
Psychedelic Bulletin #152: Policy and Business Updates
This Issue, we’re taking a deep dive into a number of policy, business and commercial stories that are relevant—in various degrees— to psychedelics. We broadly break up our coverage into two buckets: Policy and Related and Business and Commercial, with around a half-dozen stories in each.
This Issue is free. Our independent coverage of psychedelic business, policy and research is made possible by our Pα+ subscribers. Subscribe today to receive all of our Bulletins, deep dives and analysis, as well as access to the Pα+ Library and other subscriber-only content.
Policy and Related
DEA Relaunches Attempt to Place Two Psychedelics—DOI and DOC—in Schedule I
The U.S. Drug Enforcement Administration (DEA) has published a notice of proposed rulemaking that would see two psychedelics, 2,5-dimethoxy-4-iodoamphetamine (DOI) and 2,5-dimethoxy-4-chloroamphetamine (DOC), placed in Schedule I of the Controlled Substances Act (CSA).

If you’re having déjà vu, you’re not alone: DEA proposed rulemaking with the same intended outcome in spring 2022, but ultimately withdrew its proposed rule on 26th August, 2022. Ominously, the agency noted that it was “planning to publish a new proposed rule with an amended procedure.”
(Note: The agency also went after five tryptamines, but was thwarted by a small group of researchers, psychedelic startups and lawyers that challenged it. See our Special Report on the matter for more: Inside the Challenge to DEA’s Proposed Scheduling of 5 Psychedelic Tryptamines.)
“Perhaps this withdrawal is the product of a procedural quirk as opposed to a change in attitude from the agency”, we speculated in our coverage in Bulletin 116.
Indeed, the DEA has come through on its hint at re-upping this rescheduling action. In the new notice, the agency said that it withdrew the first rulemaking attempt “in order to provide additional clarity on the process for submitting hearing requests”; i.e., an administrative matter.
“The scientific, medical, and other bases for the proposed placement of DOI and DOC in schedule I remain the same in this proposed rule as they were described in the April 2022 proposed rule, except for minor updates to certain data”, the agency added.
Given that the notice was published on the 13th of December, the comment period closes on the 12th of January. That’s a tight turnaround when the holiday period is factored into things.
Comments may be submitted here.
Beyond DOI and DOC: Positional Isomers and the Case of 3-MMC
There might be more at stake here than DOI and DOC, too. As we noted in our coverage of the five tryptamines matter, the addition of molecules to Schedule I also subjects their positional isomers to potential unilateral scheduling by DEA. (Positional isomers are, generally, molecules that possess the same chemical formula as another but where the location of the functional group differs.)
These positional isomers could be numerous, and very distinctive from DOI and DOC. They might not even be psychoactive at all, for example.
A good example of this unilateral action by the DEA is the placement of 3-MMC in Schedule I of the CSA, which is effective December 13, 2023. DEA explained that 3-MMC (3-methylmethcathinone) “meets the definition of a positional isomer” when compared to mephedrone (4-methylmethcathinone) and as such “has been and continues to be a schedule I controlled substance”. As DEA explained, both compounds “possess the same molecular formula and core structure, and they have the same functional groups. They only differ from one another by a repositioning of an alkyl moiety”:
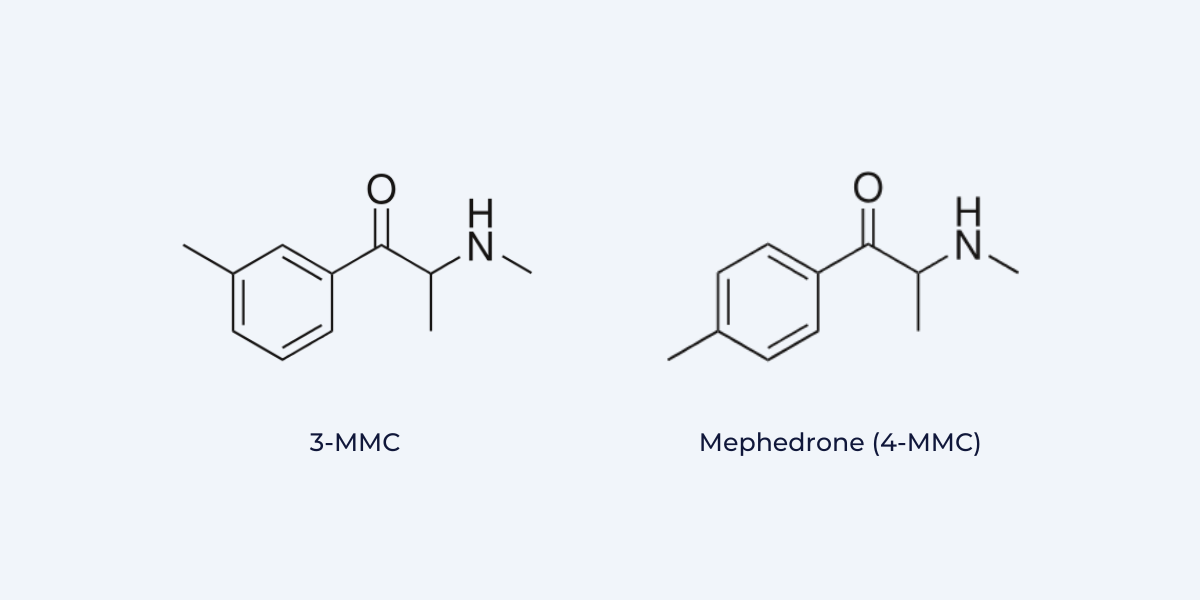
The agency found that “publishing a notice of proposed rulemaking and soliciting public comment are unnecessary”, as the rule is a “technical amendment” that it deems “insignificant in nature and impact, and inconsequential to the industry and public.”
Positional isomers of DOI and DOC (2,5-dimethoxy-4-iodoamphetamine and 2,5-dimethoxy-4-chloroamphetamine) would for example include their 3,5-dimethoxy, 2,6-dimethoxy, and 3,6-dimethoxy analogs, analogs where the alpha-methyl is exchanged for an N-methyl, and others, many of which appear never to have been made or tested.
Interestingly, a 3-MMC patent application filed by MindMed—published in 2020, but which had been abandoned in the U.S. in Q3 2021—looks set to be revived by the company, which has petitioned the Patent Office for such. The application (US16/636,044), with co-inventors Ezekiel Golan (“Dr. Z”, credited with the creation of 4-MMC), Mark Haden (formerly Executive Director of MAPS Canada), and René van Wettum, claims methods of “augmenting psychotherapeutic intervention” by administering 3-MMC. (MindMed has already received patents on the same application in Canada, Australia, and Japan; examination is in progress in Europe.)
Psychedelic Trials for Active-Duty Service Members Included in Passed NDAA
The must-pass National Defense Authorization Act (NDAA)—which stipulates the annual budget for the U.S. Department of Defense (DoD)—contains a curious easter egg this year: an amendment that instructs the DoD to provide grant funding for research into the use of psychedelics to treat PTSD, traumatic brain injury (TBI) or chronic traumatic encephalopathy (CTE) in active-duty service members.

The amendment was inserted into the NDAA via the Douglas ‘Mike’ Day Psychedelic Therapy to Save Lives Act, forwarded by Dan Crenshaw and included in the budget by fellow Republican Morgan Luttrell (who sits on a board of five representatives responsible for assembling the NDAA each year). In last year’s NDAA, Crenshaw’s amendment was dropped. This year, it snuck through.
The Act carves out $15m per fiscal year from 2024 through 2028. It’s not yet clear who might be the beneficiaries of these grants, though we do know they’re intended to flow to “federal or state government entities, academic institutions, and nonprofit entities.”
The Act does stipulate the types of studies that might be conducted:
(1) conduct one or more phase two clinical trials for the treatment of covered conditions that—
(A) include members of the Armed Forces serving on active duty as participants in the clinical trial; and
(B) use individual or group therapy assisted by covered psychedelic substances; or
(2) train practitioners to provide treatment to members of the Armed Forces serving on active duty for covered conditions using covered psychedelic substances.
In terms of psychedelics, MDMA, psilocybin, ibogaine or 5-MeO-DMT may be studied.
Jamaica Establishes Psilocybin Industry Working Group
The Jamaican Ministry of Industry, Investment and Commerce—via the Bureau of Standards of Jamaica (BSJ)—has established the Jamaica Psilocybin Industry Working Group (JPWG).
“As this new industry develops with good signs of growth, it has become necessary to study and agree the broad parameters and guidelines necessary for the standardisation of this growing psilocybin industry”, the Ministry explained in a statement.
The group is chaired by Senator Dr. Saphire Longmore, a psychiatrist who uses psilocybin in her own practice, who moved a Private Member’s Motion in the Senate back in 2020 to call for greater standardisation of the growing psilocybin industry in Jamaica.
Members of the Working Group include a mix of practitioners, business owners and researchers including Justin Townsend of MycoMeditations, Jordan James of Looking Glass Ltd, Jason Williams of Cosmic Mushrooms, Professors Jean-Pierre Louboutin and Rupika Delgado of the University of the West Indies, and Dr. Velton Gooden of the BSJ.
The group’s remit is “to study and understand the production and distribution arrangements, recommend suitable methods to facilitate, enable and encourage the standardisation of practices and the growth of the psilocybin industry in the country.” The Ministry’s statement continues, noting that “Clear consideration must be given to public education and the proper growing, processing and use of the product.”
The working group is tasked with submitting a report to the Ministry within six months, with potential recommendations to the Cabinet thereafter.
“Psilocybin is a fact of life in Jamaica”, said MycoMeditations CEO Townsend.
There are reportedly over twenty retreats on the Caribbean island, which represents a small but not insignificant source of revenue for the local economy according to Longmore. However, Longmore is also interested in leveraging psilocybin to address “some of the psychological issues some Jamaicans face, especially people who have a history of trauma.”
This growing industry is buoyed by the fact that psilocybin mushrooms have never been prohibited in Jamaica, and that psilocybin itself is not a controlled substance under Jamaica’s Dangerous Drugs Act. Accordingly, psilocybin mushrooms are legal for possession, use, sale, and cultivation. (See our Worldwide Psychedelic Laws Tracker.)

Whether it be via economic benefits to the local economy or mental health benefits to Jamaicans, this newly-formed Working Group looks set to ensure that the budding psilocybin industry benefits locals as well as tourists.
New York Fines Payors Over Lack of Mental Health Parity
New York’s Governor, Kathy Hochul, issued $2.6 million in fines to five health insurers last month. Hochul’s statement explained that the fines were for violating state regulations and Medicaid rate payment law.
Following “a comprehensive examination” of these payors’ reimbursement of behavioural health service claims in particular, the state decided that the five insurers were failing to adequately reimburse behavioural and mental health treatments. As such, New Yorkers were facing “unnecessary barriers to mental health care”, Hochul said.
Office of Mental Health Commissioner Dr. Ann Sullivan added that “companies that continue to flout these regulations are imposing a formidable barrier that ultimately discourages New Yorkers from getting the mental health care they need.”
While statute is supposed to ensure parity between Medicaid payments for physical and behavioural healthcare, in reality, there is little enforcement. Might other states follow New York’s lead?
European Parliament Calls for Direct Funding and Bloc Strategy on Mental Health
Staying with the topic of mental health parity, there have been further promising developments in Europe.
In June, the European Commission (EC) presented its strategy for a ‘comprehensive approach to mental health’, which seeks to put mental health on par with physical health “by introducing 20 flagship initiatives and €1.23 billion in funding from different financial instruments.”
Six months later, Members of the European Parliament (MEPs) have voted in favour of a report calling for greater funding to address mental health challenges in the bloc. The December 12th vote was passed by a wide majority.
The European Parliament report goes further than the EC’s by calling for a direct fund for mental health research and innovation, as opposed to funding via broader programs such as the Horizon 2020 and Horizon Europe programs.
The report also focuses on identifying and addressing the determinants of (poor) mental health, especially in vulnerable groups. As noted by PAREA’s Tadeusz Hawrot, MEPs are also calling on the EC to designate 2024 as the ‘European Year of Mental Health’.
European Medicines Agency Schedules Workshop on Psychedelics for April 2024
On the 16th and 17th of April, 2024, the European Medicines Agency (EMA) will host a multi-stakeholder workshop on psychedelics, “towards an EU regulatory framework”. The workshop will be chaired by EMA’s Chief Medical Officer, Steffen Thirstrup.
The workshop will be broadcast live. Further information and a draft agenda are available here.
Australia’s TGA Issues Psilocybin and MDMA Draft

As we have covered extensively (see, for example, Pα+Library: Bifurcated Scheduling), on July 1st 2023 Australia’s Therapeutic Goods Administration (TGA) enacted a limited rescheduling of “medicines containing the psychedelic substances psilocybin and MDMA” when used in the treatment of treatment-resistant depression and PTSD, respectively.
But, given that there is no approved drug product containing either MDMA or psilocybin in Australia, questions have arisen regarding quality standards.
In response to such questions, TGA has issued a consultation paper titled, Proposed quality standards for MDMA and psilocybin. The proposed standards were developed, “in consultation with domestic and international manufacturers supplying MDMA and psilocybin for clinical trials”, according to the document. As such, the “proposed tests, methodologies and limits are consistent with manufacturing requirements currently applied to the medicines used in clinical trials”, and thus TGA says it expects “minimum disruption to supply” following the standards’ introduction. (Note, however, that Australia’s MDMA aggregate trial participant count—for example—is very low.)
In terms of psilocybin, the proposed quality standard “restricts the medicine to a single species of mushroom – Psilocybe cubensis”, as well as synthetic psilocybin.
In terms of timelines, comments may be submitted until January 31st, 2024. TGA expects to finalise its quality standards for MDMA and psilocybin in February 2024, registering them in March.
Other Policy and Related Stories
- Treat Psychedelics Like Cannabis? (Dec 13) ↗ Vital City
- Provincetown moves to decriminalize psychedelics for adults (Dec 14) ↗ WBUR
- Could a little-known psychedelic drug treat opioid addiction? Kentucky wants to find out. (Dec 13) ↗ NBC News
- Reminder: Don’t Put Your Medical License At Risk (Dec 12) ↗ On Drugs
- To Revive Portland, Officials Seek to Ban Public Drug Use (Dec 11) ↗ NYT
- Pricing and reimbursement mechanisms for advanced therapy medicinal products in 20 countries (Nov 28) ↗ Frontiers in Pharmacology
- WHO Publishes Mental Health Gap Action Programme (mhGAP) guideline for mental, neurological and substance use disorders ↗ WHO
- Police say $49K in ‘psychedelic mushroom’ products seized in raid of store (Dec 14) ↗ Global News
- The Government of Canada announces the creation of the Canadian Drug Agency (Dec 18) ↗ Government of Canada
Stay Informed in 2024 with Pα+
Subscribe to Pα+ to receive all of our Bulletins, deep dives and analysis, as well as access to the Pα+ Library and other subscriber-only content.
Your subscription goes directly toward supporting our coverage, not glossy graphic design or marketing budgets. Learn more and subscribe here, or get in touch to discuss team plans.

Featured Psychedelic Jobs
There are some fantastic opportunities on our psychedelic job board at the moment, including:
- Program Director at UC Berkeley’s Center for the Science of Psychedelics.
- Executive Director at the Healing Advocacy Fund.
- Sr. Director, US Communications at Compass Pathways.
- (Associate) Director of Regulator Affairs at atai Life Sciences.
Browse more roles and get more job posts to your inbox by signing up for alerts here. Make an account to join our free talent pool, too.
Business and Commercial
Terran’s Fast-Follower Strategy
Earlier this year, we published a deep dive into Terran Biosciences’ apparent intellectual property strategy. We concluded by asking a question:
One might also ask why Terran would repeatedly wade into crowded waters, targeting compounds that are subject to existing IP claims (in some cases, from multiple parties) and found in the pipelines of other companies. In many cases, if Terran sought to develop and commercialise the candidates claimed in its own IP portfolio, it would need to hope that others’ doors were open, too.
Terran, which had been very quiet on its strategy until that point, has now begun publicly sharing its own narrative around strategy.
Perhaps most notably it has done so via an hour-long talk and Q&A at Wonderland Miami (one of multiple appearances that Terran’s CEO, Sam Clark, made in the conference’s agenda) and a press release dated December 8th.
In his Wonderland presentation, Clark shared that his company has developed ‘the world’s first psilocybin salt’, which he said ‘enables Terran and our collaborators to [pursue the] 505(b)(2) pathway … 5 years after psilocybin is approved’. This strategy was reiterated in the aforementioned press release, which announced the grant of four patents, two of which cover psilocybin Hcl and edisylate.
“Terran plans to use its new forms of psilocybin to pursue a rapid 505(b)(2) FDA approval pathway if one of the existing forms of psilocybin currently in clinical trials is approved”, the press release explained.
(A 505(b)(2) New Drug Application (NDA) allows an applicant to rely on studies conducted by a third-party, regardless of whether that party lends permission to do so. As such, it effectively allows an applicant to request approval for a substantially similar drug product without repeating the drug development process from scratch. The applicant must demonstrate similarity to the approved drug product.)
In his talk, Clark mentioned that his company’s discovery of three new psilocybin salts ‘gets around the idea of monopolising all of the polymorphs’. Here, he’s clearly referring to Compass Pathways’ granted patents covering some forms of psilocybin, though it’s not clear that Compass’ IP strategy relies on a total monopolisation of psilocybin polymorphs.
Clark and Terran are keen to position themselves as “moving quickly toward the goal of providing patients with affordable and accessible psilocybin treatment options”. However, it’s not yet possible to be sure of the company’s motive. As we explained in our deep dive, Terran has repeatedly filed patents on certain forms of other companies’ drug candidates, very soon after they came to light.
Take Delix’s tabernanthalog, for example. The compound was generated by David Olson’s lab at UC Davis and is under development by Olson’s drug development company, Delix Therapeutics. However, as we described in our analysis, Terran has sought to claim various forms and methods of using tabernanthalog in its own patent filings.
As announced in the aforementioned press release, Terran has received a notice of allowance for a patent covering a “novel form of tabernanthalog monofumarate and the method of use for treating neurological and psychiatric disorders.”
Is Terran’s primary motivation to provide patients with affordable and accessible treatment options by busting potential ‘monopolies’ after their respective data exclusivity elapses? Or, is it looking to piggyback others’ innovation and generate revenue by licensing out its IP to the highest bidder? Only time will tell.
The Price Isn’t Right? Market Reacts to FDA Approval of Gene Therapies
On the 8th of December, the FDA approved two gene therapies for the treatment of sickle cell disease (SCD): bluebird bio’s lovotibeglogene autotemcel (lovo-cel), marketed as Lyfgenia, and Vertex Pharmaceuticals and CRISPR Therapeutics’ exagamglogene autotemcel (exa-cel), marketed as Casgevy.
The decision came after the UK’s Medicines and Healthcare products Regulatory Agency (MHRA) approved Casgevy, which marked the world’s first CRISPR-based gene-edited therapy to win regulatory approval.
One might expect such news to positively impact a company’s share price, but that’s not what happened in either case.
Shares of bluebird, which were trading around the $4.26 mark on the 1st of December, dropped to $2.86 on the day of the approval announcement. The drop in share price was the most significant since the stock began trading over ten years ago in June 2013.
Vertex stock, meanwhile, took a slight ding: down around 1% on the day of approval.
Granted, Vertex is a much larger company boasting a market cap of over $100 billion, versus bluebird’s $350 million; and the latter is entirely focused on gene therapies with Lyfgenia marking its first approved treatment.
But what else might explain these very different reactions to concurrent approval news?
Now that these treatments are approved, the focus turns to commercialisation; of which pricing and reimbursement are major elements.
So let’s look at pricing: Casgevy has a U.S. list price of $2.2 million per patient, while Lyfgenia’s is set at $3.1 million.
Lyfgenia’s significantly higher list price, coupled with a black box warning related to instances of blood cancer (which Casgevy does not feature), represent substantial headwinds for its launch. (Another headache for bluebird was that it did not receive a priority review voucher from FDA, which it had intended to sell to Novartis for $103m.)
Investors and analysts appear concerned regarding payor appetite to cover the treatment.

Presumably hoping to assuage such concerns, last Thursday bluebird bio announced that it has signed an outcomes-based agreement with a commercial payer representing around 100 million covered patients in the U.S., and added that it’s in “advanced discussions with a number of the nation’s other large commercial payers and more than 15 Medicaid agencies collectively representing 80% of individuals with sickle cell disease in the U.S.” The company’s share price recovered slightly to $3.30 upon the news.
With cash runway through Q2 2024, it will be interesting to see whether bluebird is able to get payors on-side and reach profitability in the coming quarters. Given that it missed out on $103m in non-dilutive financing that it expected to realise through the sale of a priority review voucher, the company may be in the market for alternative financing soon.
While bluebird’s Lyfgenia looks expensive next to Casgevy, the latter is not cheap, either. This raises the broader question of whether such treatments are fit for market, or whether they resemble something akin to ‘concept cars’ that demonstrate capabilities ahead of their widespread, commercial viability.
Tryp Therapeutics to Combine with Exopharm Limited
Tryp Therapeutics, which claims to be developing oral psilocybin and IV psilocin for several indications including binge eating disorder and fibromyalgia, has entered into a definitive agreement with Exopharm Limited under which Exopharm will acquire all of the issued and outstanding common shares of Tryp.
The transaction is valued at around US $10 million. The combined entity is then expected to relist on the Australian Securities Exchange (ASX), where Exopharm had formerly traded with a market cap of around US $3m. Exopharm—which describes itself as “a leader in exosome technology”—would need to raise at least AUD $6m (US $4m) via a public offering in order to relist.
Group Therapy May Save Labour and Costs
Using data on the costs associated with psychedelic therapy at two trial sites (Sunstone Therapies’ Compass-funded study of psilocybin group therapy in patients with cancer and major depressive disorder (MDD) and a Social Neuroscience & Psychotherapy (SNaP) Lab, VA Portland Health Care System study of MDMA-assisted group therapy for veterans with PTSD), Elliot Marseille et al. (2023) compared the resource intensity of group protocols to individual.
The study found that group therapy “saved 50.9% of clinician costs for MDMA-PTSD and 34.7% for psilocybin-MDD, or $3,467 and $981 per patient, respectively.” In terms of how this impacts the number of clinicians needed to treat, the paper finds:
“To treat all eligible PTSD and MDD patients in the U.S. in 10 years with group therapy, 6,711 fewer full-time equivalent (FTE) clinicians for MDMA-PTSD and 1,159 fewer for FTE clinicians for psilocybin-MDD would be needed, saving up to $10.3 billion and $2.0 billion respectively, discounted at 3% annually.”
As such, the study finds that group therapy could provide a double-whammy benefit: both significantly reducing costs and clinician labour intensity.
Mydecine Clarifies January 2022 Statements Regarding Special Access Program
In late November Mydecine Innovations Group issued a press release “to clarify certain statements made in the Company’s news release of January 13, 2022”. That press release announced the launch of the company’s “Special Access Support and Supply Program in Canada”, which it said would “provide products and services to physicians, clinics, and hospitals in Canada who are looking to treat patients through psychedelic-assisted psychotherapy.”
“With the new addition of psilocybin and MDMA to the approved list of substances under Health Canada’s Special Access Program (SAP)”, the company announced in the January 2022 press release, “the company aims to fill a critical gap in the market by allowing thousands of healthcare professionals and clinics the resources needed to bring these treatments to patients in need.”
It appears someone—perhaps Health Canada—was not best pleased by that press release. In its November 2023 release, the company noted that psilocybin and/or MDMA is provided through its “partner”, Applied Pharmaceutical Innovation, and that “Mydecine does not directly have the ability to grant patients access to controlled substances through the Program.”
It also added that “any previous references to the Program in its press release, social media or website was not intended to promote the availability of unauthorized drugs to Canadians, nor to suggest that these unauthorized drugs have any authorized therapeutic uses in Canada.”
Other Business and Commercial Stories
- Compass Pathways appoints Teri Loxam as Chief Financial Officer (Dec 7) ↗ Press Release
- Beckley Retreats Launches Signature Program for Personal Discovery and Transformation (Nov 28) ↗ Press Release
- Beckley Retreats Announces Expanded Scholarship Program, Alongside New Specialty Programs for Underserved Communities (Dec 11) ↗ Press Release
- ATMA and Cena Life Launch Nationwide Psychedelic-Assisted Therapy Healthcare Network (Dec 7) ↗ Press Release
- Clearmind Medicine Announces Positive Results from its Weight Loss and Metabolic Disorder Program with its Proprietary Psychedelic- Based Treatment (Nov 28) ↗ Press Release
Selected Media Clippings
- Daily Mail: Bizarre moment naked woman on magic mushrooms wraps her leg around a passenger and slaps a cop in wild rampage at Chilean airport (Nov 21)
- Courier Journal: Ibogaine helped me beat heroin addiction, but I oppose Kentucky funding psychedelic research (Dec 17)
- The Guardian: Magic mushroom use grows in England and Wales, ONS figures show (Dec 14)
- VICE: Psychedelic Toad Hunters Are Under Attack by Mexican Cartels (Dec 11)
- Independent: Deontay Wilder claims he is ‘reborn’ after taking psychedelic South American drug (Dec 19)
- Oregon Live: Over 700 people used psychedelic mushrooms under Oregon’s program in 2023 (Dec 19)
- The Guardian: Peyote is the darling of the psychedelics renaissance. Indigenous users say it co-opts ‘a sacred way of life’ (Dec 19)
Join thousands of psychedelics insiders
Get the weekly psychedelic medicine briefing.
A free weekly digest covering trials, regulation, policy, and access.
By signing up, you agree to our privacy policy. You can unsubscribe at any time.