
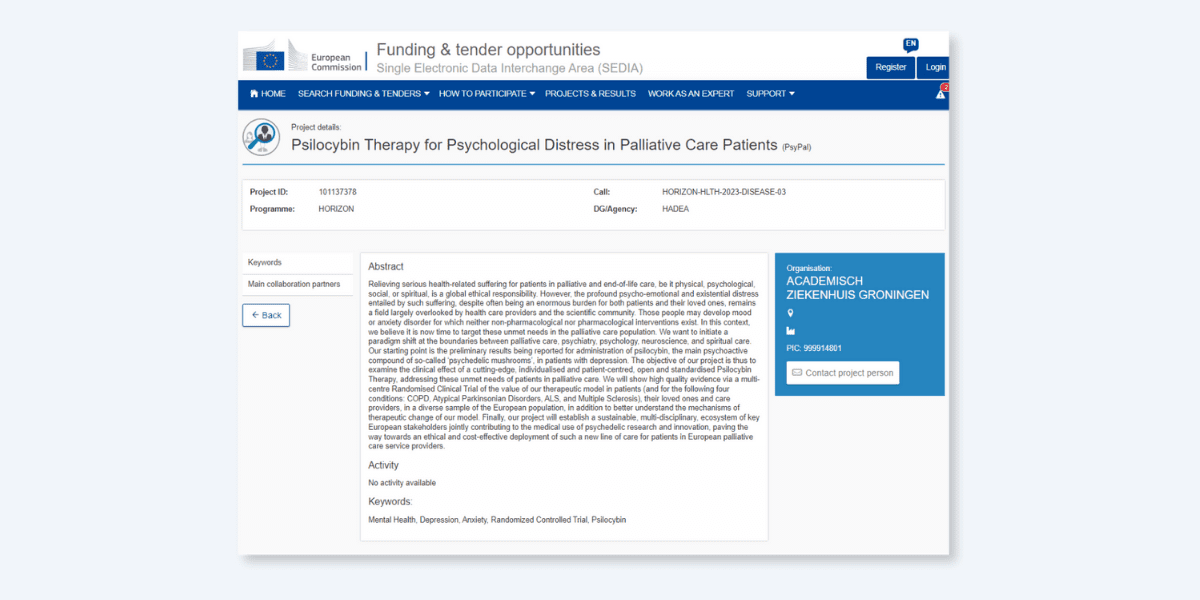
European Union Grants €6.5M for Multi-Site Psilocybin Study in Palliative Patients
The European Union has awarded €6.5 million to support a multi-site randomised controlled trial of psilocybin to ease psychological and existential distress in a variety of progressive illnesses like multiple sclerosis (MS) and chronic obstructive pulmonary disorder (COPD).
According to the recipients, this is “the first ever European grant to fund clinical research into psychedelic-assisted therapy.”
A consortium of organisations across Europe has been successful in their efforts to score EU funding for an ambitious study of psilocybin therapy.
The project, dubbed PsyPal, will explore the potential of psilocybin therapy in addressing psychological and existential distress in four progressive, incurable diseases across four clinical sites on the continent:
- Chronic obstructive pulmonary disorder (COPD) at the University Medical Centre Groningen (UMCG) in the Netherlands
- Atypical Parkinsonism (AP) at the Champalimaud Foundation in Portugal
- Multiple sclerosis (MS) at the National Institute of Mental Health in the Czech Republic
- Amyotrophic lateral sclerosis (ALS) at the University of Copenhagen and the Bispebjerg Hospital in Denmark
These diseases are life-altering, often contributing to a loss of autonomy and significant psychological distress. In people with MS, for example, a lifetime incidence of clinically diagnosable depression occurs in around half of patients.
While elements of the study protocol remain unpublished and subject to finalisation, Psychedelic Alpha understands that two dosing sessions will be employed, with recipients receiving either psilocybin (a lower dose will be administered in the first of the two sessions) or placebo.
Patients will receive three preparation sessions, one of which will focus on each individual’s specific healthcare needs. Psychedelic Alpha understands that the dosing sessions will be followed by one integration session.
OPEN Foundation Director, Joost Breeksema, explained to Psychedelic Alpha that managing these four different patient populations’ physical and somatic issues will be a key area of focus for the study team. The study will be “truly novel”, he said.
Psilocybin therapy has shown promise in improving psycho-spiritual wellbeing in patients with life-threatening cancer, as demonstrated by Griffiths et al. (2016), Shnayder et al. (2023), and others.
But the consortium are keen to highlight that the study is the first clinical trial to investigate the safety and efficacy of psilocybin in non-oncology palliatively treated patients.
Robert Schoevers, UMGC head of psychiatry, and principal investigator of the multi-site study, said: “We are eager to see if we can ease the suffering of these patients whilst also examining longer-term patient and family outcomes of this treatment, something that often gets overlooked but that is of enormous importance.”
It will hope to achieve these broader goals by using peer support and online tools, aiming to enhance participants’ coping mechanisms. This is consistent with a strong focus on post-trial support, according to those close to the project.
“Many people have remaining questions about the meaning of their experience” following psychedelic therapies, Breeksema told us, and can find themselves “isolated following the conclusion of a trial.”
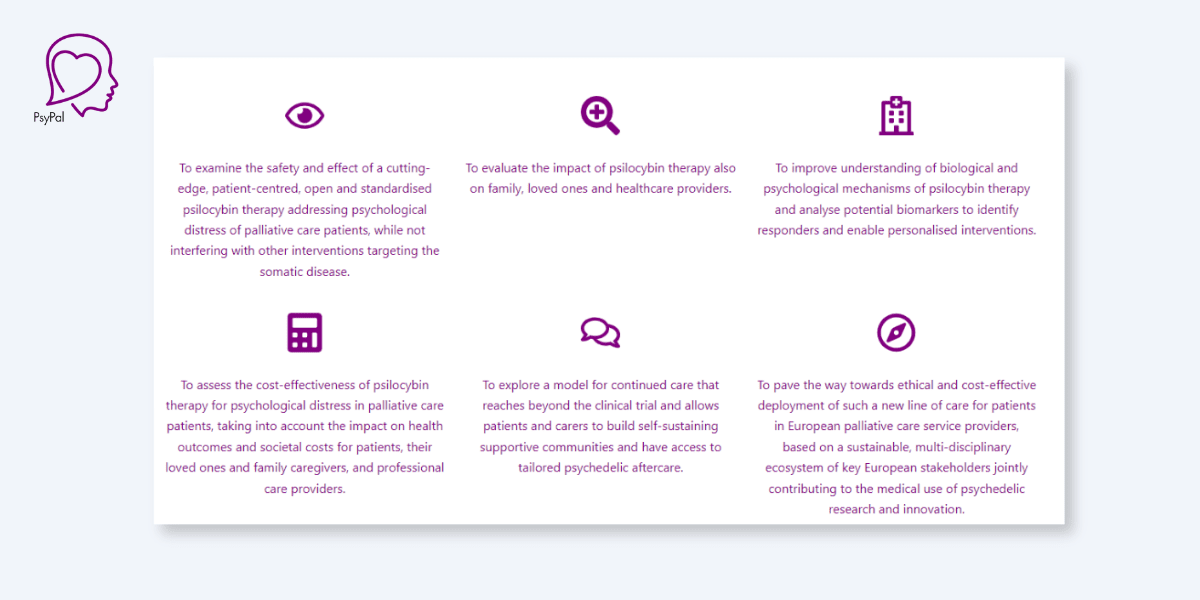
The study will also seek to assess the cost-effectiveness of the intervention with mind to costs and benefits levied at the patient, healthcare system and societal levels. All of this work could contribute to a smoother future roll-out of psychedelic therapies across the bloc.
“This project holds significance beyond the clinical study” Tadeusz Hawrot, Founder and Executive Director of the Psychedelic Access and Research European Alliance (PAREA), told Psychedelic Alpha. “I see it as paving the way for the entire field of psychedelic therapies.”
“For example, PsyPal will create a best practice manual for psilocybin therapy in palliative care and establish a training model for therapists”, he continued, adding that PsyPal “is laying the groundwork for European standards in psychedelic treatments more broadly, something we should start working on now.”
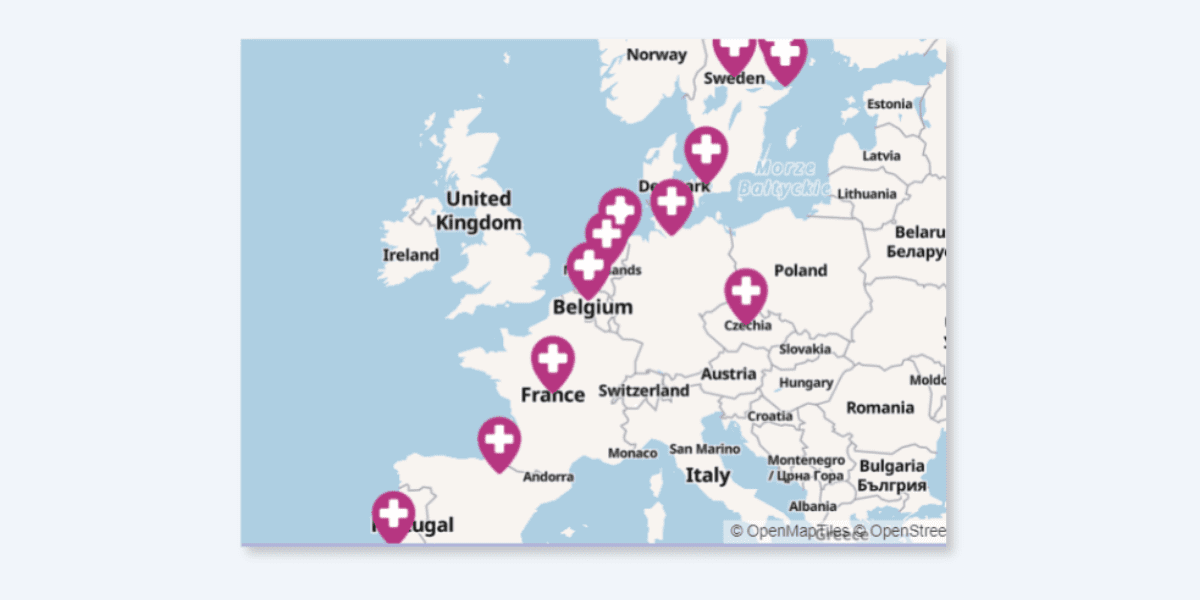
While it’s primarily coordinated by the University Medical Centre Groningen in collaboration with HumanKindLabs, the study involves a consortium of 19 European organisations from nine countries:
- 29k
- A+ Science
- Bispebjerg Hospital
- Champalimaud Foundation
- European Association for Palliative Care (EAPC)
- European Federation of Neurological Associations (EFNA)
- European Psychiatric Association (EPA)
- HumanKindLabs
- IESE Business School
- Lung Alliance Netherlands
- Madopa
- National Institute of Mental Health in the Czech Republic
- Norrsken Mind
- OPEN Foundation
- University Medical Center Groningen (UMCG)
- University of Copenhagen
- University of Groningen
- University of Stockholm
- Uppsala University
- PAREA is also actively involved
Indeed, establishing this interdisciplinary ecosystem of stakeholders in the European psychedelic research (and potential “ethical and cost-effective deployment”) scene is a broader goal of the study.
The funding flows from Horizon Europe, the supranational body’s primary scientific funding stream that boasts a budget of €95.5 billion for a seven-year period starting in 2021. Specifically, the funding was awarded via a call titled, Novel approaches for palliative and end-of-life care for non-cancer patients.
Breeksema told Psychedelic Alpha that the coalition had previously submitted a grant application on palliative care in oncology, which was unsuccessful. (The study was resubmitted to the Swedish Research Council and ultimately funded.)
The present project hopes to enrol 100 patients across the four sites. Work to finalise the protocol and commence enrollment takes place today and throughout the calendar year.
If all goes to plan, the trial will begin in earnest on the 1st of January, 2025. We might expect to see results from the study in 2027.
Pα Take: This is an exciting development, not only due to the funding’s source but also its application: studying psilocybin therapy in diseases as diverse as COPD and ALS, along with comorbid neuropsychiatric issues, is unchartered territory. Managing such a study will certainly be a challenge: from the varied patient population to the nearly 20 interdisciplinary partner organisations, this group has its work cut out. We will be following this project closely and plan to publish a deeper dive into the protocol when it’s made public. We will also be keeping an eye out for further public funding in Europe, which has already seen ERC-funded projects and grants from Dutch authorities. ∎
Stay Informed in 2024 with Pα+
This free article is made possible by our paid subscribers, who gain access to our Pα+ exclusive content.
Subscribe to Pα+ to receive all of our Bulletins, deep dives and analysis, as well as access to the Pα+ Library and other subscriber-only content.
Your subscription goes directly toward supporting our coverage, not glossy graphic design or marketing budgets. Learn more and subscribe here, or get in touch to discuss team plans.

Join thousands of psychedelics insiders
Get the weekly psychedelic medicine briefing.
A free weekly digest covering trials, regulation, policy, and access.
By signing up, you agree to our privacy policy. You can unsubscribe at any time.