
Psychedelic Bulletin #119: More Misery for MindMed; Another Psychedelics Co. Ceases Operations; A Brief Dispatch from ICPR
This Week:
- 📉 More Misery for MindMed
- 🍦 Onsero Therapeutics Looks to Skip the Trip
- ⛔ No Safe HAVN: Another Psychedelics Company Grinds to a Halt
- ⚖️ An Update on Terran vs Compass
- 📓 A Very Brief Dispatch from ICPR
and more…
Psychedelic Sector News
More Misery for MindMed
Earlier this week MindMed announced a public offering priced at $4.25 per unit, which included one common share and one warrant exercisable at that same price. MindMed had closed the preceding trading day at $6.12, meaning the offering represented a significant discount.
Predictably, then, MindMed traded down toward the offering price after hours. It didn’t stop there, though: MNMD closed Friday at $3.50, putting the company’s market cap at around $100m.
All this is happening amidst the spectre of a shareholder activist situation, which we discussed in Bulletin 114. The group are seeking to prevent MindMed from raising dilutive funding, at least in Canada. Perhaps that’s why the press release announcing the offering specifically stipulated that, “no distribution under the underwritten public offering shall occur in Canada or to a person resident in Canada.”
MindMed stock, which trades as MNMD on the Nasdaq, is down over 90% in the past year.
Onsero Therapeutics Looks to Skip the Trip
A team of nearly thirty researchers claim to have identified a new compound that has antidepressant but not psychedelic effects in mice, in a new publication in Nature. The co-authors, which represent institutions such as UNC-Chapel Hill, UCSF, Yale, Duke and Stanford, whittled down a virtual library of 75 million molecules to just seventeen candidates. From there, more conventional drug development practices were applied. Meghana Keshavan provides a write-up of the work in STAT.
The apparent non-hallucinogenic and antidepressant compound (in mice) was patented by UNC, UCSF and Yale and licenced to Onsero Therapeutics, of which Dr. Bryan Roth (a co-author) is a founder. Onsero was formerly known as Noetic Foundry, which was incorporated as Noetic Labs Inc.
No Safe HAVN: Another Psychedelics Company Grinds to a Halt
HAVN Life Sciences “is no longer a going concern”, the company explained in a very short press release, as it “is unable to meet its financial obligations as they come due.” The company will attempt to secure new financing and embark on a restructuring plan, which will surely be a difficult task. The company seemed to be lacking direction for quite some time, too (see our post-mortem of Mindcure, which briefly mentions HAVN).
This makes HAVN the latest psychedelics company to effectively cease operations, and we can’t say we’re surprised: back in July, we reported that HAVN had been “running on fumes for months” according to our data. This presumably explained their decision to enter into what we described as a “Death Spiral Financing” with “alternative financing solutions” provider Alpha Blue Ocean.
The death spiral appears to have come to its logical conclusion, then. It leaves us wondering: which psychedelics company will be next?
An Update on Terran vs Compass
In Bulletin 114 (August 12) we reported on a lawsuit that alleges Compass Pathways “milked” a researcher for trade secrets before “secretly” filing a patent on the same technology.
This week, Compass responded to the lawsuit by lodging a motion to dismiss the complaint claiming that the Maryland Court lacks jurisdiction over Compass (which is headquartered in the UK) and that Terran lacks standing, among other issues.
Readers may review both the motion to dismiss and supporting document.
Your Projects
- Christine Ziemer (Missouri Western State University) has started a Psychedelic Educators Network for resource sharing and meet-ups among those working in higher education that are actively teaching (or are interested in teaching) psychedelics-related courses. Educators interested in joining the network can fill out this form.
- Heather Lundy at Quantum Mental Health is looking to eliminate the cost barrier to psychedelic therapy by providing psychedelic prep and integration at just $75/hour.
- TheraPsil and Drug Science are working on providing access to psilocybin in Canada via the Special Access Program under Project Solace.
Readers can share their projects by replying to any Bulletin email.
Featured Psychedelic Jobs
- Usona Institute is hiring an Ops and Admin Manager.
- Johns Hopkins are looking for a Research Program Supervisor.
- atai Life Sciences are hiring a Director of Venture Operations.
Browse more roles and get more job posts to your inbox by signing up for alerts here.
Weekend Reading
A Very Brief Dispatch from ICPR
I (Josh) had a great time attending the first portion of the Interdisciplinary Conference on Psychedelic Research (ICPR) in Haarlem, the Netherlands. The Psychedelic Science, Ethics & Business day took place immediately prior to the main conference, and offered a space to discuss the rapid development of the so-called psychedelic ‘sector’.
One particularly interesting talk came from Elliot Marseille, a health economist who is director of the Global Initiative for Psychedelic Science Economics (GIPSE) at UC Berkeley and UC San Francisco. Marseille has co-authored a number of publications on the cost-effectiveness of MAPS’ MDMA-assisted therapy for PTSD, which we discussed briefly in a previous Bulletin.
Marseille was keen to reiterate an important caveat to the MDMA-AT cost-effectiveness data: the patients in MAPS’ clinical trials are, in many respects, the most ‘extreme’ PTSD sufferers. As such, we might expect the efficacy of the therapy to reduce when addressing mild or moderate cases of PTSD, which will, of course, have a significant impact on the cost-effectiveness of the modality.
MAPS PBC’s COO, Michael Mullette, also tempered expectations on several fronts. Out of all the challenges and tasks ahead, Mullette said that establishing a suitable pool of appropriately trained therapists was his primary concern. How will MAPS PBC incentivise a therapist to sit with an MDMA-AT patient for eight hours, versus sitting with seven or eight ‘conventional’ therapy patients for one hour, for example?
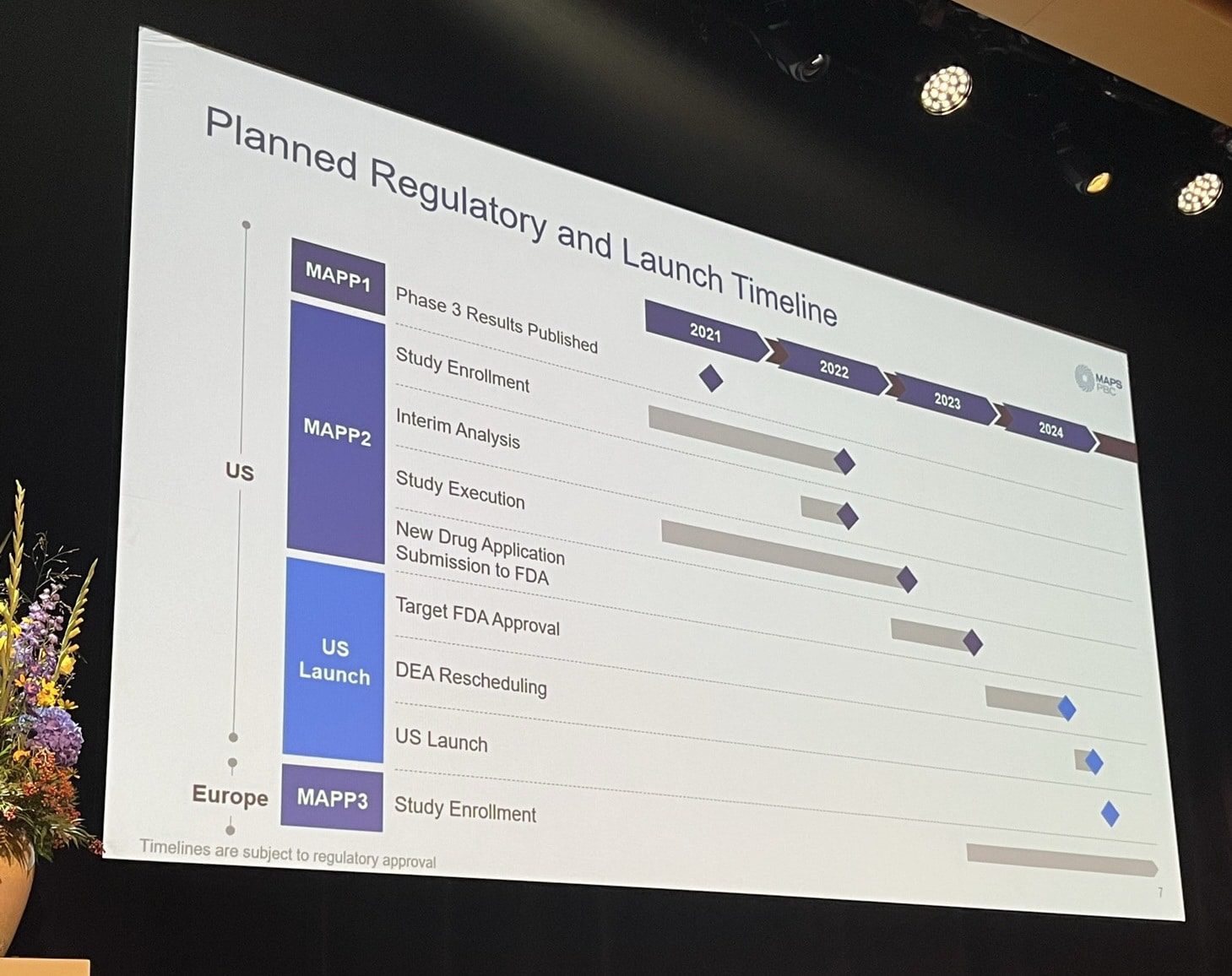
We also noticed that the timeline for a potential approval and roll-out of MDMA-AT in the U.S. has been pushed back slightly. The company is targeting FDA approval toward the end of 2024, with a U.S. launch taking place sometime thereafter.
Elsewhere, The European Research Council (which has a 2021-2027 budget of over €16 billion) sent two representatives to encourage researchers to apply for funding. “We believe that psychedelic sciences have the opportunity to bring ground-breaking outcomes […] to humanity,” exclaimed Lionel Thelen, a Senior Research Programme Officer at the Council.
The interdisciplinary nature of psychedelic research means that they may be considered for a variety of the ERC’s funding categories (or, “panels”, in ERC parlance), not just Life Sciences. What’s more, the Council explicitly encourages interdisciplinary projects through its Synergy grants which allow for, say, a scientist and an anthropologist to serve as principal investigators on a project.
Despite this large pot of funding, and a clear alignment between the nature of many psychedelic research projects and the ERC’s remit, only one psychedelics-related project has been funded by the ERC to date (Healing Encounters by Emilia Sanabria). As someone who has worked on ERC-funded projects in a past life, I implore readers that are engaged in research to explore ERC funding for their work: “the sole criteria is excellence”, explained Thelen (well, that and spending >50% of your time in Europe…), and there are many truly excellent minds in our audience here.
Aside from these specific appearances, I was surprised by the general tenor of the Science, Ethics & Business day, which was decidedly more optimistic about the intersection of psychedelics and business than similarly-billed gatherings. Rather than a blanket aversion to business involvement in research, a number of (certainly not all, by any means) the researchers present expressed an enthusiasm about the prospect of securing corporate funding, and limited their concerns to ensuring they are paid appropriately and secure fair compensation where findings go on to be commercialised.
I was only able to attend the first day of ICPR proper, but was pleased to catch Erika Dyck’s presentation on the global history of psychedelic research. Looking at the dominant repositories of scientific research, such as PubMed and other formal aggregators of research outputs, gives us only a partial picture of the ebbs and flows of psychedelic research over time and geographies.
Dyck shared some impressive visualisations to tell a high-level story: the majority of naturally-derived psychedelics (like mescaline from peyote and psilocybin from mushrooms) research originated in the Americas before finding its way to Europe, whereas synthetic psychedelics charted a different course with LSD, for example, originating in Europe.
For those looking for a more fulsome write-up of ICPR, Blossom’s Twitter threads are the place to go.
Assorted Reading
- Nature Outlook published an issue on trends in psychedelic research (sponsored by atai).
- Politico looks at the challenges regulators face regarding psychedelics.
- The Economist explores the psychedelic renaissance in medicine on its podcast, and in an earlier article.
- A new publication by Neşe Devenot et al. explores the ethical concerns and financial interests that abound in the psychedelics space (shorter write-up here).
- Psychedelic Science Review interviews Dr. Alex Kwan: from dendrites to drug discovery.
- Abigail Calder (who wrote an Opinions piece for us earlier this year) and Gregor Hasler’s review of psychedelic-induced neuroplasticity is well worth a read. If you can’t find the time, you should at least admire Figure 1.
Weekly Bulletins
Join our newsletter to have our Weekly Bulletin delivered to your inbox every Friday evening. We summarise the week’s most important developments and share our Weekend Reading suggestions.
Live Updates
Join us on Twitter for the latest news and analysis.
Other Channels
You can also find us on LinkedIn, Instragram, and Facebook.
Join thousands of psychedelics insiders
Get the weekly psychedelic medicine briefing.
A free weekly digest covering trials, regulation, policy, and access.
By signing up, you agree to our privacy policy. You can unsubscribe at any time.