
BREAKING: MAPS PBC Files New Drug Application for MDMA-Assisted Therapy for PTSD
MAPS Public Benefit Corporation (PBC) has submitted its New Drug Application (NDA) to the FDA in order to seek marketing approval of MDMA-assisted therapy for PTSD.
The organisation and its parent non-profit, MAPS, has been conducting clinical studies of MDMA-assisted therapy (MDMA-AT; though, a MAPS PBC representative told Psychedelic Alpha that it prefers that the ‘AT’ not be abbreviated, so as not to risk undervaluing the importance of therapy) for over twenty years, making this a significant milestone.
And it’s not just a milestone for MAPS: this is the first psychedelic-assisted therapy submitted to the FDA for approval.
“The filing of our NDA is the culmination of more than 30 years of clinical research, advocacy, collaboration and dedication to bring a potential new option to adults living with PTSD,” said MAPS PBC’s CEO, Amy Emerson.
“If approved, MDMA-assisted therapy would be the first psychedelic-assisted therapy, which we hope will drive additional investment into new research in mental health”, she continued.
When Might MDMA Be Approved?
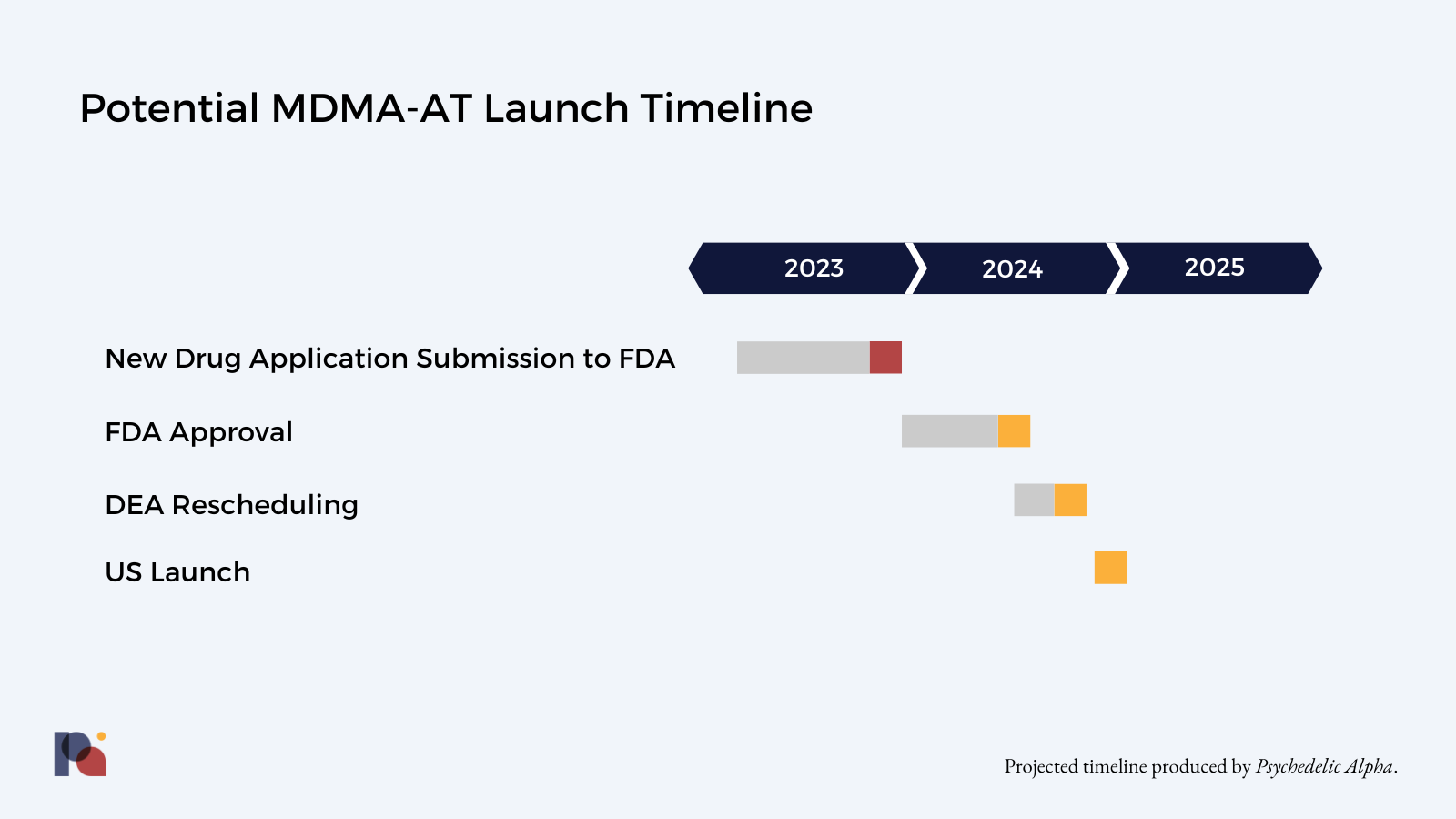
The natural question arising is: when might we expect to hear about a decision from the FDA?
The answer—perhaps unsurprisingly—is that it’s complicated
If the FDA decides that MAPS PBC’s NDA is complete, it generally aims to make a decision within 6 to 10 months.
However, the fact that MDMA will be considered a new molecular entity in the eyes of the FDA will likely lengthen the process—usually tagging on a couple of months to the approval timeline.
It’s also very likely that MDMA-AT will be subject to a Risk Evaluation and Mitigation Strategy (REMS; see our Library entry for more), which will take time to devise, negotiate and implement.
On the other hand, MAPS PBC does have some tailwinds. Notably, MDMA-AT’s Breakthrough Therapy Designation, which aims to expedite the review process. Indeed, MAPS PBC will request that FDA grant Priority Review of the package.
MAPS PBC also secured a Special Protocol Assessment in 2017, which represents some level of agreement between the company and the FDA with regards to study design. This should ensure that the Phase 3 data MAPS PBC includes in its NDA is acceptable to the FDA for the purposes of market approval. The FDA also agreed to rolling review of portions of MAPS PBC’s NDA in November 2021.
These tailwinds and headwinds might cancel each other out to some extent, meaning that the upper end of FDA’s “6 to 10 months” timeline might be a reasonable expectation.
As part of that deliberation, we should expect to see the FDA convene an Advisory Committee, perhaps during the summer.
If the FDA approves MDMA-AT for PTSD, DEA rescheduling would then take place. This could take up to three months at the federal level, with more than twenty states also having to reschedule at the state level. (See our Library entry on bifurcated scheduling for more.)
As such, we might hope to see MDMA-AT approved for PTSD in (late) 2024. If all goes to plan, we might even see the first patients receiving the therapy before the end of next year, though numbers would be small.
While an approval would be an enormous success for MAPS, MAPS PBC and psychedelic therapy more broadly, there will be little time to celebrate: rolling out this first-in-class treatment will be no small feat.
Aside from commercialisation (which includes choosing a brand name for MDMA, such as RYSANSO) and roll-out-related efforts, MAPS PBC may also be required to undertake further studies of MDMA-assisted therapy, such as in paediatric populations.
Pα Take: Now that MAPS PBC has submitted its New Drug Application, we might expect to hear of a potential approval as early as 2024. We’re expecting any decision to come late in the year, meaning the therapy might reach small numbers of patients in late 2024 or early 2025. ∎
Related Content from Psychedelic Alpha
- Psychedelic REMS – Pα+ Library.
- Bifurcated Scheduling – Pα+ Library.
- Second Phase 3 Study of MDMA-assisted Therapy Reports Positive Results, Paving Way for New Drug Application – Psychedelic Alpha (September 2023).
- From Approvals to Access – Psychedelic Alpha (January 2023)

Join thousands of psychedelics insiders
Get the weekly psychedelic medicine briefing.
A free weekly digest covering trials, regulation, policy, and access.
By signing up, you agree to our privacy policy. You can unsubscribe at any time.