
MEP Action Group Meets in Brussels to Discuss Regulatory Pathways for Psychedelics
We share our notes from the inaugural meeting of the MEP Action Group for the Medical Use of Psychedelics which took place in Brussels on Monday.
Pα+ subscribers can access an enhanced version of this article, which includes additional commentary and analysis. Click here to Join Pα+ today.
On Monday, the inaugural meeting of the MEP Action Group for the Medical Use of Psychedelics took place at the European Parliament in Brussels.
The meeting was co-organised by PAREA and PsychedelicsEUROPE, who kindly invited me (Josh Hardman) to present alongside a handful of others: from researchers and industry through to patients and policy advocates.
The MEP Action Group, formed in May, seeks to promote the clinical use of psychedelics in the EU. This first meeting, Innovating Mental Health in the EU Pharma Legislation: Regulatory Pathways for Psychedelic Therapies, sought to explore how a revised EU pharma package could support this field.
Background: Revising the EU Pharma Legislation
The revised EU pharma package aims to—among other things—balance the need for equitable and efficient access to safe and effective medicines with the need to incentivise the development and launch of innovative medicines in Europe. It will be the most significant revision to the EU’s pharmaceutical legislation in two decades.
Thus far, proposed revisions have included changes to the regulatory exclusivity regime. At present, new medicinal products receive 8 years of data protection followed by 2 years of market protection, effectively providing a 10-year monopoly that staves off generic entry. Sponsors can also obtain an extra year should they achieve authorisation for at least one additional indication within 8 years of approval, if the product demonstrates significant clinical benefit versus existing treatments.
An early proposal for the revised pharma strategy looks set to cut the standard data protection period down to 6 years, with a full 10 years of exclusivity achievable only where certain criteria are met. The 2-year market exclusivity period remains. Extensions to the data protection period might be achieved in the following ways:
- An additional 2 years of data protection is awarded where a new product is launched across all EU member states within 2 years of approval (this is extended to 3 years for some entities, including SMEs and nonprofits).
- 6 months can be tacked on if the product addresses an unmet medical need: a life-threatening or severely debilitating disease with insufficient existing alternatives.
- 6 months can also be tacked on when a medicinal product contains a new active substance and where clinical trials used an evidence-based comparator.
- An additional year can be obtained if marketing authorisation for an additional indication is achieved for a condition where there is significant clinical benefit versus existing treatments.
Aside from these changes to exclusivity periods, the revised package might lead to significant changes in the European Medicines Agency’s (EMA) composition in an effort to make it more efficient (which could, for example, speed up review timelines) and deepen its scientific advice program, for example.
Regulatory sandboxes might also be introduced, which would enable the EMA to consider broader data sources such as real-world data and evidence of the off-label use of existing medicines.
The period of revising the far-reaching pharma package offers a key opening for psychedelic research and drug development, according to attendees at the MEP Action Group event. In terms of timelines, it could be a lengthy process, with no set timeline for its adoption. Many expect the process to extend into 2026 and beyond.
Here, we provide brief notes on the meeting. You can watch a recording of the slides plus audio below:
I (Josh Hardman) began by providing a brief overview of select milestones at the intersection of psychedelic drug development and policy/regulation. I noted that despite a suite of breakthrough therapy designations in the U.S., and a bout of Innovation Passports in the UK, psychedelic drug development candidates do not appear to have been designated as ‘priority medicines’ under EMA’s PRIME scheme.
In fact, not a single psychiatric drug has made it through to approval with the help of PRIME, with only two such candidates ever deemed eligible (both of which were ultimately shelved by their sponsors). Compare that with oncology, which has seen 34 candidates granted PRIME status.
(In spite of this lack of fast-track type designations, EMA has been publicly supportive of psychedelic drug development activity, with Chief Medical Officer Steffen Thirstrup attending ECNP’s New Frontiers meeting on psychedelics in March (read our Dispatch), penning a supportive post on EMA’s LinkedIn page, and co-authoring a Lancet article on the topic, for example.)
I then turned to public and practitioner attitudes, pointing out that a (slim) majority of the public appears to be open to trying psychedelic therapies for mental health issues. I also cited Barnett et al.’s two surveys that show a drastic increase in the proportion of surveyed psychiatrists that agreed psychedelics show promise in treating psychiatric disorders.
Having looked at the changing landscape of global psychedelics policies and perspectives, I moved on to the crux of my presentation: the geographical distribution of psychedelic drug development and potential approvals.
At the most mature end of the clinical development spectrum, MDMA-assisted therapy looks set to be approved in the U.S. as early as next year. Psilocybin, meanwhile, could be eyeing FDA approval in the next half-decade, too. (The day after I prepared my presentation, COMPASS Pathways announced that it has received authorisation to begin recruiting at a number of European trial sites as part of one of its Phase 3 trials.)
However, I’m concerned by the apparent lack of approval-enabling programs in Europe. There are a great deal of early-to-mid stage trials across the continent, with many of psychedelic research’s great insights seeded by European researchers and participants, but it looks like Europeans could be waiting substantially longer than their U.S. counterparts to access any approved therapies.
In this sense, one might argue that Europeans are psychedelic guinea pigs, or lab rats.
One striking example is the fact that the world’s first clinical trial of MDMA-assisted psychotherapy commenced in Spain in the year 2000. However, it was shut down by local authorities after just a half-dozen of the 29 participants had been treated. 24 years later, MDMA-assisted therapy could be approved for PTSD in the U.S., which makes me wonder: how far behind might approval and access be in Spain, where the first clinical trial participants tested the modality over two decades ago?

On to potential solutions, through the lens of the EU pharmaceutical legislation revision. Raising funds to support pivotal studies in Europe is a major issue for (psychedelic) drug developers, especially today.
The EU could support such efforts through mechanisms like incentives and grants. These need not be handouts, either. Support could, for example, be linked to public health (economic) priorities, such as by requiring the inclusion of a comparative element in trials that would contribute to the goals of EMA and member states in identifying the most (cost-)effective treatments.
Deeper and earlier support from EMA could also give funders the confidence to support large-scale and late-stage psychedelic programs. At present, EMA’s scientific advice facility is chronically underused by psychiatric drug developers, a fact that is presumably acknowledged by the draft revised strategy’s focus on offering advice by default as opposed to waiting for a sponsor to reach out. Beyond scientific advice, I think early engagement on matters such as health technology assessment and logistical considerations could help psychedelic drug developers better anticipate the EU landscape.
Ideally, EMA would be willing to recognise much of the data produced outside of its bounds, which would significantly reduce the resource and time burden of conducting large-scale European trials specifically to enable EMA approval.
Another challenge for psychedelic therapies is the fact that they are much more resource-intensive than standard of care, which raises questions around the likelihood of reimbursement in EU member states as well as increasing trial complexity and cost.
Plans for an EU-wide health technology assessment (HTA) agency—while some way off—could prioritise a societal perspective when conducting cost-benefit analyses of medicines, in order to capture the broad societal costs of mental health disorders and (both direct and indirect) benefits of interventions such as psychedelic therapies.
What’s more, the EU could take the lead in supporting studies that seek to uncover more cost-effective models of psychedelic therapy, such as group therapy protocols or comparative trials.
As aforementioned, the revised pharma package might reserve some portion of market exclusivity for sponsors that launch across all 27 member states within two or three years of marketing authorisation. The purpose of this proposed revision is understandable: to reduce inequalities in access to innovative therapies across the bloc’s member states.
However, this will be very challenging for psychedelic-assisted therapies, given the logistical and infrastructural demands and constraints they invoke. At a minimum, developing and sharing ‘cookie cutter’ models at the EU level, for member states to adopt as they see fit, could enable a more efficient launch of psychedelic therapies across member states. But I still feel it’s a tall ask.
My concluding remarks were as follows:
- Europeans are aware of the potential of psychedelic therapies.
- Many of the world’s most influential and accomplished psychedelic researchers and drug developers are based in Europe.
- The bulk of regulatory flexibility and encouragement for late-stage psychedelic drug development is occurring outside of Europe.
- Europeans risk being psychedelic guinea pigs, seeding breakthroughs before late-stage, approval-enabling clinical programs are offshored to North America.
- There are concrete steps the EU and its agencies can take to support this field, which offers genuine potential for addressing significant unmet needs in Europe.

Next up was Tadeusz Hawrot, Founder and Executive Director of PAREA, who set out the scale of the unmet need that psychedelic therapies might contribute to addressing.
According to EU Health at a Glance 2018, 1 in 6 people in Europe have a mental health issue, with 1 in 2 members of the EU population experiencing mental illness at some point in their lifetime. The economic and social impact of such illnesses is enormous. So much so that the World Economic Forum’s Global Risks Report 2023 calls out deteriorating mental health as a key risk to economies and societies.
And, treatment is wholly inadequate: WHO Europe estimates three out of four people with major depression do not receive adequate treatment. Every year in Europe alone, 130,000 die by suicide: i.e., since 2016, a million Europeans have lost their lives to suicide. (In the Q&A section, David Nutt suggested that suicide rates were a key reason why TGA decided to enact a limited rescheduling of psilocybin and MDMA for treatment-resistant depression and PTSD, respectively, earlier this year.)
Forward-looking projections suggest that depression will become the leading cause of disease burden by the end of the decade. Crucially, estimates of disease-burden-reduction potential by 2040 are very pessimistic when it comes to mental health disorders. One estimate from McKinsey reported that just 14% of the burden is expected to be preventable in a healthy growth scenario.
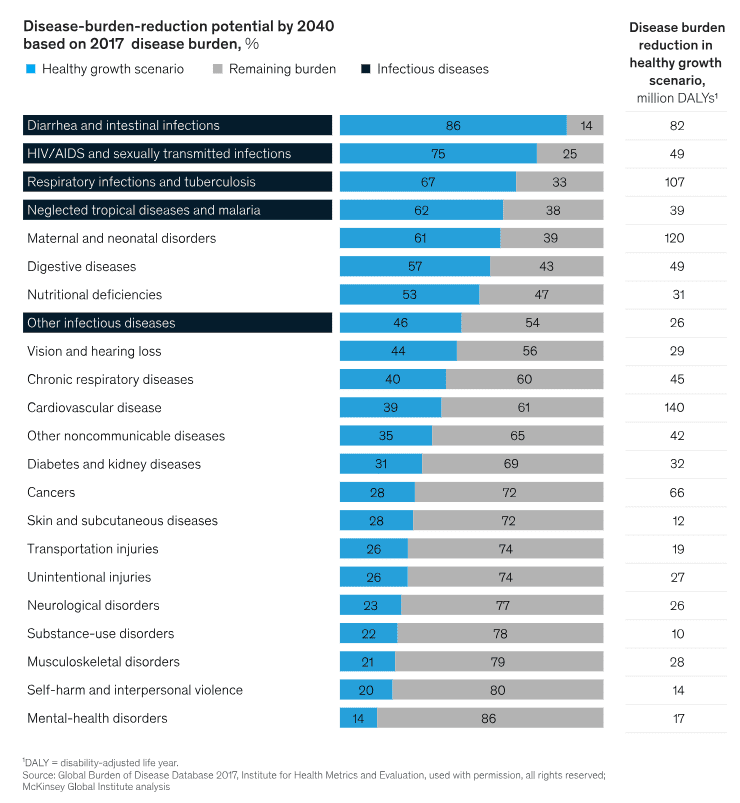
A stagnation in the development of new therapeutics for mental health disorders entrenches this somewhat bleak outlook. But it also emphasises the need to support promising new treatments, such as psychedelic-assisted therapies.
Defining Unmet Medical Needs
The meeting’s presenters shared a suite of actions that EU, its agencies, and member states could take to catalyse the development and roll-out of psychedelic therapies, such as funding and incentives as well as EU-level logistical and reimbursement planning to get ahead of foreseeable hiccups.
An aligned approach is to push the envelope when it comes to the definition of ‘unmet medical needs’. Ensuring that the dire reality of mental health is captured by such a definition could spur broader support for the research and development of potential innovations, which psychedelic therapies might find themselves amongst.
To this end, PAREA launched a new policy paper, Unmet Medical Needs: Aligning Medical Innovation with Societal Health Needs. The paper calls for a broader conception of ‘unmet medical needs’, arguing that a narrow focus on ‘life-threatening or severely debilitating conditions’ neglect the immense social and healthcare costs of mental health and substance use disorders.
If these conditions were brought under the umbrella of ‘unmet medical needs’ through a fine-tuning of such criteria, we might see them more appropriately prioritised when it comes to R&D incentives.
The position paper calls for the establishment of a European Hub for Mental Health R&D. That hub would bring EU and Member State institutions, healthcare funders and philanthropists into conversation with one another, establishing a common research infrastructure supplemented by guidance and support.

Next, Maryam Jabir shared her experience as a participant in a phase 2b study of psilocybin therapy for treatment-resistant depression.
Having struggled with depression for many years, Maryam said she had tried repeated bouts of psychotherapy to the point she had almost begun gamifying her sessions: trying to make her therapist laugh, for example. Her mind was “like a Gordian Knot” by this point, she said.
It’s perhaps unsurprising, then, that Maryam was intrigued when she came across a media report that included a quote from a psilocybin clinical trial participant about how the drug might ‘reset’ one’s brain. She applied to the trial.
While she says that she was not expecting a silver bullet, she hoped it might provide “a raft, or even some driftwood” from which she could pull herself ashore and start to get better.
(Note: you really should listen to Maryam’s testimony in her own words. Here’s a link to the recording, which should take you to the relevant segment at 33:18.)
Going into the trial, Maryam says she deliberately tried to under-research psilocybin to avoid priming herself, and it wasn’t until her second preparation session with the study therapist that she realised she might have a full psychedelic experience. She was also apprehensive that she might not be in the ‘active’ arm of the trial, meaning the 25mg psilocybin group (as opposed to the 10mg or 1mg dose). Maryam suggested that was one of her primary concerns… she hadn’t even considered that she could ‘get worse’ after ingesting psilocybin.
She did, indeed, have “what is euphemistically called a challenging experience”, which resembled “a hurricane”. Upon coming out of the experience, Maryam said that she felt “like I had failed the team, the drug, my mum”. It was a very difficult time, she said.
“I also want to be very clear”, Maryam said, that the dose of psilocybin ‘didn’t make anything worse’, but it did make her confront many difficult things all at once. She questioned what is deemed an ‘ideal response’ in psilocybin therapy: “Is it a feel-good trip, or is it progress? Is it happiness, or is it healing?”
“If you’re skeptical about psychedelic therapy or if hearing my story is concerning you right now, I’d just invite you to reimagine the concept of what therapeutic means. For me, it’s as I said to my therapist: I’m past just wanting to feel good, I want to feel better. The first thing I said when they did take me out of the experience was: I can’t go on like this alone, I need help. And for me, that was a huge thing to acknowledge.”
While her experience was clearly difficult—“one of the hardest things I’ve ever gone through”—Maryam said she would do it all again, “exactly as it happened”. The benefits of her experience “took the scenic route”, she said, adding that “it’s undeniable to me what a force these treatments could have on mental health care; a force that has the potential to heal”, as opposed to damage control.
She was very appreciative of the trial team. “It’s frustrating to know that I can’t seek treatment again” she added, alluding to the lack of options, adding that “I would never feel safe enough to do it without similar support available.”
Maryam said she went from having excellent care during the trial to almost none. Peer-led integration groups in society are so important, she said. (Coincidentally, a few hours prior to Maryam’s talk, a paper published in Neuroethics titled: When the Trial Ends: The Case for Post-Trial Provisions in Clinical Psychedelic Research.)

Next up, Tomáš Pálenícek looked at the standard of care for depression, before comparing it to emerging evidence on psychedelics as antidepressants. The data will be familiar to readers, but a couple of Phase 2 trials at the Czech National Institute of Mental Health might have flown under the radar.
The trials both seek to compare psilocybin with ketamine, with a midazolam group as a type of active control. The first compares the two drugs in treatment-resistant depression, while the second looks at depression comorbid with a cancer diagnosis.
Tomáš shared some of the challenges and costs in running clinical trials with psychedelics, such as obtaining GMP drug product, training therapists, and generally meeting clinical trial approval criteria with more modest budgets compared to industry sponsors.

Viktor Chvátal, Secretary-General of PsychedelicsEUROPE, spoke next. He emphasised his organisation’s key goals: the harmonisation of rules to “open the EU single market for medical use of psychedelics”; inclusion of psychedelics in key EU initiatives such as the pharmaceutical package and Horizon; and, financial allocation for psychedelic research from both the EU and member states.
Viktor urged for more private sector involvement in such efforts and explained that his organisation will seek to engage payers on the topic of reimbursement.

Last up was Damian Kettlewell, CEO of psilocybin drug developer Clairvoyant Therapeutics, which is engaged in a Phase 2b study of psilocybin therapy for alcohol use disorder (AUD). He noted positive data from Bogenschutz et al.’s (2022) study of psilocybin-assisted therapy for AUD, which found an 80% reduction in heavy drinking days among those in the psilocybin group, and said his company’s trial design is very similar. (Read our Deep Dive on Bogenschutz et al.’s study.)
The company’s objective is to be the “1st company globally to medicalize psilocybin”, with a “speed-to-market strategy designed to enable psilocybin therapy in the EU, UK & Canada” beginning 2026. Put differently, the company hopes to beat COMPASS Pathways to market approval for a drug product containing psilocybin anywhere in the world, but their arguably more hedged goal is to gain approval of psilocybin therapy for AUD in certain ex-US jurisdictions.
Kettlewell shared that the company has, as of November 3rd, randomised 63 participants in its Phase 2b study that pits a 25mg dose of psilocybin against placebo across two dosing sessions (4 weeks apart), in which it expects to complete enrollment—targeting 128 total participants—by April 2024. Interim results representing 64 participants are expected in February 2024. The majority of participants are from the EU, with four active sites in Finland.
The company is bearish on the rigour of psilocybin patents, with Kettlewell saying that “we do not believe that patents are strong blockers […] in regards to the classics like psilocybin, MDMA, DMT and LSD.” Instead, it’s hoping to benefit from data exclusivity: currently set at around 10 years in the EU, though this could change with the aforementioned revisions to the EU pharmaceutical legislation.
Reflecting on their experience working on trials in the EU, Kettlewell said that patient recruitment has been very strong in Finland, with “very high interest” from therapists, too. He also said Clairvoyant is hopeful of R&D funding opportunities, adding that this is a motivation for the company to establish a presence in Portugal.
In terms of challenges, Kettlewell said that he perceives some “reluctance to psychedelic research” in Eastern Europe. He added that it’s challenging for a non-EU company to access EU funding options. Finally, he noted that the proposed changes to data exclusivity in the EU were generating uncertainty, which is unsurprising as the company’s commercial strategy hinges heavily on the outsized data exclusivity period offered by EU vs., say, the US.

In closing the meeting, Deirdre Ryan, President of Pain Alliance Europe, noted that ‘unmet needs’ in this area are often “life and death for people”. “We really need champions at the national level, the member states, to get behind this,” she added, noting that “the EU will always tell us: it has to come from the Member States; and the Member States always say ‘no, we need the EU to tell us what to do.”
She encouraged a bridging of the void, with patients, regulators, payers, researchers and industry to come together in a multi-stakeholder manner to further support this field and other mental health treatments.
Ryan also reminded people that elections are coming soon, alluding to the June 2024 ballot.
Pα Take: The inaugural meeting of this new MEP Action Group offers plenty of food for thought regarding how psychedelic therapies and their developers might benefit from the revised EU pharma strategy. Realising such benefits will require a continued, multi-stakeholder effort, especially as Europe heads into elections next year. We look forward to covering developments in this geography. ∎
Join the List
Our Bulletins are delivered to over ten thousand psychedelic insiders. Join the list:
Join the Conversation
We would love to hear from you. Join us on:
↗ Twitter, for ‘live’ updates, news and analysis.
↗ LinkedIn
↗ Instagram
↗ Facebook
Join thousands of psychedelics insiders
Get the weekly psychedelic medicine briefing.
A free weekly digest covering trials, regulation, policy, and access.
By signing up, you agree to our privacy policy. You can unsubscribe at any time.