
Cybin Shares Positive Interim Topline Data for Deuterated Psilocybin Analog Candidate
Today, Cybin shared topline, interim data from a Phase 2 study of its deuterated psilocybin analog candidate (CYB003) in major depressive disorder (MDD).
Below is our quick reaction to Cybin’s announcement, including comments from Cybin CEO, Doug Drysdale.
Earlier today, psychedelic drug developer Cybin shared interim topline data from a study of its deuterated psilocybin analog candidate, CYB003, for major depressive disorder (MDD). CYB003 was compared to placebo; both administered alongside the company’s ‘EMBARK’ manualised psychotherapy protocol.
Cybin CEO Doug Drysdale told Psychedelic Alpha that today’s topline data reflects 21 participants from the Phase 2a side of the Phase 1/2a trial. 15 of those received a 12mg dose of CYB003, while 6 received a placebo dose.
That’s around two-thirds of the 36 MDD patients that the Phase 2 study calls for. 24 of these patients are assigned to the 12mg group, with 12 assigned to a 16mg group; though today’s topline data only refers to the 12mg group.

Before we jump in, do remember our cautionary notes regarding interpretation of interim, topline data issued by sponsors. In this case, it’s worth adding that this data does not represent the full patient cohort, nor does it represent the 16mg arm. It’s also a small group of patients, with just 15 MDD patients having received the 12mg dose of CYB003.
Let’s take a look…
Positive Efficacy Signal
The data appear to be positive, with a mean MADRS score reduction of 14.08 in the CYB003 group versus placebo at week 3, the primary endpoint. These results are, according to Cybin, statistically significant at this timepoint.
Based on this data, the company claims that 53.3% of the 21-patient cohort were responders at week 3 after a single dose, while 20% were in remission. This is compared to zero in the placebo group. (Response is defined as a ≥50% reduction in MADRS, with remission being MADRS scores ≤10.)
Tantalisingly, the subset of patients in the CYB003 cohort appear to sustain their response up to three weeks, as opposed to a gradual weakening of antidepressant effect as seen in other trials of psychedelics for depression.

Muted Placebo Response
As you can see in the graph above, the placebo response was relatively small, especially when compared to other psychedelic clinical trials (though, as we always mention, inter-trial comparisons are fraught with difficulties, especially when one is going off interim/topline data, and should be interpreted with caution).
Take, for example, COMPASS Pathways’ Phase 2b psilocybin treatment for TRD data:
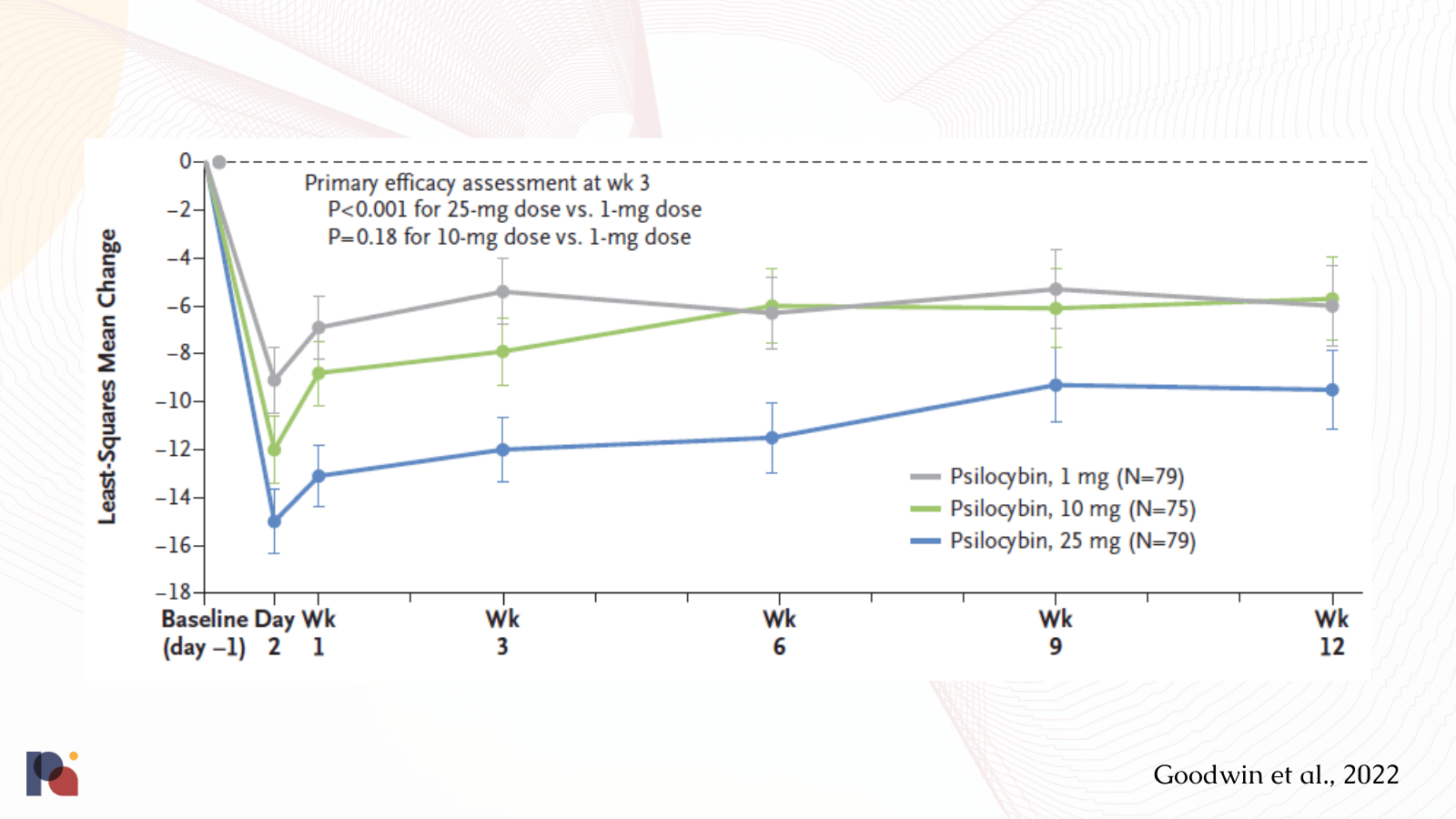
The placebo effect in this trial (i.e., the 1mg COMP360 psilocybin group) was substantial, with a c.9 point mean reduction in MADRS score at Day 2. By Week 1, this had reduced to a c.7 point reduction, ultimately levelling out to around 6 points at week 12.
Usona Institute, meanwhile, recently published (Raison et al., 2023) its long-awaited Phase 2 study of psilocybin therapy for MDD.
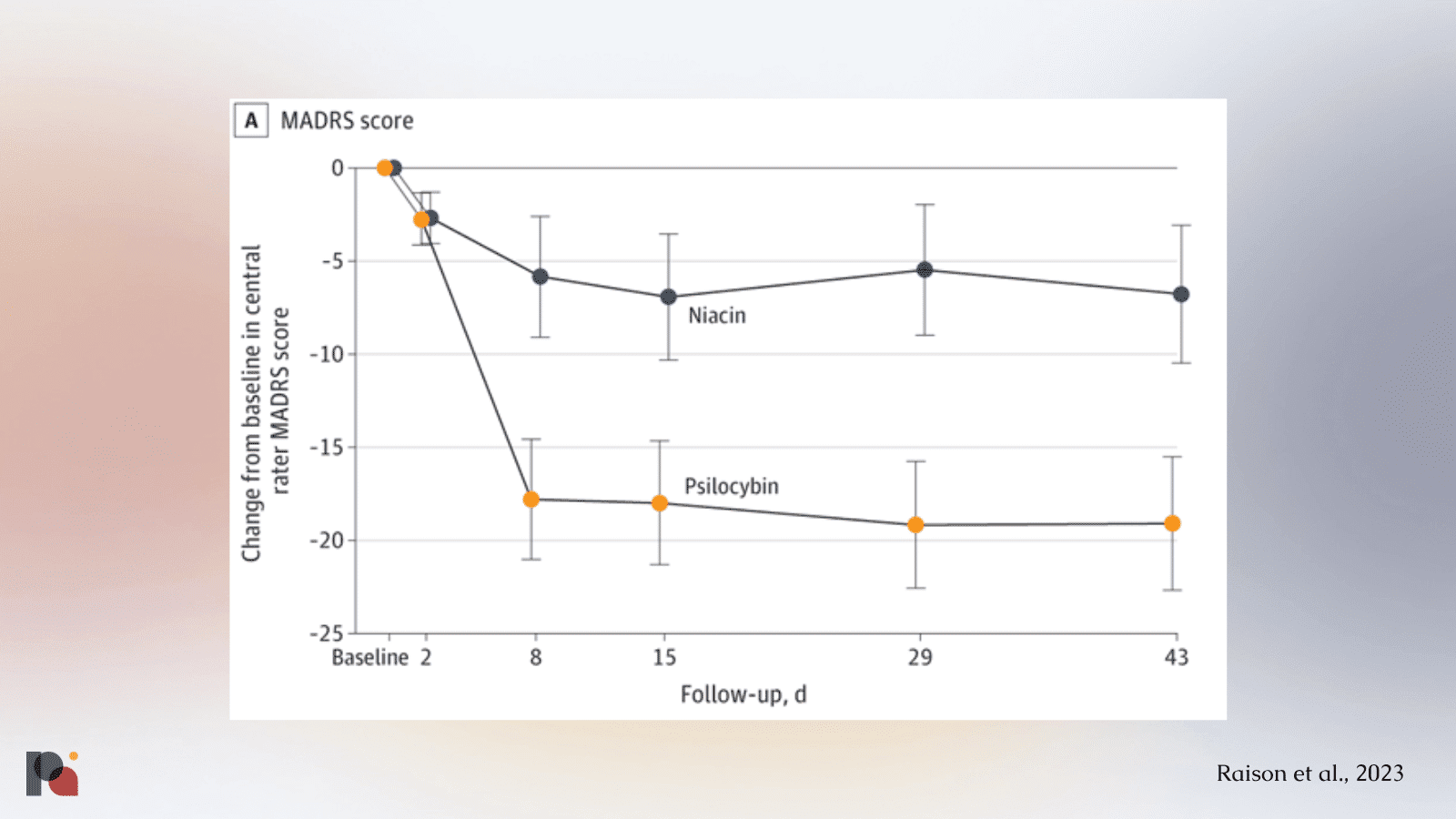
We again see a substantial placebo effect in the niacin group, though not quite as substantial as that seen in COMPASS’ Phase 2b study.
It will be interesting to see if Cybin’s lower placebo effect—which seems to hover around -6 or -7 until Day 10, before receding toward -3 by week 3—is borne out in the full Phase 2a readout.
It is worth noting, again, that inter-trial comparisons are treacherous, especially when dealing with incomplete data. It’s also worth noting that both Usona and COMPASS employed somewhat ‘active’ placebos, though neither were necessarily successful in adequately blinding participants.
Safety Signal
In terms of safety, the company reports that there were no treatment-related serious adverse events observed in this 12mg group.
Deuteration
This preliminary data provides an early look at how deuterated psychedelics perform in the clinic compared to their non-deuterated counterparts. Besides offering a potentially clearer route to obtaining patent protection (and indeed, Cybin recently announced patent grants covering its deuterated psilocybin program), the process of deuteration can also change the pharmacokinetics of the compound.
In the case of CYB003, Cybin hoped that deuterating a psilocybin analog might lead to a more clinically useful—and convenient—compound: i.e., faster onset of the psychedelic effects, shorter overall duration of effects, and a favourable safety profile.
Cybin’s peek under the curtain suggests that psychedelic effects were observed within around fifteen minutes of dosing, with the average duration of “peak effects lasting 2 hours”, according to Drysdale.
The company statement doesn’t give an idea of how long the total duration of effects was, but Drysdale told us that, anecdotally, it was around 4.5 hours. Though, he added, this varied from patient to patient.
And, as mentioned, the company did not report any treatment-related serious adverse events, though there’s no reason to believe that this safety profile is related to the deuteration of the compound.
Next Steps
Based on the findings of this Phase 1/2a study Cybin hopes to progress straight to Phase 3, recruiting patients by the end of Q1 2024. First, the company will need to submit topline data to FDA and request an end-of-phase 2A (EOP2A) meeting for Q1 2024. That’s an ambitious timeline.
In terms of further data redouts, Drysdale told us that we can expect a readout from the 16mg cohort, and two-dose data at six weeks, in late Q4. We should expect data from the optional 12-week open label period early next year (Q1).
Aside from CYB003, Cybin also expects to share topline Phase 1 data from its studies of CYB004 and SPL028, its deuterated DMT compounds (the latter having recently been acquired via its purchase of Small Pharma), before the end of the year. The company hopes to initiate a Phase 2 study in generalised anxiety disorder in Q1 2024.
Pα Take: It’s worth stressing once again that this is a small N study, and today’s interim data reflects a subset of that already-small N. Nonetheless, the data appear positive, with a substantial mean MADRS score reduction and safety profile that gives other psychedelic candidates a real run for their money. It now remains to be seen whether these results are reflected in the full Phase 2a data and ultimately sustained in a larger Phase 3 program. With plans to commence a Phase 3 program as early as Q1 2024, along with a Phase 2 study of its CYB004 deuterated DMT candidate for generalised anxiety disorder, it’s going to be a busy couple of quarters for Cybin staff and investigators! ∎
Join the List
Our Bulletins are delivered to over ten thousand psychedelic insiders. Join the list:
Join the Conversation
We would love to hear from you. Join us on:
↗ Twitter, for ‘live’ updates, news and analysis.
↗ LinkedIn
↗ Instagram
↗ Facebook
Join thousands of psychedelics insiders
Get the weekly psychedelic medicine briefing.
A free weekly digest covering trials, regulation, policy, and access.
By signing up, you agree to our privacy policy. You can unsubscribe at any time.