
Breaking: Lykos Therapeutics Moved to Patent MDMA In Late 2022, Filing Reveals
Written by Josh Hardman and Noah Smith with support from Editor-at-Large and patent attorney Graham Pechenik of Calyx Law.
In December 2022 a small group of MAPS PBC’s (now Lykos Therapeutics) employees, consultants and lawyers oversaw the filing of the first in a series of four provisional patent applications seeking to cover MDMA having specific particle sizes (US63/430,287). A Patent Cooperation Treaty (PCT) application claiming priority to this and three other provisional applications1 was filed on December 4, 2023, and published today.
Over the past few years, sources at the company have repeatedly told Psychedelic Alpha that it had not filed any patents, to their knowledge, meaning it’s unclear the extent to which portions of its staff were left in the dark regarding its IP strategy.
As such, while some had noticed a softening stance on patents in the past year or so among company execs, even some of its employees might be a little surprised by today’s publication. Whether the company kept shtum out of concerns for information leakage—as is customary at many firms—or whether leaders at Lykos knew it might cause consternation internally is a matter of speculation.
But it might come as a shock to many in the broader psychedelics field, too.
Just last week, for example, neuroscientist and Tactogen CEO Matthew Baggott noted on Twitter that the company “doesn’t have any patents” (which, it’s worth noting, remains true today; the company’s application remains pending), but might gain leverage through things like market exclusivity and REMS-related matters. And, speaking at the ICER meeting in late May, Heroic Hearts Project’s Jesse Gould told the panel that, “The fact that this unpatentable molecule has made it this far [is remarkable].”
It is true that messaging from the company on IP-related matters has become increasingly confusing of late, with some at least tacitly maintaining the company’s anti-patent stance while others offered equivocal answers that… in light of their equivocalness… suggested the company had indeed filed.
For example, when journalist Olivia Goldhill asked Lykos COO Michael Mulette whether the company had filed any patents, he replied: “We have no patents that we’ve openly discussed”. That was last June, after the company had filed all four of its provisional patent applications. It was enough of a hint, at least, for Olivia Goldhill to proclaim in the title of her post on LinkedIn that this marked “The end of an anti-patent era” for MAPS and Lykos.
In this piece, we unpack the present patent application before recounting a brief history of MAPS and Lykos’ changing stances on patents and starting to speculate on what the future might hold in light of this development—although more will follow in our monthly Psychedelic Patent Analysis series, after we’ve had time to digest.
Looking Under the Hood of Lykos’ Patent Apps.
Lykos’ published patent application, broadly, seeks to cover pharmaceutical compositions comprising MDMA and other excipients, methods of treatment using the compositions, and processes of their preparation.
Generically, MDMA itself has long been regarded as an unpatentable molecule (and as a compound itself, of course is). Nevertheless, Lykos, like other psychedelic drug developers, is pursuing claims to specific forms and formulations of the compound using narrowed claim limitations (see: Pα+ Psychedelic Patent Analysis: February 2024).

While other efforts have been focused on specific characterised salts and solid forms or single enantiomers and enantiomeric ratios, in this instance, Lykos has put its focus on the average particle size of the claimed compositions.
The range and distribution of the particle sizes of the active pharmaceutical ingredient (API) in a drug formulation can affect the drug’s flowability, processability, stability, and shelf life as an ingredient, as well as its dissolution, absorption, and bioavailability—and hence efficacy—in the body after administered. As a key parameter for pharmaceutical manufacturing, particle size patents are common in the industry, and have been granted (and later upheld as non-obvious and inventive), especially where the claimed particle sizes contribute to specific advantages over others.
Specifically, Lykos’ patent application claims a composition comprising a salt or solvate of MDMA along with one or more excipients, where the average particle size in the composition is, at its broadest, between 50 and 400 microns, and, at its narrowest, between 100 and 200 microns. From the claims, it is not immediately clear whether those particle size limitations are intended to apply only to the MDMA API or to the average size of all particles, excipients included, in the composition (the process claims for instance only describe milling the MDMA). Claims 1-3, for example, recite:

Further dependent claims cover specific MDMA salt and solvate forms that are used in the composition, dosages, excipients, and dosage forms, all of which, together, presumably cover the formulation of MDMA HCl Lykos is now seeking approval for.

The company received a written opinion from a US patent examiner acting as the international searching authority (ISA) for the first three claims of its application (claims 4-32 contain “multiple dependencies” and were not entitled to an opinion). While the claimed compositions, with aforementioned particle size limitations, were found to be novel, the examiner found the same claims to be obvious in view of Awakn Life Sciences’ WO2022/150525 PCT, on MDMA for alcohol use disorder.
Notably, the examiner wrote that, while the precise particle sizes (or size ranges) were not specifically disclosed in the reviewed prior art, “it would have been obvious to one of ordinary skill in the art to change the particle size through routine experimentation.” To support their conclusion, the examiner pointed to disclosure in Awakn’s ‘525 PCT that “in preparing a formulation, it may be necessary to mill the active agent to provide the appropriate particle size.”
Of course, even if novel, an invention must also be non-obvious to be patentable. While this opinion is therefore unfavourable, it is important to note that the ISA’s findings are not binding, and findings of obviousness are generally to be expected at the PCT stage. Once in national prosecution, the burden is generally on the applicant to make its case for non-obviousness, for example with data demonstrating the surprising or unexpected effects of its invention (such as the advantages of the claimed particle sizes), and by amending and narrowing its claims. Further, only the first 3 of the company’s 32 claims in the application were reviewed by the examiner for this written opinion. Limitations from the further dependent claims (or additional limitations, imported from the specification) could still be found sufficiently novel and non-obvious to be patentable, especially when supported by evidence of unexpected effects.
In addition to pointing towards the company’s desire to build a moat around the subject of its FDA New Drug Application, its MDMA-assisted therapy for PTSD intervention, the patent application also seems to point towards the company’s forthcoming ambitions for its Midomafetamine product.
In the application, Lykos unsurprisingly claims the use of its composition for the treatment of PTSD. Interestingly, however, the only other indication recited in the application’s claims are eating disorders. In fact, the company’s second- and third-filed provisional applications are focused almost exclusively on methods of treating various eating disorders. Indeed, while the first and fourth provisionals (like the PCT application) are entitled “Compositions Comprising MDMA and Methods of Using Same”, the second and third provisionals are entitled “Methods of Treating Eating Disorders”. As such, it may be the case that eating disorders such as anorexia nervosa, bulimia nervosa, or binge-eating disorder are among the next target indications Lykos hopes to pursue approval for.
A Brief History of MAPS & Lykos’ Patent Philosophy
In order to contextualise today’s revelation, it’s worth looking back at some of MAPS’, and Lykos’, history with regard to how the entities have discussed the role of intellectual property in their efforts to bring MDMA to the masses.
Learning from Ibogaine, Doblin Lands on ‘Anti-Patent Strategy’
While debates around patenting psychedelics have reached a crescendo in recent years, MAPS’ founder, Rick Doblin, has been engaging with the topic for around four decades.
In the summer of 1985, a year before he founded MAPS, Doblin published ‘A Proposal for Orphan Pharmaceuticals Inc.: A Psychedelic Medicine Pharmaceutical Corporation’.
“The non-patentable status of MDMA deserves some analysis”, he wrote in an early section of the report, noting that while bog-standard MDMA “could not be patented”, “a version of MDMA designed for maximum assimilation into the body could be.”

While obtaining a patent on the drug itself might be tricky—Merck had patented MDMA over a hundred years ago in 1912 after it found it to be a potentially useful intermediary in the production of a styptic product2—Doblin pointed out that “it might be possible to receive use patents for MDMA”, citing the example of “a New York physician”, who had received a use patent for ibogaine that same year. (Doblin was referring to Howard Lotsof, who sought to develop ibogaine for addictions after finding its utility in managing his own heroin addiction.)
Almost prophetically, given today’s entactogen drug discovery activity, Doblin also highlighted that “there are several new compounds that could be patented that deserve experimentation”, explaining that Orphan Pharmaceuticals, if formed, would “explore various possible chemical modifications of the basic MDMA molecule, and to seek to determine which substance to put through the lengthy testing process.” In fact, this would be the first line item on the prospective company’s research agenda, he said.
That ibogaine IP, however, turned out to be a warning signal for Doblin. Lotsof had filed for patents covering the use of ibogaine in the treatment of substance use disorders via his newly-formed company, NDA International. Shortly thereafter, however, the nascent ibogaine drug development scene was apparently thwarted by litigation between various parties.
Worried by these developments, Doblin engaged Lotsof’s patent attorneys to review the MDMA IP landscape, scoping out whether MDMA use patents existed. “I decided to hire a patent attorney to try and insure [sic] that MDMA’s potential medical use would not be bottled up by anyone else’s use patent”, he recalled in a Winter 1992-93 issue of the MAPS Newsletter, adding that “[i]t could be a major setback if MAPS funded MDMA research and secured approval for its medical use only to have someone else control it through an inexpensively obtained use patent”. (Lykos’ patent certainly wouldn’t be inexpensively obtained, it’s worth noting!)
This was the crux of the organisation’s ‘anti-patent strategy’, under Doblin, which saw large swathes of protocols, data, and methodologies deposited into the public domain—a practice unthinkable to mainstream pharma companies—and reliance on non-patent forms of exclusivity3 to recoup the costs of bringing a drug to market.
While Doblin had anticipated contemporary debates and dynamics around psychedelic patents, and was cognisant of the potentially deleterious effects that IP disputes had had on ibogaine’s early drug development push, he could not have anticipated the quantity and character of patent-related activity that has unfolded in the past half-decade.
Indeed, writing in an op-ed for Psychedelic Alpha in January 2023 he said precisely that: “At the time of the creation of MAPS PBC, I did not anticipate the rise of hundreds of for-profit psychedelic pharmaceutical companies that would follow our research playbook.”
“They also follow the traditional pharma playbook,” he continued, “patenting everything from novel methods of manufacturing psychedelic drug products, to new molecules that may or may not offer advantages over the psychedelics already in the public domain.”
In hindsight, might Doblin have been implicitly painting Lykos as one of those companies following the ‘traditional pharma playbook’, given the present patent was filed just a month prior?
Beyond Method of Use Patents
Despite his foresight, it seems that Doblin had not anticipated that this activity would extend to the filing of patents other than use patents, such as composition of matter and methods of manufacturing MDMA drug product itself.
In that 2019 Boston Magazine interview, for example, Doblin explained: “MDMA was invented in 1912, so there’s no composition-of-matter patent”, adding that “you can still get usage patents for different uses.” That’s why he had hired the lawyers to “develop a strategy so nobody could ever patent any of the uses of MDMA.”
By Spring 2021, however, Doblin was acutely aware of the risk that someone might pursue claims over “ways of manufacturing and encapsulating MDMA”, telling journalist Shayla Love that, for MAPS, “It’s not off the table.”
By this point, MAPS PBC, as it was still named, had made its treatment manual freely available online, and in December 2021 it published its “Fully Validated, Multi-Kilogram cGMP Synthesis of MDMA” in an open access journal article.
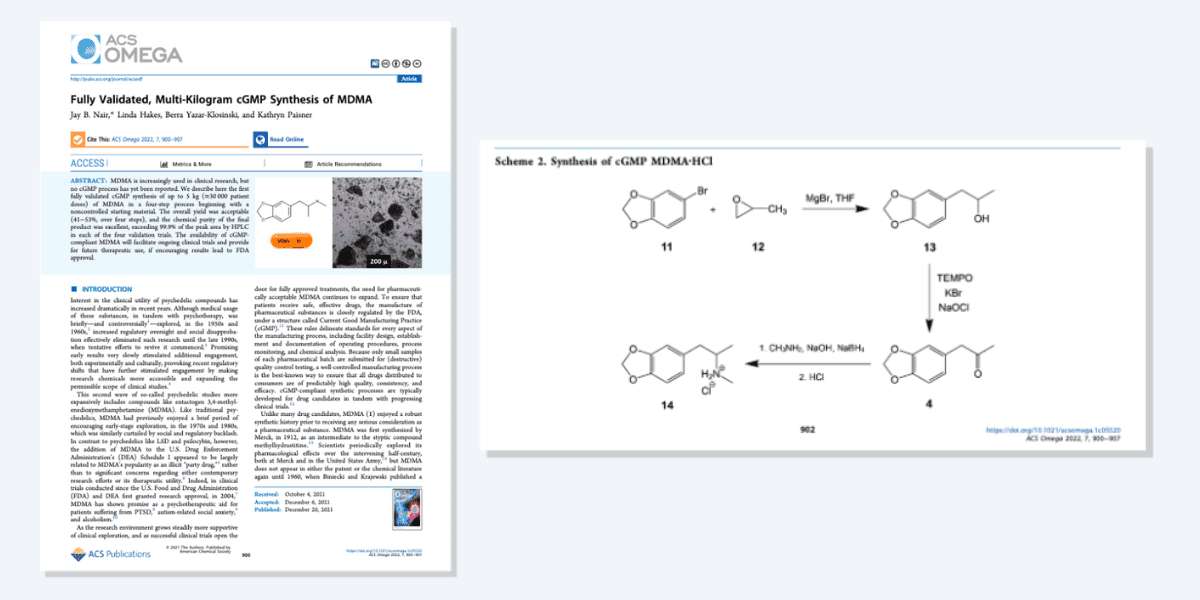
So, the bulk manufacturing method of MDMA and its hydrochloride salt was thrust into the public domain, constituting prior art. The method described in the paper yields enough MDMA for around 30,000, it claims.
Indeed, a MAPS press release explicitly noted that “[p]ublication of this process established ‘prior art’, contributing to MAPS’ patient access strategy by making intellectual property public.”
However, that publication did not mention later stages in the path to producing dosage-form-ready forms of MDMA, like the milling of MDMA to achieve specific particle sizes. It was these elements that Lykos sought, a year after the publication of the open access methods article, to protect with patents.
One might presume Doblin would dissent from this move, given that in March 2021 he Tweeted a statement explaining that MAPS, “will also make public the IP that we have created relating to the production and encapsulation of medical-grade MDMA in commercial quantities, rather than seeking to patent that information.” (Doblin was responding to concerns raised by Tim Ferriss around “the patent land grab warming up in the for-profit psychedelic world.”)
While it’s unclear the extent to which Doblin might have dissented from the patent’s filing, it’s very likely that his younger self would be shocked that his nonprofit’s progenitor would be responsible for filing such claims.
A Change of Name, and Character?
So, did Doblin do a complete 180 on his oft-mentioned anti-patent strategy? Probably not. It’s more likely that MAPS PBC, now Lykos, became increasingly influenced by the gravitational pull of capital versus the wishes of its nonprofit owner, MAPS.
Indeed, in late 2021 the nonprofit announced a revenue financing model led by a venture capital fund that would see a share of future revenues handed over to its financiers. Importantly, however, the nonprofit did not cede governance or equity to these outside backers, retaining full control of MAPS PBC.
But this non-equity fundraising effort didn’t see the organisation through to FDA approval, and the company ultimately took on equity investment in the form of a $100M+ Series A and concurrent rebrand to Lykos, announced in January 2024.
While the lead investors in Lykos were touted as ‘mission-aligned’, their backing was not philanthropic. So, in order to shore up investor returns and lay the runway for future rounds, it’s likely that Lykos took on more of a conventional pharma sheen than some of its earlier supporters might have been comfortable with.
While Doblin and co. might have hoped that a five-ish year period of non-patent exclusivity might be enough to make their funders whole, the reality of just how challenging the commercialisation of MDMA-assisted therapy might be (with a “limited roll out” that’s “staged and controlled” (See An Embattled Lykos Seeks to Reassure Committee, Agency with Slow and Steady Roll Out) metering market penetration and thus revenues) might have led Lykos to seek additional exclusivity via patent protection.
As such, this patent filing is likely best understood within a broader narrative arc of Lykos, which (as we wrote in our earlier coverage) “appears to be entering something akin to puberty” whereby it aims “to simultaneously find its own voice and direction while respecting its lineage and embodying key tenets of the MAPS mission.”
With the announcement of the present patent filing, it’s only natural that some will think the pendulum has swung too far.
How Will Lykos Wield Its Patents, If Granted?
But it remains to be seen how Lykos will wield its patents, if issued.
Just because it has filed for patents, it’s not a given that the company would choose to wield them in the manner that a conventional pharma company might, such as through inflicting 30-month stays on would-be generic entrants, and trying to block all entrants until the patent expires (at least).
Indeed, in Spring 2022 the company’s CEO, Amy Emerson, told Life Science Leader that it “is not interested in using patents or intellectual property to discourage other companies from competing in the emerging psychedelic-assisted therapy space.”
In a statement sent to the publication, Lykos continued: “Intellectual property tools also can be applied within a patient access framework to promote health equity, and if we were to pursue a patent it would be for that purpose.”
It’s unclear what that means. Is it possible that, even if it does wield its patent to block others from entering the MDMA treatment market, Lykos may still claim to be doing so in service of the public benefit?
At least one narrative could be spun, here. The company has previously emphasised that portions of revenues from the sale of its drug product would be recycled into patient benefit activities, for example (See Looking Ahead at Patient Access to MDMA-Assisted Therapy), so it could argue that its use of patents to enforce a longer period of exclusivity would support greater levels of patient benefit activities.
However, without knowing things like Lykos’ intended pricing of the MDMA drug product, it’s difficult to appraise whether such a scenario would lead to better patient access and public benefit than the speedier entry of generic, cheaper MDMA.
Of course, another big ‘if’ is whether the company will even obtain FDA approval for its MDMA-assisted therapy at all (see FDA Advisory Committee Snubs MDMA-Assisted Therapy for PTSD in Overwhelming “No” Vote).
Next Steps
On the procedural side, it could be some time before Lykos gets clarity on the strength of its patent applications.
The company now has until June 5, 2025, 30 months from its earliest priority date, to file a national stage application in the United States (and in most other jurisdictions). Should the company pursue the standard prosecution pathway, at the point of filing Lykos can, based on present pendency data from the USPTO, expect to wait a little over 20 months before receiving a first office action, and would expect at least another 6 months to a year from that until receiving an issued patent (if the claims are allowable); thus, perhaps around the start of 2028. (Lykos also could enter national phase early, and could pay to “fast track” its examination in the US, potentially resulting in a granted patent in under a year, but even the lengthiest of examinations look to place a final patent outcome comfortably before the expiration of any future data exclusivity.)
Assuming the company gets approval for its drug candidate, should Lykos eventually see its patent granted it will undoubtedly seek to have it included in FDA’s Orange Book prior to exhausting its 5 years of data exclusivity. Such an inclusion would then allow the company to attempt to delay generic or 505(b)(2) entry for an additional 2.5 years by way of a 30-month stay. That is, presuming that the entrant’s drug product could be alleged to infringe at least one of Lykos’ patent claims—Terran, for instance, which has recently patented novel salts and polymorphs of MDMA hemifumarate, asserts that it “has prepared in advance to get around those 30-month stays”.
Were Lykos’ patent to stand up to invalidity challenges, it could potentially extend Lykos’ exclusivity against generic applicants until its expiry, which would be at least until December 4, 2043 (plus any applicable patent term adjustment or extension). Potentially—because a generic manufacturer would still have to infringe; advice by one pharmaceutical consultant (as quoted in an FTC complaint) is that “all generic drug companies know” that particle size patents “may be easily circumvented” (of course, the ultimate breadth of any issuing claims from Lykos’ application remains to be seen).
Moreover, Lykos’ isn’t the only MDMA IP at play—despite having a first shot on goal at approval, several other companies are jostling to secure a slice of the entactogen patent landscape (see Patenting MDMA, its Enantiomers and Mixtures Thereof). Hence, apart from generic and 505(b)(2) competitors seeking approval for the same or another racemic MDMA drug product, Lykos may have to compete with companies developing other forms of MDMA, too.
As for Doblin’s mission to bring genericised psychedelics to market, there’s still reason to believe it’s something he’s pursuing.
The MAPS founder has said on the record, for example, that ibogaine—the alleged victim of that earlier psychedelic patent war—could be a good candidate for developing in an open science manner, “to be sold as a generic medicine immediately after approval.” There are efforts underway to appropriate state-level opioid abatement funds for this purpose, too.
Others might look to different geographies, like Australia or the Netherlands, which could be fertile ground for realising paths to MDMA access that don’t rely on conventional drug development channels.
Pα: While some might be surprised or even upset by today’s development, others might ask: Why shouldn’t Lykos have filed for a patent? Given that hundreds of other entities have entered the scene and sought to cleave off portions of the psychedelic drug development landscape for themselves, isn’t it only natural for Lykos to do the same for themselves, even as a defensive measure?
In any case, it will likely be the manner in which the company chooses to wield this patent—if issued—against present and future competitors, if at all, that will decide its impact on patient access and public benefit.
There’s plenty more to unpack here, so look out for further analysis and reaction via our regular Psychedelic Patent Analysis series (if you haven’t already, join Pα+ today to receive these premium insights). ∎
Join thousands of psychedelics insiders
Get the weekly psychedelic medicine briefing.
A free weekly digest covering trials, regulation, policy, and access.
By signing up, you agree to our privacy policy. You can unsubscribe at any time.
- Subsequent provisional applications claim methods of treating an eating disorder with MDMA having specific particle sizes (US63/449,928 and US63/463,169, filed March 3, 2023 and May 1, 2023); and claim further compositions and dosage forms of MDMA having specific particle sizes (US63/463,170, also filed May 1, 2023).
- Speaking in 2000, Julie Holland explained that, “Merck was looking for a good vasoconstrictive substance to use as a styptic, to stop bleeding, and when they stumbled across this chemical in their synthesis, as any good chemical company would do, they patented their finding.” In 1985, a statement from a group of psychedelic researchers and practitioners read: “That patent has expired, and MDMA is not under patent and cannot be patented. There is no financial incentive to research MDMA.” It was received wisdom, then, that the drug could not be patented; at least among those on the psychedelic research scene at the time.
- Before MAPS settled on PTSD as its lead indication it had explored MDMA in the treatment of pain in end-stage cancer patients, which Doblin had hoped could benefit from Orphan Drug Designation, for example.