
May 2025 Psychedelic Patent Update: Gilgamesh Broadens Moat Around Lead Candidate Following Positive Phase 2a Readout; Lykos Publishes Further Continuation Applications; Reconnect Joins Crowded 5-MeO-DMT Patent Landscape
Written by Psychedelic Alpha’s Noah Smith with support from Editor-at-Large and patent attorney Graham Pechenik of Calyx Law.
The Psychedelic Patent Update provides headline figures and data regarding published psychedelic patent applications and grants, as well as a brief summary of a select handful of patent-related actions.
In this Bulletin:
- Overview and Interactive Database
- Gilgamesh Pharmaceuticals Broadens Moat Around Lead Tryptamine Candidate
- Final Round of Publications from Lykos Therapeutics
- Reconnect Labs Joins Competitive 5-MeO-DMT IP Landscape
***
General Overview
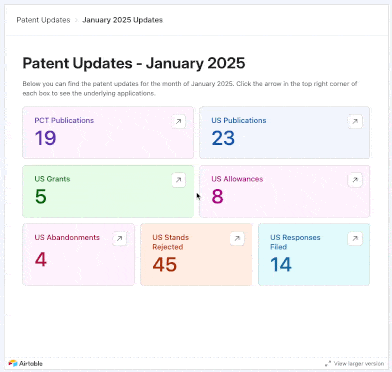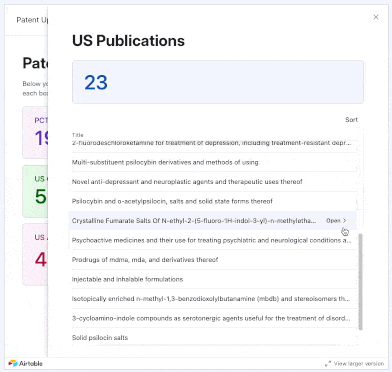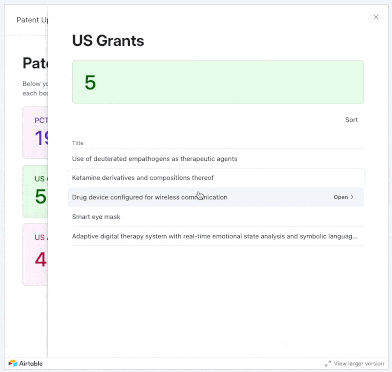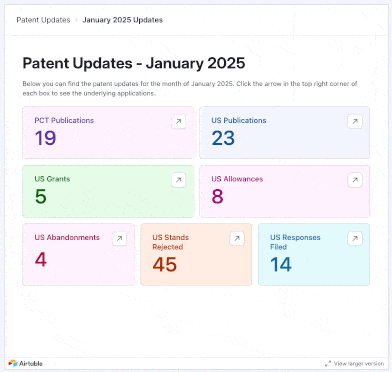
We added 17 PCTs and 34 U.S. patent applications focused on psychedelics to our tracker in May 2025. New entries came from the likes of Reconnect Labs, Mindset Pharma (acquired by Otsuka), Bexson Biomedical, Kuleon Bioscience, Lykos Therapeutics, Sage Therapeutics, and more.
Using our interface, you can explore applications that were not only published or granted in these months, but also those that were allowed, abandoned, rejected, or even instances where applicants filed responses to final and non-final rejections from Examiners.
Log-in to your Pα+ account, or join today, to explore the data and continue reading.
Join Today
Independent data-driven reporting, analysis and commentary on the psychedelics space: from business and drug development through to policy reform and research.
- Regular Bulletins featuring news, analysis, and research
- Articles and deep dives across psychedelic drug development, policy, and research
- Exclusive interviews with researchers, executives, and policymakers
- Bespoke resources and tools including the Psychedelic Bill Tracker
- Quarterly video briefings and slide decks