
The Obscure Advisory Panel Blocking Dozens of Psychedelic Studies in California
Dozens of new studies involving Schedule I and II substances—many of which employ psychedelics—are unable to commence in California, despite having clearance from the FDA, DEA and Institutional Review Boards.
The cause of the hold-up? An obscure—yet apparently mandatory—checkpoint in the path to study initiation: the Research Advisory Panel of California (RAP-C).
California is simultaneously the state with the most clinical trials in the U.S., representing over a quarter of registered trials in the country1, while also being the only state to feature such a panel, which may be becoming its Achilles’ heel.
Here, look at what RAP-C is, how it operates, its current impasse, and potential paths forward.
***
Editor’s note: shortly after this piece published, Psychedelic Alpha received the following comment from the Attorney General’s Office:
The Attorney General’s Office has historically provided administrative and legal support to RAPC. The Attorney General’s Office is now working with the Governor’s Office and the Legislature on a legislative solution to the procedural issue that has impacted RAPC’s meetings this year. That issue is the potential application of the Bagley-Keene Open Meeting Act to RAPC’s meetings. If legislative efforts are successful, this issue could be resolved by early 2024.
RAP-What?
The Research Advisory Panel of California (RAP-C) was established as the California Research Advisory Panel in 1965 under California law (specifically, Health and Safety Code sections §11480 and §11481).
In some respects, it was established to encourage and coordinate further research into cannabis and psychedelics; at least according to the opening sentence of §11480.
Its material purpose, though, is to “hold hearings on, and in other ways study,” research projects investigating “the nature and effects of cannabis or hallucinogenic drugs”, as well as “research projects concerning the treatment of abuse of controlled substances.”
Crucially, the panel is afforded the authority to approve such research projects, as well as withdraw its approval “at any time”.
According to the Attorney General, investigators or sponsors must submit applications to the panel for any research projects planned to be conducted in California where:
- A Schedule I or Schedule II controlled substance is the main study drug or a comparison drug for human studies.
- A Schedule I controlled substance is used in non-human studies.
- Any controlled substance use disorder treatment research employs any scheduled or non-scheduled medication.
Sponsors should submit their research projects to the panel only after they have received all other necessary approvals, such as those from the Drug Enforcement Administration (DEA), Food and Drug Administration (FDA, such as Investigational New Drug (IND) applications), Institutional Review Boards (IRBs), Data and Safety Monitoring Boards (DSMBs), and so on.
In effect, the panel is the last hurdle investigators or sponsors must pass before their studies are approved for initiation.
Once a proposal is submitted, the panel may approve or reject it, as well as propose amendments.
The panel consists of five members, including researchers who have substantial experience investigating scheduled substances including psychedelics, such as Boris Heifets and Jennifer Mitchell. Members are unremunerated volunteers.
While the Panel’s substantive activity is reviewing relevant research projects, it has occasionally issued proposals of its own, including ones on drug policy2.
RAP-C Reviews All Psychedelics Studies in California
During the 2022 calendar year, RAP-C reviewed 54 applications for research, 36 of which it approved, 3 were not approved or withdrawn, and 15 were pending finalisation at year end (see figure). 18 “substantially amended studies” were also approved during the 2022 calendar year. It’s not immediately clear what these amendments looked like, or whether the “not approved or withdrawn” studies were nixed due to RAP-C’s concerns.
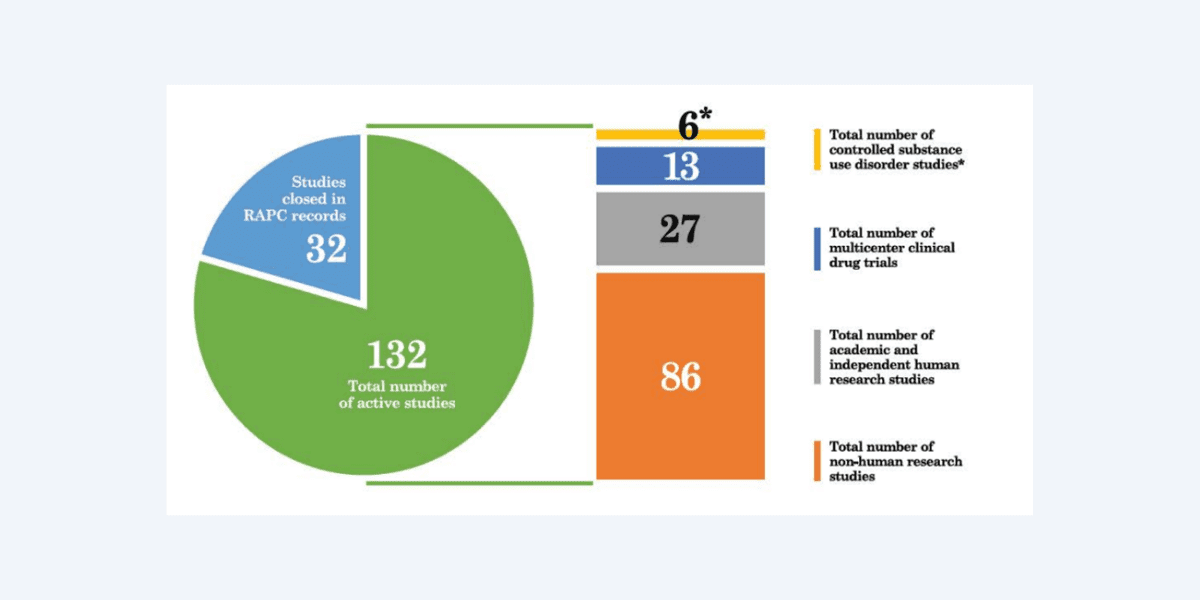
According to our analysis, 14 of the 36 (around 39%) research studies approved in 2022 sought to primarily investigate psychedelics. These include Compass Pathways’ psilocybin therapy in anorexia nervosa proof of concept study, MindMed’s Phase 2 study of LSD for the treatment of anxiety symptoms, and UCSF’s comparison study of oral and sublingual psilocin which uses Filament Health’s candidates.
Amendments to studies from the likes of Compass and MAPS PBC (now Lykos) were also approved in 2022.
Ongoing studies, according to 2022’s annual report, include work on psilocybin therapy for phantom limb and chronic lower back pain; MDMA-assisted therapy for PTSD; psilocybin’s effects on visual perception; psilocybin for depression and anxiety in Parkinson’s disease; and more.
The Present Impasse
Later on, we look at how lead times for RAP-C approval are already lengthy. But that’s not the cause of the present, total inability to commence new Schedule I and II research (and substance use disorder studies more generally) in the state.
The current blockage, and backlog, is due to the Panel’s apparent inability to meet.
The final two meetings of the 2023 calendar year, scheduled for October 27th and December 8th, were cancelled; with applications submitted up to September 1st, 2023, languishing in bureaucratic limbo.
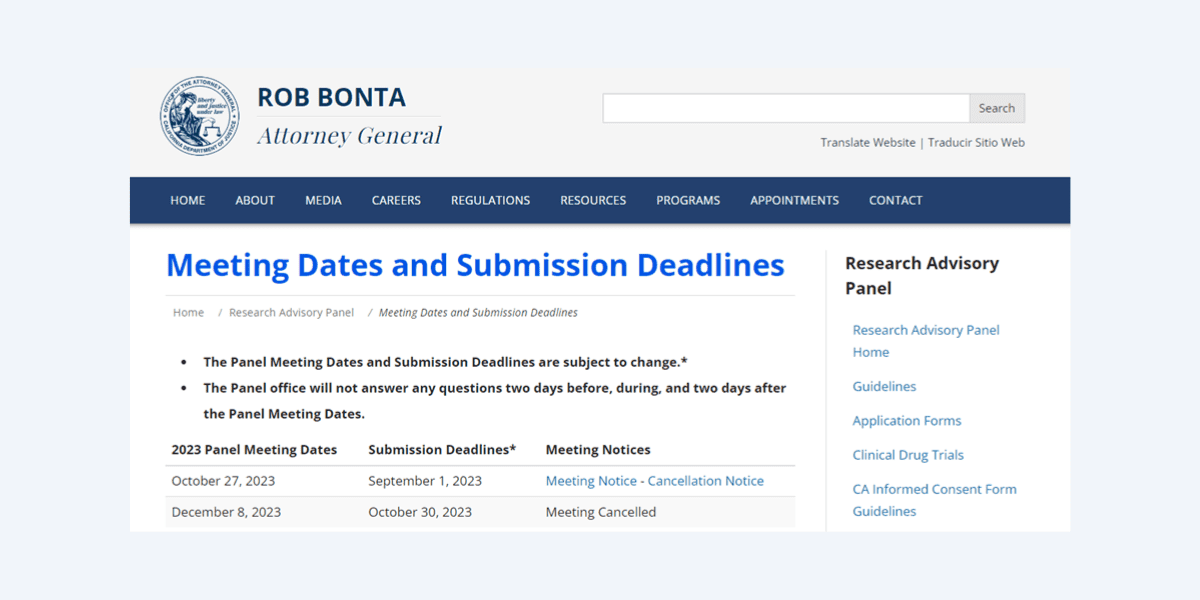
Given that the panel only meets six times a year, these two unexplained cancellations have wiped out a third of 2023’s approval window, with dozens of studies on hold before they have even commenced. In the case of human studies, research subjects cannot be screened or enrolled until approved.
The two-sentence Cancellation Notice for the October 27th meeting provides no details on why the meeting was scrapped. Psychedelic Alpha reached out to the Panel’s administrative contact multiple times over the course of two weeks, but did not receive a response.

The Meeting Agenda, meanwhile, gives some insight into the types of research that are held in limbo.
Thirteen studies involving controlled substances are listed, which hope to investigate “the nature and effects of cannabis or hallucinogenic drugs”. The vast majority of applicants are academic institutions, such as UCSF, UCLA and Stanford.
There are two drug developers in the mix, too. One is Beckley Psytech, which is perhaps hoping to add a California study location to a Phase 2 study of its 5-MeO-DMT candidate (BPL-003) for treatment-resistant depression (TRD). Another is Herophilus, though it’s not clear which of its candidates might be the subject of the RAP-C application.

In terms of studies that seek to evaluate potential treatments or interventions for substance use disorders, two studies sponsored by the National Institute on Drug Abuse (NIDA) were due to be reviewed, and one from Cari Health, which is “developing the next generation of Remote Medication Monitors”.
There is no Cancellation Notice for the December 8th meeting. Instead, the Notices column simply reads “Meeting Cancelled”. We can only assume that a similar number of studies might have been submitted to the Panel in the meantime.
Psychedelic Alpha understands that there are dozens of studies indefinitely prevented from commencing due to these meeting cancellations, with a substantial portion of these involving psychedelics.
The Cause? Bagley-Keene
Why might these meetings have been cancelled so abruptly, neutering the Panel’s primary purpose and slamming the brakes on dozens of new studies in California?
A number of sources told Psychedelic Alpha that the issue lies in the Bagley-Keene Open Meeting Act, a state body equivalent of the Brown Act that generally requires state boards and commissions to conduct and service meetings in public (unless the Act specifically allows them to meet in closed session).
According to sources close to the matter, recent changes to Bagley-Keene3 (which was first enacted in 1967 but underwent a number of amendments to account for increases in meeting remotely during the pandemic), or its interpretation by the current attorney General’s office, have led to incongruencies with the Panel’s methods and the wishes of sponsors and investigators4.
At the sharp end, RAP-C regularly reviews proprietary documents—such as study protocols—that a sponsor may not wish to become public.
There’s also the matter of scientific review, which is conventionally undertaken anonymously.
Bagley-Keene, then, appears to compromise both sponsors’ and academics’ expectation of privacy, and in turn could compromise not only the work of the volunteer Panel (who may no longer wish to scrutinise their peers’ work in public) but also make sponsors (especially industry) unwilling to risk their proprietary plans seeing the light of day.
Beyond the Blockage: What’s RAP-C’s Purpose?
But even without the current gridlock, those that Psychedelic Alpha spoke to raised more fundamental issues with California’s unique appendage.
The application process itself requires at least nine constituent elements for human research.
Granted, much of this information will already be at hand, as it’s likely been submitted to an IRB or as part of an IND, and so on. But this begs the question: what’s the purpose of this Panel? If all of the constituent elements of the RAP-C application—such as IRB- and FDA-approved Research Protocols and Consent Form Checklists—have been reviewed and approved by other bodies, RAP-C is presumably a rubber-stamping process.
While it likely served a purpose in the past, Psychopharmacology researcher Joshua Woolley, speaking in a personal capacity, told Psychedelic Alpha, it’s hard to parse out an additional benefit of the Panel today, given that we now have IRBs, DEA, FDA, DSMBs, and so on.
Daniel Kelly, Director of the Pacific Neuroscience Institute, agreed that there is already a substantial (“extreme”, in his words) level of oversight for clinical trials assessing Schedule I compounds without RAP-C, citing the same types of agencies, bodies and boards as Woolley.
Assistant Clinical Professor at UCSF, Brian Anderson, supposed that, where human research with controlled substances has already undergone “rigorous scientific review” by federal funding agencies like the National Institutes of Health (NIH), additional scientific review by RAP-C “may provide only a marginal benefit”.
In these cases, Anderson suggested that RAP-C “should perhaps only perform a rubberstamp function of certifying that a sufficient review was already completed”.
In many cases, then, it appears RAP-C is duplicating efforts, given that studies may have been reviewed by agencies like DEA, and in some cases funding bodies like NIH.
Researchers Psychedelic Alpha spoke to said that this wouldn’t be a major issue in itself. But those rubber stamps and back-and-forth take time, and money.
“It has become a major impediment to the launching of psychedelic-assisted therapy clinical trials for mental health disorders in California”, Dr. Kelly told us, referring to just one type of study that falls under the auspices of RAP-C.
The Price of a Rubber Stamp
“A response can be expected within 30 business days after the Panel meeting”, according to the Attorney General’s office. That’s up to six weeks without including the potential gap between submission and the next RAP-C meeting. Given that the Panel meets six times per year, there’s a baked-in limit as to how quickly it can respond to proposals.
The submission deadline can be up to 7 weeks before the meeting date, meaning the actual time between submitting an application and receiving a response can be more than a few months and often around a half-year according to those that have been through the process; assuming those meetings aren’t cancelled.
Researchers told Psychedelic Alpha that they’re increasingly expected to run studies in 12-18 months, meaning these delays are substantial.
Should RAP-C propose amendments to the research proposal, further delays ensue5.
In the case of a study of MDMA-assisted therapy for anxiety associated with life-threatening illness, RAP-C requested “several changes to the protocol and Informed Consent form”, according to a MAPS Bulletin from 2014.
The study was approved by RAP-C on December 15th, 2014, three months after the study achieved IRB approval. Having tangled with RAP-C multiple times, it’s perhaps not surprising that MAPS’ founder, Rick Doblin, told us that the Panel is “more appropriately known by its former name”, referring to the acronym of California Research Advisory Panel 6.
Woolley, meanwhile, told us that RAP-C review has always caused ‘many months of extra delays’, postponing the commencement of studies.
It doesn’t look like these months-long delays to research commencement are new, either. A letter dated February 8th 1977 to Alexander (‘Sasha’) Shulgin demonstrates relatively slow interactions. Shulgin had promptly submitted an amendment to his research protocol on December 7, 1976, just two weeks after the Panel had sent him a letter. Two months later, the Panel denied his research protocol, citing “major deficiencies” and withdrew its approval of an earlier study 7 8. (Interactions with the Panel were described in TiHKAL by Shura, which is widely regarded to be a pseudonym for Sasha9.)
Shulgin’s research wasn’t the only to come under scrutiny by the Panel. Speaking to the PsychedeRx podcast, Mark Geyer, UC San Diego Distinguished Professor of Psychiatry and Neurosciences Emeritus, explained how his Department Chair Arnold Mandell was dosing mescaline to pigeons in the mid-1970s, with a DEA license in hand.
Despite their DEA-licensed bona fides, Geyer recalls a cease and desist letter arriving at UC San Diego ordering them to stop their study10. That letter came from the Research Advisory Panel which, according to Geyer, many academics didn’t even know existed at the time.
Once an application is approved, investigators or sponsors must also submit an annual progress report to the Panel in order to maintain approval of the project11. In this capacity, RAP-C monitors ongoing, approved projects.
At the end of the 2022 calendar year, RAP-C was monitoring 132 research projects12. For multi-site studies, the sponsor or contract research organisation (CRO) is responsible for the report’s submission, not the California trial site itself.
These delays and added administrative burden result in substantial costs. According to Woolley, these costs can add up to something in the region of $100,000 – $250,000 per study reviewed.
Given that studies submitted to RAP-C are supposedly cleared by all other agencies and boards, in many cases the disbursement of study-related overhead such as researcher and staff costs has already begun.
That money is not only coming from industry sponsors and philanthropy, but also millions of taxpayer dollars.
Our Reporting, Powered By You
This is the first coverage of RAP-C’s current impasse and how it’s impacting psychedelic research. Our reporting is made possible by Pα+ subscribers.
Consider subscribing to Pα+ to receive all of our content and resources. Your subscription goes directly toward supporting our coverage, not glossy graphic design or marketing budgets. Learn more and subscribe here, or get in touch to discuss team and corporate plans. Thank you.
The Fallout
The fallout from the present hold-up is simple: new human studies involving Schedule I and II substances—along with non-human studies involving Schedule I substances and those that investigate substance use disorders—cannot commence until the issue is resolved.
But the researchers we spoke to suggested that RAP-C, even without the current impasse, is damaging both clinical and basic research in California.
“The unfortunate reality for California psychedelic researchers is that the longer this RAP-C dysfunction continues, the more likely our clinical research teams will be sidelined” – Daniel Kelly
Taken together, they say, the time and costs associated with RAP-C might disincentivise sponsors from basing studies, and trial sites, in the state; which affects investigators and Californians alike. What’s more, sponsors of multi-site studies may skip the state, due to the aforementioned delays and reporting requirements (which fall on the sponsor, according to RAP-C rules).
“The unfortunate reality for California psychedelic researchers is that the longer this RAP-C dysfunction continues, the more likely our clinical research teams will be sidelined from contributing to critically important Phase 1, 2 and 3 clinical trials for medicines such as psilocybin, LSD and 5-MeO-DMT”, Kelly told Psychedelic Alpha.
This is especially concerning for the field given that California represents such a large portion of clinical trial sites in the US.
This should all be understood in the context of an increasingly competitive (psychedelic) research and drug development milieu, where studies are expected to be completed in under two years and where there are exponentially more appropriate trial sites available than there were a decade ago.
This isn’t just an issue with industry-sponsored studies, either. Psychedelic Alpha heard from researchers whose research proposals were rejected by NIH, with RAP-C delays presumed to be the cause.
Woolley told us that several philanthropy-supported trials are stuck awaiting RAP-C approval for ‘several months’.
Next Steps
When it comes to solutions, all of those we spoke to agreed that the immediate priority is to get the Panel sitting once more and clearing the backlog.
Keith Heinzerling, Director of the TRIP Center at Pacific Neuroscience Institute, told us that “the only urgent time-sensitive matter is to get RAP-C working again as soon as possible, clear the backlog of studies waiting for approval, and be ready for the new wave of studies that will be submitted soon.”
According to sources close to the matter, the hold-up is hoped to be resolved this month. This could be achieved via an exemption of RAP-C from Bagley-Keene (or a re-interpretation of the statute), or by allowing it to hold its meetings in closed sessions13.
“The only urgent time-sensitive matter is to get RAP-C working again as soon as possible, clear the backlog of studies waiting for approval, and be ready for the new wave of studies that will be submitted soon.” – Keith Heinzerling
Once the hold-up is resolved and the backlog cleared, there doesn’t appear to be a consensus on the future direction of RAP-C.
Several of the researchers we spoke to would like to see a multi-stakeholder review, which would aim to re-align RAP-C with the stated aim of encouraging further research into scheduled substances in the state.
Some of the researchers we spoke to called for the nuclear option: total dissolution of the Panel14 and deference to the likes of the DEA, FDA and IRBs. Historically, some have opted to ignore the Panel altogether, though this does not appear to be a reliable strategy15.
Others would be content with seeing the Panel explicitly classified a rubber stamping agency, which would only serve to confirm whether approvals were received by the aforementioned agencies and boards. But, unless the Panel’s schedule were also changed to allow much more frequent meetings, it’s difficult to see how this wouldn’t still lead to delays.
Some, however, maintained that RAP-C plays an important role and should not necessarily be dissolved.
Anderson, for example, told Psychedelic Alpha that relatively few IRBs have in-depth knowledge of the risks associated with psychedelics, or experience in reviewing psychedelic therapy clinical trials at all. Indeed, Anderson said that his experience in submitting human psychedelic study protocols to IRBs at three different University of California campuses revealed “variability” in the IRBs’ “level of familiarity and comfort with psychedelics”.
In lieu of such experience or finalised Guidance for Industry from the FDA, Anderson suggests that RAP-C “has provided somewhat of a layer of stabilization and standardization over psychedelic research in California over the past several years.”
Outside of human research, Anderson suggested RAP-C’s review of non-clinical research with Schedule I drugs could serve “an important quality assurance check given that some basic science research with psychedelics … does not require any IRB or FDA review at all.” In these scenarios, Anderson said, RAP-C “might be the only scientific review that stands between a potential manufacturer and a DEA research registration.”
Whether California will be able to turn RAP-C from the Achilles’ heel of scheduled substance research into its unique selling point is yet to be seen. Indeed, Governor Newsom promised that California “will be on the front-end of leading” psychedelic research when he vetoed a decriminalization bill last October.
For now, all eyes are on whether the Attorney General’s Office can reach a solution to the current impasse and release dozens of research projects—including psychedelic trials—from regulatory limbo.

Josh Hardman
Josh has been writing, talking and working on the business, science and policy of psychedelics since he launched Psychedelic Alpha in early 2020. You can connect with him on LinkedIn or via email.
Stay Informed in 2024 with Pα+
This free article—the first to cover RAP-C’s impact on psychedelic research—is made possible by our paid subscribers, who gain access to our Pα+ exclusive content.
Subscribe to Pα+ to receive all of our Bulletins, deep dives and analysis, as well as access to the Pα+ Library and other subscriber-only content.
Your subscription goes directly toward supporting our coverage, not glossy graphic design or marketing budgets. Learn more and subscribe here, or get in touch to discuss team plans.

Join thousands of psychedelics insiders
Get the weekly psychedelic medicine briefing.
A free weekly digest covering trials, regulation, policy, and access.
By signing up, you agree to our privacy policy. You can unsubscribe at any time.
- According to the U.S. National Library of Medicine: ClinicalTrials.gov
- In its 1990 report, the California Research Advisory Panel made recommendations about drug policy, including to permit the possession of syringes and needles, to permit the cultivation of marijuana for personal use, and to forbid the sale or consumption of alcohol in hospitals and schools. See This Date in UCSF History: Professors Stir Drug Debate (Synapse).
- Indeed, the cancelled October 27th, 2023 meeting had Bagley-Keene Open Meeting Act Training as agenda point four.
- RAP-C isn’t the only body that appears to be struggling to reconcile Bagley-Keene with its usual operating style. “I am well acquainted with the frustration that the Bagley-Keene sometimes induces, and the fact that people frequently feel like they’re being deprived of their ability to engage in natural communication patterns” said Ted Prim, former California Deputy Attorney General, in a January 2022 training video.
- One researcher, who wished to remain anonymous, suggested that RAP-C makes amendments frivolously, so as to justify its existence. But this view wasn’t common among those we spoke to, who almost universally praised the Panel members and expressed that the delays precipitated by RAP-C are a function of its schedule and existence, not its members.
- Doblin isn’t alone: in TiHKAL, Shulgin explains that the Panel’s name was “finally shortened to Research Advisory Panel [of California] when the acronym of their original name became publicized”.
- That study was Shulgin’s marijuana project, The Origins and Potencies of Marijuana, which was first approved seven years earlier in March 1970. The Panel cited “violation of the controlled substances act and failure to obtain Panel approval for recently-completed non-marijuana Schedule I drug research”. The Panel sent a carbon copy of its letter to the DEA. See Beryl Lipton’s reporting for more.
- Files surrounding the 1994 raid on Shulgin farm “repeatedly point to a decision by the California Research Advisory Panel (CRAP) to reject his proposed marijuana research, which they claim he failed to properly disclose when applying for DEA credentials” according to Muckrock. Shulgin received a $25,000 fine and had his DEA license revoked. Shulgin continues to appear at RAP-C, even posthumously: in 2022, RAP-C approved Synthesis and Structure-Activity Relationships of Psychoactive Drugs Acting on Biogenic Amine Systems submitted by Nicholas Cozzi of the Alexander Shulgin Research Institute (ASRI).
- Shulgin—who was one-part psychonaut, one-part philatelist—abandoned the aforementioned RAP-C approved study on the potency of marijuana strains and their seeds after realising he could obtain Marijuana Tax Stamps, with which he was content. This represented “the ultimate, magical overlap, a synthesis of my two compulsions”, he wrote. The ensuing story is well worth a read, for it includes Shulgin’s summoning to a Grand Jury panel in Washington where an apparent stamp thief is under investigation.
- It was later published as a Brief Report.
- Sasha Shulgin failed to submit his 1976 annual progress report. It didn’t matter too much, though: the Panel withdrew its approval for his studies just months later, asking him to instead turn in a final project report.
- This has remained relatively stable over time; for reference, in 2012 it was monitoring 120 studies.
- Bagley-Keene allows for certain meetings to be closed to the public.
- At least two investigators told us that the fact that all trials that seek to investigate substance use disorder treatments fall within the remit of RAP-C, whether they employ a controlled substance or not, seems particularly odd. They called for these studies to be dropped from RAP-C’s sphere of influence entirely.
- Writing in the Food and Drug Law Journal, Jeffrey Gibbs supposed that, “it is unclear whether the body is able to prevent or halt research studies that are never presented to it” (2004). However, the aforementioned experiences of Shulgin and Mandell suggest otherwise.