
A weekly round-up of the
latest coverage in psychedelics.
Receive the free weekly digest straight to your inbox every week
By signing up, you agree to our privacy policy. You can unsubscribe at any time.
📼 Watch our Q1 2026 Quarterly Briefing Video.
It’s been a very quiet week in the psychedelics field, largely owing to the U.S. holiday.
We’re taking this moment to prepare for our 2025 Year in Review, and this time we’re looking to you, our audience, to contribute. Find out more and pitch a contribution. (The deadline for submissions is Friday 5th.)
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
I just returned from a trip to Washington, D.C., where I found myself immersed in a notably optimistic psychedelics clique, with many expecting several significant developments in the next 18 months.
One of those developments has already come to fruition: ARPA-H’s $100M initiative to develop objective measures of mental and behavioural health. The initiative, which is one of the agency’s largest ever, looks set to feature psychedelic research substantially. (We were the first to cover that program’s reveal last Friday.)
There is plenty more coming down the pike, I am told, with 2026 set to be an incredibly busy year for the field. I will be writing up some of my field notes (a kind of ‘View from D.C.’) in the coming weeks, exclusively for our Pα+ subscribers.
Elsewhere this week, the largest trial of LSD microdosing to date found that it underperformed placebo across all endpoints in a population of 89 patients with major depressive disorder. The null finding is the latest blow to drug developers seeking to prove the efficacy of microdosing, and it becomes the second mid-stage low-dose LSD program to flop.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor

Psychedelic Bulletin 214
I had a great time speaking at the UW-Madison Psychedelic Symposium this week. It’s my second time attending this event, which I appreciate for its remarkably interdisciplinary agenda: there was everything from preclinical studies of psychoplastogens (Katherine Nautiyal) through to the history of touch in psychedelic practice (Zoë Dubus).
For my part, I provided a kind of ‘State of the Union’, presenting on the current state of psychedelic drug development, industry and policy, before considering some potential future directions and open questions. I was also glad to participate in a lively panel discussion. Besides being among my favourite meetings in the field, not least due to the Midwestern hospitality of the hosts and organisers, I was also grateful to have an excuse to spend some time updating my slides and adding some new ones. I hope to share an annotated copy with our Pα+ subscribers soon.
Psychedelics were also on the agenda at the ‘MAHA Summit’ in DC, a closed-door affair with a line-up including Vice President JD Vance and Health and Human Services Secretary RFK Jr. Psychedelics were discussed on stage by HHS lawyer and “psychedelics czar” Matt Zorn, who chatted with AtaiBeckley founder and chairman Christian Angermayer.
Speaking of the HHS Secretary, a new book by journalist Olivia Nuzzi, who says she had a close relationship with RFK Jr., claims that he told her he uses psychedelics and has smoked DMT.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor

We take a deep dive into the readout, which we discussed with AtaiBeckley CEO, Srinivas Rao. ∎
This week got off to a busy start with Compass Pathways announcing on Tuesday morning that it is bringing forward its launch planning by 9-12 months, following a positive meeting with FDA and the completion of enrolment in its second, larger Phase 3 study. The psilocybin developer is expected to be the first to secure FDA approval for the drug, which could come as early as late next year or early 2027.
That same day, we also learned, via an SEC filing, that AbbVie paid $900 million upfront for Gilgamesh Pharmaceuticals’ lead psychedelic candidate, bretisilocin. Until then, there had been no public disclosure of how much of the acquisition deal, which is valued at up to $1.2bn, was paid in cash. The answer, we now know, is 75%: well above what many expected. Some investors and operators in the field view this as evidence of AbbVie’s conviction around the candidate.
Also in the psychedelic drug development world, the merging of atai Life Sciences and Beckley Psytech closed this week, with the former now trading as AtaiBeckley.
Beyond the psychedelics industry, Global Psychedelic Week debuted with online and in-person programming across the world. I was pleased to participate by moderating a panel on funding, as well as having a discussion with psychedelics researcher Robin Carhart-Harris.
I am heading to Wisconsin next week for the UW-Madison Psychedelic Symposium, so please cross your fingers and toes for minimal flight disruptions! I had a great time last year and am looking forward to delivering the final presentation of the meeting on Friday afternoon. Thereafter, I have a short stint on the East Coast. I am excited to again meet with some of our readers in person.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor

Breaking: Compass Pathways Accelerates Psilocybin Launch Timeline by 9-12 Months
We take a deep dive into the news, which we discussed with Compass Pathways CEO, Kabir Nath. ∎

Psychedelic Bulletin 213
It’s been an incredibly active start to the fourth and final quarter of 2025.
In October, funding for psychedelics companies appears to have caught up with the positive sentiment exhibited in our Q3 Psychedelic Investor Survey. More than half a billion dollars was raised across three rounds alone last month (by atai, Cybin, and MindMed), meaning Q4 2025 will see the largest inflows to the space since mid-2021. There are still two months left of the quarter, meaning the heady days of 2021 could still be topped.
On the lobbying front, too, spend is ramping up. Our Psychedelics Federal Lobbying Tracker shows that psychedelic drug developers spent more than ever on U.S. federal lobbying in Q3, though the aggregate reported spend remains relatively modest at just north of $300k over the three-month period. Interestingly, Lykos Therapeutics has ceased its formal spend on federal lobbying, while Compass Pathways has more than doubled its own. What’s more, the Association for Prescription Psychedelics (which represents most of the late-stage developers in the field) began spending cash on lobbying activities in Q3, according to filings. (Pα+ subscribers can read our analysis of the latest data.)
On Friday, the Australian Government’s Department of Veterans’ Affairs announced that it has decided to fund psychedelic assisted therapy for veterans who meet certain criteria. It’s the latest in a string of psychedelics-related headlines from down under, which we will be covering in more detail very soon.
In the 212th Issue of our Psychedelic Bulletin, we looked at several other stories, including Delix Therapeutics’ efficacy signal from a small open-label study of its lead neuroplastogen and NRx Pharmaceuticals’ efforts to have FDA ban its competitors’ ketamine products.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
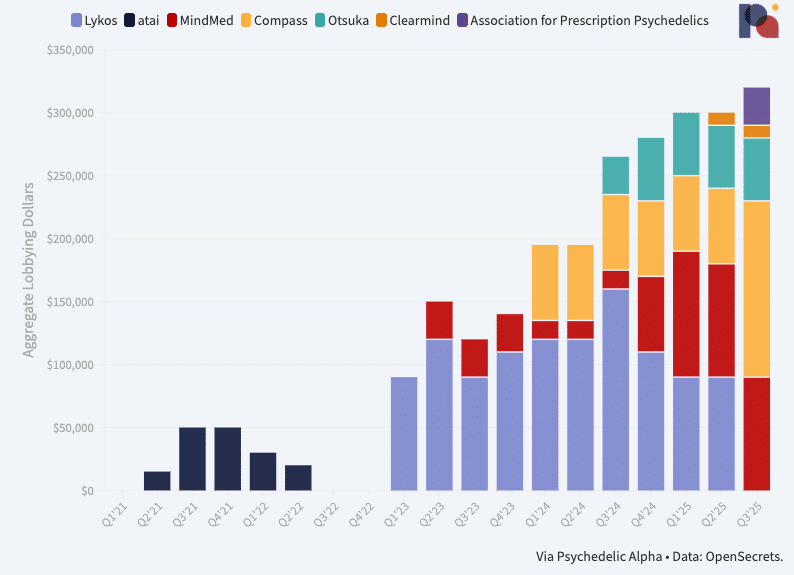
New: Psychedelics Federal Lobbying Tracker
Here, we share several charts that visualise the extent of psychedelics organisations’ lobbying activities at the federal level, at least according to filings. ∎


Psychedelic Bulletin 212
Free subscribers receive a weekly round-up of news on psychedelics, as well as the occasional article.
Receive regular bulletins, articles, interviews with insiders and quick-take analysis of major stories and developments.
Teams, groups and corporate pricing plans are available, please get in touch via email to learn more.
Front-row access to the psychedelics space: breaking bulletins, in-depth articles, insider interviews and sharp analysis of the moments that matter—from major trial results and funding rounds to policy shifts shaping the future. Plus, full access to our complete archive and Library.
Learn More| Free | ||
|---|---|---|
| Weekly Psychedelic News Feed | ||
| Occasional Articles & Free Resources | ||
| Psychedelic Bulletins (In-Depth Briefings, Multiple per Month) | ||
| Quick-Take Analysis of Major Developments | ||
| In-Depth Articles & Deep Dives | ||
| Exclusive Interviews with Insiders & KOLs | ||
| Quarterly Video Briefings | ||
| Exclusive Tools & Data Resources | ||
| Library of Primers & Explainers | ||
|
Subscribe Now |
||
Independent data-driven reporting, analysis and commentary on the psychedelics space: from business and drug development through to policy reform and research.
By signing up, you agree to our privacy policy. You can unsubscribe at any time.
Aside from group pricing, we also offer bespoke reports and regular briefings. Get in touch to discuss.