
Single Dose of Psilocybin Reduces Migraines for at Least 2 Weeks
New research from Yale finds that a single low dose of psilocybin reduces the frequency of migraine headaches for 2 weeks.
The study, published Thursday 12th November, sought to add empirical rigour to anecdotal evidence suggesting that a low dose of psilocybin (and other 5-HT2A agonists) may have an enduring positive effect on headache disorders.
To this end, the research team undertook a double-blind, placebo-controlled cross-over study in which 10 adult migraine sufferers received either psilocybin or placebo.
The psilocybin dose in this study is low, at 0.143mg/kg. As such, an adult weighing 75kg would receive around 10 mg of psilocybin.
Doses for psilocybin-assisted therapy, in contrast, are much larger. In their seminal study of dose-related effects, Griffiths et al. (2011) evaluated psilocybin administered in doses up to 30mg (per 70kg). When studying psilocybin-assisted therapy for treatment-resistant depression, Carhart-Harris et al. (2016) administered two doses seven days apart, with the larger dose being 25mg.
The low dose of psilocybin was delivered in 2 sessions, with a 2-week interval between. Participants recorded the incidence of migraines in headache diaries, with entries beginning 2 weeks prior to the first session, and ending 2 weeks following the second session.
Ultimately, the study found that the reduction in the frequency of migraine days after receiving psilocybin was significantly larger than placebo.
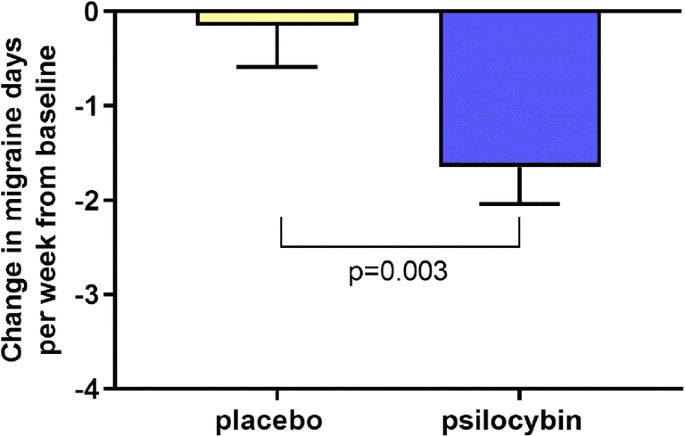
The study reports that psilocybin was well-tolerated by all participants, and that the effect size was not correlated with the intensity of psychotropic effects during psilocybin adminsitration.
It’s important to note that this was a small (n=10), exploratory study. Yet, its results are promising and pave the way for further research.
This new research contributes to a growing body of evidence suggesting that the acute psychotropic effects of 5-HT2A agonists, namely psilocybin, are not the sole vehicle for therapeutic benefit. Rather, it appears that psilocybin can have an enduring therapeutic benefit that is distinct from its psychedelic effect, demonstrating significant scope for further research.
References
Clinical Trial NCT03341689
Carhart-Harris, R. L., Bolstridge, M., Rucker, J., Day, C. M. J., Erritzoe, D., Kaelen, M., … Nutt, D. J. (2016). Psilocybin with psychological support for treatment-resistant depression: an open-label feasibility study. The Lancet Psychiatry, 3(7), 619–627. doi:10.1016/s2215-0366(16)30065-7
Griffiths, R. R., Johnson, M. W., Richards, W. A., Richards, B. D., McCann, U., & Jesse, R. (2011). Psilocybin occasioned mystical-type experiences: immediate and persisting dose-related effects. Psychopharmacology, 218(4), 649–665. doi:10.1007/s00213-011-2358-5
Schindler, E. A. D., Sewell, R. A., Gottschalk, C. H., Luddy, C., Flynn, L. T., Lindsey, H., … D’Souza, D. C. (2020). Exploratory Controlled Study of the Migraine-Suppressing Effects of Psilocybin. Neurotherapeutics. doi:10.1007/s13311-020-00962-y
Join thousands of psychedelics insiders
Get the weekly psychedelic medicine briefing.
A free weekly digest covering trials, regulation, policy, and access.
By signing up, you agree to our privacy policy. You can unsubscribe at any time.