
Psychedelic Bulletin #116 – Ketamine Combo Co. Scores $10m; DEA Drops Plan to Schedule 2 Psychedelics, for Now; Analysis of Oregon Psilocybin Opt-Outs
This Week:
- 💰 Ketamine Combination Therapy Startup Freedom Biosciences Raises $10m Seed
- ⚖️ DEA Abandons Plan to Schedule 2 Psychedelics, for Now
- 🗺️ Analysis: Majority of Oregon Counties Will Send Psilocybin Opt-Out to November Vote, but Maps Can Be Misleading
- 🍺 New Study Shows Promise of Psilocybin for Alcohol Use Disorder (more analysis next week)
and more…
Psychedelic Sector News
New Study Shows Promise of Psilocybin for Alcohol Use Disorder
We stopped the presses to write this one up, but decided it deserved more space than this week’s Bulletin allows for. So, expect our commentary on this study to feature in our next Bulletin.
The TL;DR is: Psilocybin-assisted psychotherapy “produced robust decreases in the percentage of heavy drinking days compared with those produced by active placebo and psychotherapy.”
This is the largest psilocybin trial to be published, despite it being smaller than investigators had originally planned. If you’re keen to get a head-start you can read the journal article here.
The study also received a great deal of media attention, which might help you get a foothold before our deep dive:
- Smithsonian: Psychedelic ‘Magic Mushroom’ Ingredient Could Help Treat Alcohol Addiction
- NBC News: Psychedelic drug helped people with alcohol use disorder reduce drinking, study shows
- The Verge: Psychedelics help people with alcoholism drink less
- NYU Langone: Psychedelic Drug Therapy May Help Treat Alcohol Addiction
- ABC News: ‘Magic mushroom’ psychedelic may help heavy drinkers quit
- Axios: Psychedelics with psychotherapy helped heavy drinkers cut back, study finds
- WSJ: Psilocybin, Psychedelic Compound in Magic Mushrooms, Shown Effective for Alcohol Addiction
- Science Alert: ‘Magic Mushroom’ Psychedelic May Help Ease Alcohol Addiction, New Results Hint
- Morning Brew: Psychedelic drugs treat alcoholism in new study
Ketamine Combination Therapy Startup Freedom Biosciences Raises $10m Seed
Freedom Bio is the latest psychedelics startup on the block, having raised over $10m in a seed round led by MBX Capital. Perhaps we should call it a psychedelic-like startup, as its lead development program is a combination therapy with ketamine to treat major depressive disorder. The company claims its candidate will extend the antidepressant effects of ketamine.
We don’t always cover ketamine companies, but this one is quite interesting given its drug development focus (as opposed to being a ketamine clinic, for example) and esteemed co-founders, one of which is Yale Psychiatry chair John Krystal. Our Advisor Michael Haichin takes a closer look at the research that he presumes is underlying this startup…
Based on the reporting of the seed financing, from John Krystal’s involvement to mention of extending ketamine’s antidepressant effect, Haichin and others have speculated that Freedom Biosciences’ lead program, FREE001, is a combination of ketamine and rapamycin.
Ketamine and other potential rapid-acting antidepressants are thought to exert antidepressant effects by starting a signaling cascade eventually leading to the activation of the mechanistic target of rapamycin complex 1 (mTORC1) and subsequent increased formation of synapses between neurons. Also known as sirolimus, rapamycin is a medication most commonly used to suppress the immune system and is an inhibitor of the mechanistic target of rapamycin complex 1 (mTORC1).
In 2020 John Krystal and colleagues from Yale published the results of a trial (n=20) examining the combination of ketamine and rapamycin, testing the hypothesis that rapamycin pretreatment would block ketamine’s antidepressant effects. Paradoxically, the combination prolonged the antidepressant effect up to 14 days compared to ketamine and a placebo. These results raised several questions at the time of publication and warrant further replication due to the small number of participants, however the potential to extend the antidepressant effects and decrease the frequency of redosing is undoubtedly appealing.
The same publication indicated the lead authors Chadi Abdallah and John Krystal filed a patent application for using mTORC1 inhibitors to augment the effects of antidepressants on August 20, 2018. That application, entitled “Combination Therapy for Treating or Preventing Depression or Other Mood Diseases” was first published as PCT Pub. No. WO2020/041329A1 and is currently awaiting examination in the U.S. as Appl. No. 17/269,470. An international preliminary report on patentability found all claims novel, but none inventive, largely finding the claims obvious over Yang 2018, “Mechanistic Target of Rapamycin–Independent Antidepressant Effects of (R)-Ketamine in a Social Defeat Stress Model” (notably, work from Kenji Hashimoto’s Chiba University lab, the IP from which Perception Neuroscience has an exclusive licence). Pending U.S. claims are broadly drawn to methods “of treating or preventing a mood disorder” by administering “a rapid-acting antidepressant (RAAD)” together with an mTOR inhibitor or immunosuppressant. Disclosed examples of RAADs include NMDA receptor modulators and ketamine, R-ketamine, and S-ketamine specifically, and examples of mTOR inhibitors include rapamycin. The lone example in the application discusses the results of the published trial and concludes: “Rapamycin was found to enhance the antidepressant effect of ketamine, and thus combinations of ketamine and rapamycin or other potent immunosuppressant approaches can be used to enhance the rapid antidepressant effects of ketamine.”
The company plans to enter the clinic next year.
DEA Abandons Plan to Schedule 2 Psychedelics, for Now
The Drug Enforcement Administration (DEA) has backpedalled on another attempt to schedule psychedelics, this time DOI and DOC.
In spring, the DEA announced its intention to place DOI and DOC, two phenethylamine psychedelics, in Schedule I of the Controlled Substances Act. Nearly 100 public comments were submitted in response to the proposal, the vast majority of which opposed the DEA’s intended course of action.
Some public comments suggested that scheduling these two drugs would further entrench the War on Drugs, which one individual said would be at odds with “the current cultural climate of the country”. These commenters often also note the low levels of abuse by the general public of DOI, in particular.
Others focused on the damage that the proposed scheduling would have on research, given that DOI is used extensively in the study of 5-HT2 receptors. The regulatory hurdles that Schedule I status would place on researchers looking to use these phenethylamines would also hinder federally funded research.
SSDP’s University of Michigan chapter submitted a comment along these lines, as did prominent researchers like Charles Nichols. A letter submitted by Harrison Elder was co-signed by a number of other researchers including Alaina Jaster, Frederick Barrett, Matt Baggott, and Boris Heifets. Another letter, submitted by Gilgamesh Pharmaceuticals co-founder and Columbia scientist Mike Cunningham, was co-signed by other psychedelic researchers: from Small Pharma’s Carol Routledge through to Heffter’s Dennis McKenna. It’s even signed by a couple of NIDA researchers. Notably, these researchers tend to focus on DOI, with DOC not even mentioned in many submissions.
Beyond public pressure—which early-career researchers including Alaina Jaster were largely responsible for mustering—Panacea Plant Sciences’ David Heldreth made a formal filing to oppose the scheduling, as he did in the recent case of the five tryptamines.
In a document filed on the 26th August, the Administration announced that it is withdrawing the proposed rule, terminating all proceedings.
However, the DEA has noted that it “is planning to publish a new proposed rule with an amended procedure.” Perhaps this withdrawal is the product of a procedural quirk as opposed to a change in attitude from the agency.
Interested in these recent challenges to the DEA’s scheduling of psychedelics? Read our 8,000-word Special Report: Inside the Challenge to DEA’s Proposed Scheduling of 5 Psychedelic Tryptamines.
Note: Just as we were wrapping this section, Matthew Baggott shared his (characteristically insightful) thoughts in a mini Twitter thread…
I think this victory may indicate a big change in how DEA handles psychedelics. What they do next will show how they plan to regulate psychedelics. DEA will come back with a new proposal to schedule DOC, and I wouldn’t be too surprised if it’s Schedule IV. https://t.co/Tar1G9ODyt
— Matt Baggott (@mattbagg) August 29, 2022
Analysis: Majority of Oregon Counties Will Send Psilocybin Opt-Out to Public Vote in November, but Maps Can Be Misleading
As the Oregon Psilocybin Services Opt-Out landscape firms up after a filing deadline and ahead of the November 2022 vote, we take a step back to review the situation…
Refresher: Oregon Psilocybin Services Act Opt-Outs
Oregon’s Psilocybin Services Act (Measure 109) allows for local jurisdictions to vote to opt-out of allowing psilocybin services within their borders. Under Measure 109, local jurisdictions are automatically opted-in unless they elect to adopt an opt-out ordinance and refer it to local voters on the November 8, 2022 general election. Our map tracks the status of Measure 109 in these jurisdictions.
Incorporated city land is subject to city regulation. Unincorporated land outside of incorporated city limits is subject to county control. As a result, a county’s vote to opt-out only applies to its unincorporated areas; cities within such counties could choose to remain opted in.
Even where a jurisdiction does not opt-out, land use analysis for real estate is a fact-intensive process, implicating local zoning and time, place, and manner restrictions.
Our map is produced in collaboration with Emerge Law Group, Calyx Law. With special thanks to Healing Advocacy Fund for additional on-the-ground data.
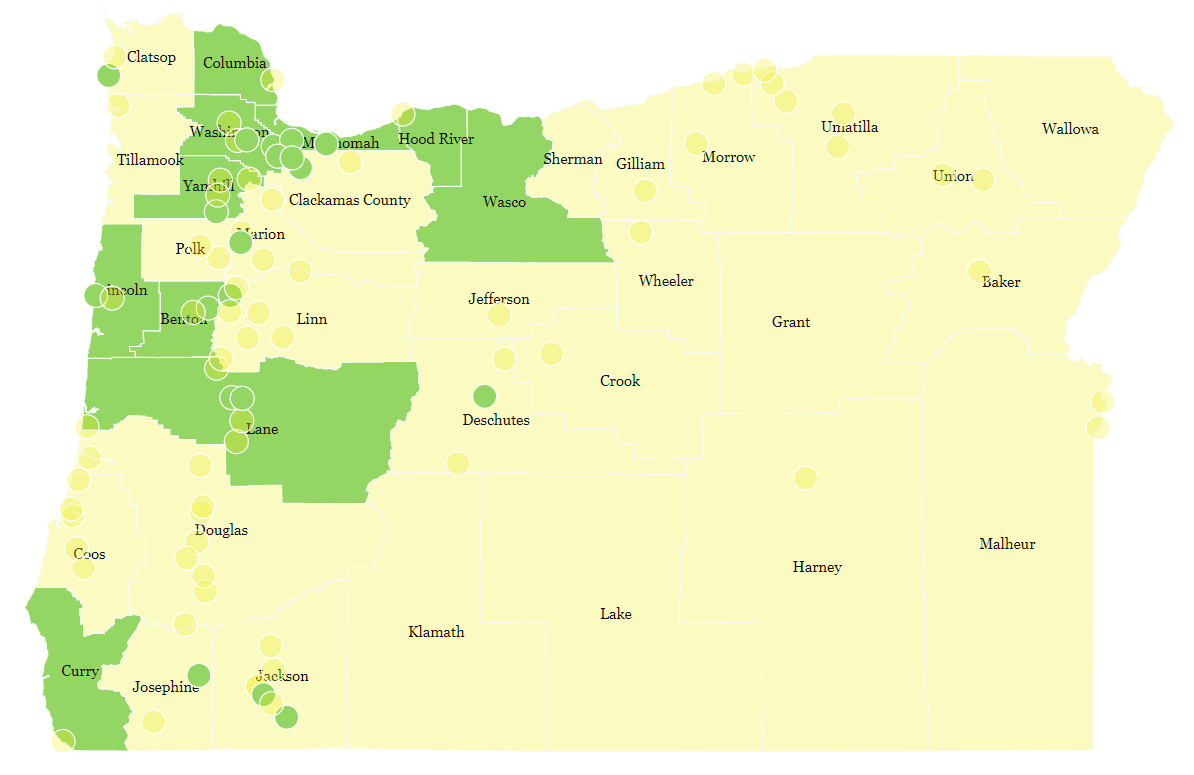
The map’s pretty yellow. But Oregon’s counties vary a great deal when it comes to their populations (more on that later), so let’s look at the five most populous ones to begin with.
With a combined population of around 2.5 million, these five counties represent just over 60% of Oregonians.
Three of the five counties will not have an Opt-Out Ordinance on November’s ballot, meaning Psilocybin Services should go ahead unhindered. Notably, this includes Multnomah and Washington, the two most populous counties.
At a statewide level, some trends can be observed; many of which might be predictable to those with even a cursory understanding of the State’s demographic and party political cleavages…
There’s clear correlation between those counties that will have Opt-Out Ordinance on the November ballot and their party political leanings. Counties like Multnomah, Lane and Benton are all firmly Democratic, while counties like Klamath, Lake and Harney are Republican strongholds.
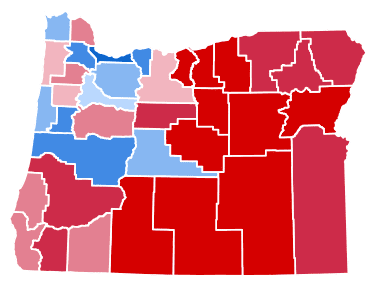
These political cleavages are geographic, too, with the most southern and eastern counties almost entirely Republican, and moving to put a potential Opt-Out of psilocybin services to a public vote.
But, as is common, these more rural, Republican counties are much less densely populated. To illustrate this point, we cleaved off 7 of the most Eastern counties of Oregon. Together, this ‘Eastern bloc’ represents around a third of the landmass of Oregon.

However, counties like Grant, Harney and Wallowa have four figure populations. Together, these seven counties have a population of around 175,000 people: just over a fifth of the population of Multnomah.
As such, our map is visually quite misleading in showing such large swathes of yellow, or counties and cities putting potential Opt-Outs in the hands of their voting-age residents this November. If we adjusted the size of counties on the map by their population, the picture would be much different.
What’s next?
Our Opt-Out Tracker should be relatively stable in the coming weeks and months, as the deadline for sending any Opt-Out Ordinances to the Oregon Secretary of State was August 19 (there may be some minor updates, likely owing to delays in updating public information).
After that, we will be covering the election results in November.
Featured Psychedelic Jobs
- MycoMeditations is hiring a Lead Therapist.
- Sunstone Therapies is hiring a Learning Manager in Maryland, U.S.
- Psylo is hiring a Lead Computational Chemist.
Browse more roles and get more job posts to your inbox by signing up for alerts here.
Weekend Reading
Assorted Reading
- Marc Gunther’s latest for Lucid News explores the legal questions that linger as plant medicine churches proliferate.
- A new survey from Johns Hopkins finds that attitudes about death change after psychedelic experiences as well as out-of-body experiences. The university wrote-up the research in plain English here. TIME also covered the study, among others.
- A podcast from the Irish Times speaks to researchers involved in local psilocybin trials.
- Rachel Yehuda discusses how the VA is looking at treating mental health issues with psychedelics in a podcast with Federal News Network.
- A Nature Outlook piece explores how psychedelic drugs might provide relief for those who don’t respond to existing therapies, asking: “does the hype outweigh the hope?”
- USA Today asks, “Can psilocybin combat mental health issues?”
- The Guardian explores “the Black mothers finding freedom in mushrooms”.
Weekly Bulletins
Join our newsletter to have our Weekly Bulletin delivered to your inbox every Friday evening. We summarise the week’s most important developments and share our Weekend Reading suggestions.
Live Updates
Join us on Twitter for the latest news and analysis.
Other Channels
You can also find us on LinkedIn, Instragram, and Facebook.
Join thousands of psychedelics insiders
Get the weekly psychedelic medicine briefing.
A free weekly digest covering trials, regulation, policy, and access.
By signing up, you agree to our privacy policy. You can unsubscribe at any time.