
Psychedelic Research and Clinical Trials in 2021
Psychedelic Research and Clinical Trials in 2021
A Year in Review
Part of our Psychedelics 2021: A Year in Review Series
Introduction
The resurgence of research into psychedelics has continued to thrive over the past year. Dozens of trials and hundreds of studies have been published in 2021, expanding the depth and scope of scientific inquiry in this flourishing space.
If published findings from this past year are a reliable indicator of what is to come, these investigations will foreseeably expand our fundamental understanding of psychedelics and point to new, promising avenues of exploration.
However, we must also appreciate the methodological challenges inherent in psychedelic research, as well as the broader fields within which such research is nested.
It’s worth reminding ourselves of the lengthy timescales involved in drug development, with clinical trials regularly taking in excess of six years to complete. As such, it’s worth looking at studies announced and approved in 2021, and considering when we may expect to see data readouts from such work.
Just as the funding environment for psychedelic research has heated up significantly in recent years, regulators are also demonstrating an increasingly warm attitude to such work. Toward the end of this section we review some key regulatory developments in 2021, and profile a handful of new psychedelics research centres.
As you will see as you leaf through this report, psychedelic research is showing no signs of slowing. One of the most prolific psychedelics researchers, Robin Carhart-Harris, predicts “more of the same” in 2022, and data confirms that we’re witnessing the most productive period of psychedelic research (at least that which is conducted in recognised institutions).
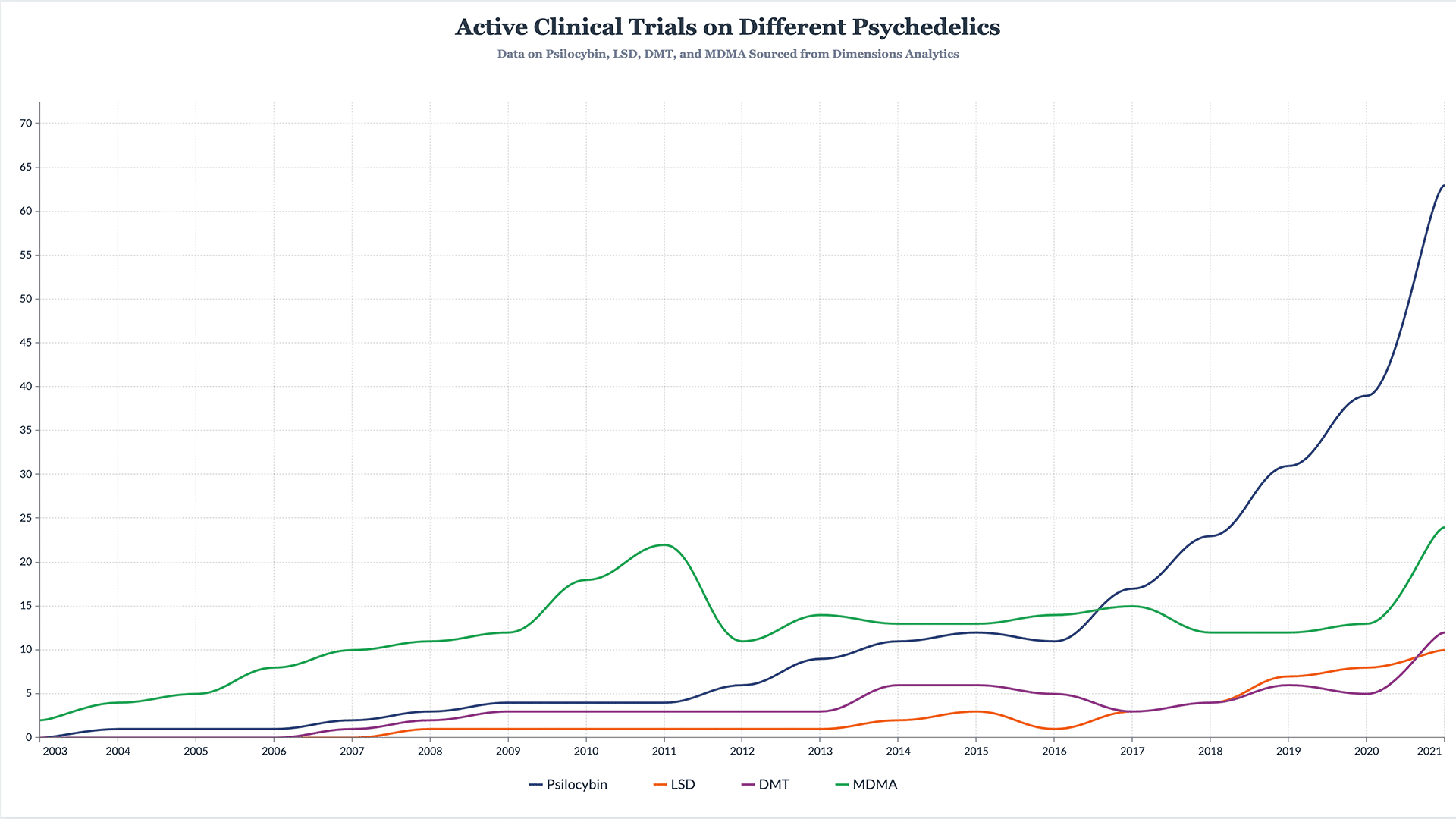
Key Publications in 2021
To begin, here are some of 2021s most notable publications…
Psilocybin Goes Head-to-Head with SSRI Antidepressant
In April 2021 results of a Phase 2 trial evaluating psilocybin-assisted therapy compared with psychotherapy and escitalopram, a common SSRI antidepressant, in the treatment of major depressive disorder (MDD) were published in the New England Journal of Medicine. This was the first randomised, controlled study of psilocybin for the treatment of MDD.
Publication in NEJM—one of the oldest and most prestigious medical journals—is, in itself, a significant moment for psychedelic research. The study was also covered widely in mainstream media outlets, including NBC; Scientific American; BBC; Rolling Stone; and the Guardian.
However, the results didn’t—at least on the face of it—appear to meet the lofty expectations of many. This is partly due to the study’s design, including the measure of depression that was chosen as the primary outcome in the study’s pre-registration.
Speaking to Psilocybin Alpha, Psilera Bioscience’s Co-Founder and CEO Chris Witowski explained, “by primary endpoints [sic] there was no difference in efficacy, however, when you look at other endpoints there seem to be better outcomes trending towards psilocybin.”
We featured exclusive expert commentary from Stanford School of Medicine’s Boris Heifets in an April Bulletin, in which he further explained some of these “very unfortunate (and unlucky) design features” that make this study “difficult to interpret.”
However, despite these methodological misfortunes Witowski explained that, “there is still promise for psilocybin in the treatment of depression but more studies are needed to prove this in the eyes of regulators; the best way to do this and increase access is through top-quality science and clinical research.”
The trial was also profiled in a BBC documentary, somewhat unimaginatively titled, The Psychedelic Drug Trial.
MAPS Publishes Results From Phase 3 Trial of MDMA-Assisted Therapy for PTSD
One of the year’s most anticipated publications came in May, when MAPS released results of its MAPP1 Phase 3 trial investigating MDMA-assisted therapy (MDMA-AT) in the treatment of PTSD.
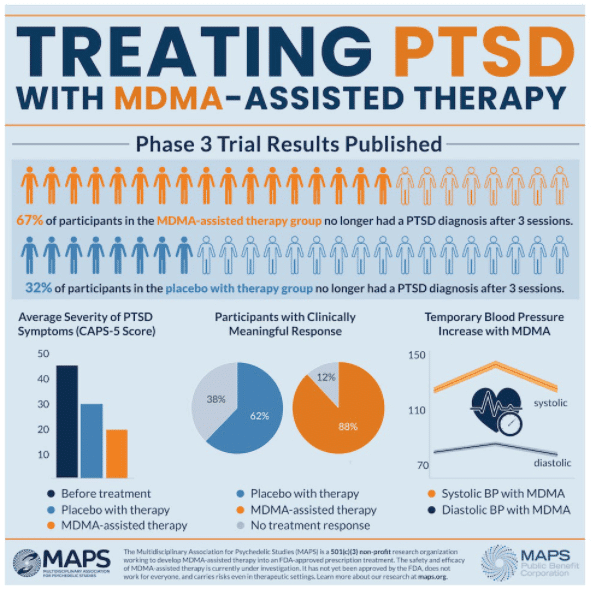
The trial, which enrolled 90 participants, found that 88% of individuals who underwent MAPS’ MDMA-AT protocol experienced clinically meaningful reductions in PTSD symptoms. Perhaps even more remarkable was the fact that 67% of participants in the treatment arm no longer met the criteria for a PTSD diagnosis 2 months post-treatment, versus 32% of those in the placebo group.
In a view shared by many, MAPS’ Founder Rick Doblin characterised these results as “outstanding.” So remarkable, in fact, that Science magazine shortlisted the nonprofit’s work for its 2021 Breakthrough of the Year, dubbing MDMA-AT “a psychedelic PTSD remedy”.
In terms of safety, there were no serious adverse events in the MDMA group. Rather, the two serious adverse events recorded were both in the placebo group. Non-serious adverse events, meanwhile, occurred to a greater extent in the placebo group than the MDMA group.

The study received a great deal of mainstream media attention, including a major write-up in the New York Times: A Psychedelic Drug Passes a Big Test for PTSD Treatment. Other write-ups include those from the BBC, TIME, Fast Company, and a second piece from the New York Times.
This is about as excited as I can get about a clinical trial. There is nothing like this in clinical trial results for a neuropsychiatric disease.”
Some important findings in the publication included:
There is no obvious impact of past SSRI usage. Importantly, the study found that there was “no obvious impact of SSRI history on the effectiveness of MDMA.” This is a reassuring finding, especially in the context of a study published in 2020 that suggested past antidepressant usage reduces the efficacy of MDMA-assisted psychotherapy for PTSD (Note: Michael Haichin authored an informative article for Psychedelic Science Review which covered the study in question). It’s not clear why we see a discrepancy between these findings, but we may expect deeper analysis following MAPS’ second Phase 3 trial. We discuss studies published in 2021 that further elaborate the potential interactions between SSRIs and psychedelics below.
Diversity and Representation. As a field, we still have a significant amount of work to do in terms of representation in clinical trials of psychedelics. Just 2 participants in this trial were black or African American, representing just over 2% of the sample size. For context, according to the US Census Bureau around 12.4% of the U.S. population is black or African American.
A significant body of literature (e.g., Roberts et al., 2010) shows that lifetime prevalence of PTSD is higher in minority groups than Whites, but that minority groups are less likely to seek treatment. While, like many clinical trials, MAPS’ study falls short of representing society in an equitable manner, it is hoped that MAPS will be in a position to deliver MDMA-Assisted Therapy to a diverse range of individuals, should it be approved. MAPS is prioritising this in their second Phase 3 trial, and in their therapist training efforts. Comments from MAPS PBC at Horizons 2021 reinforced the fact that this is high on their agenda.
Overnight Stay vs. Evening Discharge. The study also found that whether or not a participant had an overnight stay following treatment had no effect on the success of the protocol. This was achieved by allowing participants at two study sites to be discharged in the evening, as opposed to being kept at the site overnight.
Haichin points out that the ability to eliminate the overnight stay from the treatment protocol would have a significant impact on cost-effectiveness. However, he also points out that 10/14 participants that requested further integrative visits were in the MDMA arm, indicating that the amount of psychotherapy in the current model may be insufficient for some.
The Blinding Problem. As with most, if not all, psychedelic studies, the elephant in the room is the ability of the researchers to adequately prevent participants from guessing what treatment they received, and potentially influencing the results (more in this later, where we discuss a number of methodological stumbling blocks that were further highlighted in 2021).
Previous MDMA research used low-dose MDMA instead of an inactive placebo in the comparator group, which improved blinding but made participants’ PTSD worse. Since the use of low-dose MDMA made it easier to find a treatment difference (and would be unethical to give), MAPS decided—in partnership with the FDA—that an inactive placebo group was more appropriate for both Phase III trials.
“However, although blinding was not formally assessed during the study, when participants were contacted to be informed of their treatment assignment at the time of study unblinding it became apparent that at least 10% had inaccurately guessed their treatment arm. Although anecdotal, at least 7 of 44 participants in the placebo group (15.9%) inaccurately believed that they received MDMA, and at least 2 of 4 participants in the MDMA group (4.3%) inaccurately believed that they had received placebo.”
In other words, almost 90% of participants guessed whether they received MDMA-assisted psychotherapy or not. Because of this, critics will say the trial is essentially open-label, where the given treatment is known and treatment effects tend to be overestimated.
It’s been no small feat to get this far, Rick Doblin reminded Psilocybin Alpha. “MAPS trained over 800 therapists [in 2021],” he explained. “We still need to supervise each new therapist as they work with their first PTSD patient, and we still need to arrange for those therapists who want to volunteer to receive MDMA themselves in a therapeutic setting as part of their training.”
There’s plenty of work ahead, too. In our Looking Ahead to a Psychedelic 2022 section, we discuss the next steps in MAPS’ path to approval with Doblin himself.
Psilocybin for Treatment-Resistant Depression: COMPASS Pathways’ Phase 2b Results
In June, COMPASS Pathways announced that it had finished administering psilocybin therapy to all patients enrolled in its Phase 2b clinical trial. The company had been investigating the safety and efficacy of psilocybin therapy for treatment-resistant depression (TRD) since March 2019. The study, which enrolled over 200 patients, was the largest clinical trial for psilocybin therapy in history.
The population targeted by this psychedelic intervention are those with treatment-resistant depression (TRD). These individuals, of which there are thought to be at least 100 million worldwide, have failed to respond to at least two existing antidepressant treatments.
In anticipation of the results we published a Special Issue, 2b, or not to be? Preparing for COMPASS Pathways’ Data Readout, in which we explored the significance of the forthcoming results and aimed to provide a primer on their interpretation.
Not a week later, while we were attending Microdose’s Wonderland conference in Miami, the company published topline results from the study on November 9th.
The results of the trial were generally positive, finding that a 25 mg dose of the company’s synthetic psilocybin (COMP360), alongside ‘psychological support’, produces a statistically significant and clinically relevant reduction in depression symptom severity.
Summarising the findings, COMPASS CEO George Goldsmith said, “a high dose of psilocybin works immediately, the day after, for a large number of people, and continues to work.”
This appears borne out in the topline data, which showed that at least twice the number of patients in the 25 mg group demonstrated response to the therapy and remission from their treatment-resistant depression at weeks 3 and 12 compared with the 1 mg group.
However, many expressed concern about the serious adverse events reported in the study, which involved 12 patients and were more common in the 25 mg COMP360 group than in the 10 mg or 1 mg arms.
Goldsmith suggested that some of the most concerning adverse events were among non-responders, who were perhaps despairing at the therapy’s inability to help them (especially in the context of such hype and promise). On this topic, readers may find Tehseen Noorani’s piece on The Pollan Effect of interest. It’s also worth remembering that TRD sufferers are a very vulnerable group of people, with suicidal ideation, for example, not uncommon.
In this trial, COMPASS Pathways attempted to standardise their psilocybin-assisted therapy protocol as far as possible across its trial sites and individuals: a significant undertaking given the heterogeneity of psychedelic-assisted therapies.
Overcoming the incongruities between the idiosyncratic psychedelic-assisted therapy modality and the objectivity and standardisation demanded by the clinical trial regime is certainly no small feat. In fact, it was a significant contributor to the downfall of psychedelic research in the first place (see Oram, 2014; Bonson, 2017; or, Hall, 2021 for a broader overview).
For much deeper analysis on these results, including contextualisation of the serious adverse events and efficacy data, read our Special Issue: Deconstructing COMPASS Pathways’ Phase 2b Results (>3,000 words).
Upon publication of the results, COMPASS’ stock price took a tumble and dragged much of the sector down with it. CMPS had been climbing since October, closing in on $50 for the first time since February.
How might we explain this: a rational reaction to the publication of topline results, or a buy the rumour, sell the news moment? Remember, many psychedelics stocks are characterised by an unusually high level of retail investor ownership compared to other small-cap biotech companies, which may explain some of the apparent irrationality.
Microdosing: Little Doses, Little Evidence?
Anecdotal evidence has suggested that even sub-perceptual doses of psychedelics can produce a number of therapeutic or wellbeing-enhancing benefits. From general wellness benefits, to improved creativity and attention, microdosing psychedelics has garnered a substantial amount of public attention.
These positive reports have led to high expectations and hopes that clinical evidence might one day legitimise the practice in the eyes of regulators and practitioners. To this end, many researchers have been evaluating the practice.
Here’s what their efforts taught us about microdosing in 2021…
Imperial College London Researchers Find Anecdotal Benefits of Microdosing “Can Be Explained by the Placebo Effect”
In March, a group of researchers at Imperial College London published the largest placebo-controlled trial on psychedelics to date, which used an innovative citizen science approach to explore microdosing. The researchers concluded that the “anecdotal benefits of microdosing can be explained by the placebo effect.”
The research design was certainly interesting: 191 participants were asked to incorporate placebo control into their microdosing routine. However, it’s important to note that this was not supervised: instead, the study was self-blinded with participants instructed to place placebo and active capsules into envelopes with QR codes, before drawing the envelopes at random.
While this study is, of course, not comparable in rigour to a randomised controlled trial (RCT), the authors claim that their novel self-blinding approach allows them to reach the following conclusion:
“…Our study validates the positive anecdotal reports about the psychological benefits of microdosing (significant improvements from baseline in a broad range of psychological measures); however, our results also suggest that these improvements are not due to the pharmacological action of microdosing, but are rather explained by the placebo effect (lack of significant between-groups differences).”
The study was covered broadly in the media: the Guardian; FT; Science; Wired; Forbes; and others. It also encouraged a great deal of conversation and debate within the psychedelics community, especially from those who claim to have found, or witnessed, benefits from the practice.
Study Finds Correlation Between Microdosers and Lower Levels of Anxiety and Depression
Later in the year, in November, a microdosing study published in Scientific Reports caused a stir among microdosing advocates and skeptics alike.
The publication, titled Adults who microdose psychedelics report health related motivations and lower levels of anxiety and depression compared to non-microdosers, was co-authored by a mix of researchers at the University of British Columbia and those with other affiliations, including mycologist and microdosing advocate Paul Stamets.
The observational study found that:
“those who [microdose] appear to be slightly less symptomatic of depression and anxiety than their peers who report similar mental health concerns but do not microdose…”
The study also sought to understand the motivations of microdosers, with health and wellness-related motives proving to be the most prominent.
Interestingly, and perhaps frustratingly, the paper was reported with headlines such as:
- New study finds ‘microdosing’ psychedelics can be effective in treating anxiety, depression (The Hill)
- UBCO study finds psychedelic microdosing improves mental health (EurekAlert).
But, this reporting implies that causality was identified through the study: i.e., that the practice of microdosing causes, or leads to, lower levels of anxiety and depression. However, this was neither what the observational study sought to, or did, demonstrate.

Perhaps the most important thing to note here is that identifying a correlation between microdosing and better mental health does not imply that microdosing has caused a better mental state.
A co-author of the study reiterated that the purpose of the study was not to establish causality, noting that it would not be possible via an observational approach.
There were other concerns surrounding this study, including the omission of any discussion around placebo or expectancy effects, which we would expect to see in this type of study.
Read our late November Bulletin for a more detailed exploration of the study. Also note that Paul Stamets, a co-author of the paper, announced that a second microdosing study is due to be published in 2022.
Preprint Study Suggests “Expectation Effects Underlie At Least Some of the Anecdotal Benefits” of Microdosing
A preprint of a study by Cavanna et al. published in December delivered another blow to microdosing. Researchers recruited 34 individuals who planned to microdose with psilocybin mushrooms and employed a double-blind placebo-controlled design to investigate the effects of microdosing on subjective experience, behaviour, creativity, perception, cognition and brain activity.
While the researchers found that reported acute effects were significantly more intense among microdosers than those consuming placebo (which, the researchers note, could be explained by unblinding), for other measurements the group observed trends toward cognitive impairment, reductions in certain brain activity, or null results.
The researchers conclude:
“Our findings support the possibility that expectation effects underlie at least some of the anecdotal benefits attributed to microdosing with psilocybin mushrooms.”
Study Finds Microdosing Psilocybin Did Not Affect Anxiety or Depression Compared with Placebo
Another double-blind placebo-controlled microdosing study emerged in December, which sought to investigate whether microdosing psilocybin across the space of three weeks modulated emotion processing, altered interoceptive awareness, and reduced symptoms of anxiety and depression.
The researchers ultimately found that “psilocybin microdosing did not affect emotion processing or symptoms of anxiety and depression compared with placebo.” However, the researchers were stymied by the fact that most participants had tried psychedelics previously, and many easily broke the blinding. As has been recommended for many psychedelics studies, research in a substance-naive population may be more fruitful.
Beyond the ‘Benefits’: Potential Risks Associated with Microdosing
Speaking to Psilocybin Alpha, Dr. Kelan Thomas (Associate Professor of Clinical Sciences at Touro University California College of Pharmacy and Board Certified Psychiatric Pharmacist) noted:
“While psychedelic macrodoses have consistently demonstrated benefits for mental health in clinical trials, the current evidence available from double-blind randomized placebo-controlled microdosing trials show minimal positive benefit beyond the placebo response, suggesting that anecdotal reports of microdosing efficacy may be due to positive expectation effects. Pharmacology and toxicology researchers have also expressed concern over the potential risk of long-term chronic LSD and psilocybin microdosing to cause valvular heart disease due to 5-HT2B receptor binding affinity.”
Thomas has prominently sounded alarm around the potential dangers of microdosing, including in a presentation titled “Safety First: Microdosing’s Possible Benefits and Potential Risks,” and in a pair of articles on Chacruna Chronicles.
It’s also worth noting that several FDA-approved medications that are agonists of the 5-HT2B receptor, such as fenfluramine/phentermine (‘fen-phen’), have been withdrawn due to the risk of valvular heart disease. However, there is a dearth of longitudinal research on microdosing, and the doses involved are (unsurprisingly) small, so no firm conclusions can be drawn.
It’s also worth noting that participants in microdosing studies thus far are generally healthy individuals. As such, some may argue that the likelihood of seeing an improvement in wellbeing is lower, due to a relatively high baseline.
So, we shouldn’t write-off the possibility that microdosing may be beneficial to those diagnosed with depression or anxiety, for example, just yet.
It’s also possible that we might see differences in the efficacy of microdosing across different molecules. Perhaps microdosing LSD will demonstrate a different safety and efficacy profile to, say, psilocybin. Companies like MindMed are certainly hoping this will be the case. The company is sponsoring trials of LSD microdosing, including via its recently announced study of low-dose LSD effects on sleep and cognitive measures.
Psychedelics Induce Neuroplasticity & Neurogenesis?
Recent foundational scientific research has brought investigators closer to understanding how psychedelics might elicit their purported therapeutic effects.
While the full pharmacological profiles of drugs like psilocybin, LSD, DMT are not yet fully understood, the past year saw many important breakthroughs that have helped push that understanding forward. Recent discoveries appear to add support to the theory that much of the therapeutic potential of psychedelics can be attributed to their ability to elicit structural and functional changes in the brain via mechanisms like neuroplasticity and neurogenesis.
Should psychedelics act as a catalyst for neuroplasticity and neurogenesis, these compounds may prove to be viable treatments for disorders beyond those falling within the realm of mental health, such as neurodegenerative disorders.
In 2021’s most popular study on psychedelics and neuroplasticity (according to Altmetrics; see more studies ranked by attention later in this section), researchers from Yale set out to understand what effects psilocybin had on structural plasticity in vivo, how quickly these changes might occur, how enduring the effects may be, and whether the drug’s hallucinatory effects are related to structural remodeling.
Commenting on the group’s approach, co-author Dr. Alex Kwan, Associate Professor of Psychiatry at Yale School of Medicine, told Psilocybin Alpha:
“Although there have been hints of psilocybin promoting neural plasticity, we were able to visualize and track the plasticity over many days in a live brain for the first time. The approach is powerful, and opens up new ways to screen compounds, by looking at how they directly affect neural circuits.”
Through their innovative efforts, the Yale researchers found that a single dose of psilocybin can quickly lead to structural changes in the medial frontal cortex of mice. Shao et al. demonstrated that the administration of psilocybin resulted in the increased growth and formation of dendritic spines. Evidence suggested that a number of these new spines lasted long enough to develop into functional synapses, some of which were found to persist 34 days after administration. Together, their results added to the growing body of research that points towards neuroplasticity as an impetus for many of the therapeutic benefits psychedelics are believed to produce.
The researchers also attempted to shed light on the relationship between hallucinatory effects and neuroplasticity. To achieve this, they administered ketanserin, a serotonin 5-HT2A receptor antagonist, before administering psilocybin. The researchers found that psilocybin continued to elicit some of its neuroplastic effects, despite a roughly 30% reduction in available 5-HT2A receptors (the primary receptor on which psilocybin acts to generate hallucinogenic effects) in the mice treated with ketanserin. While these results seem to suggest that neuroplasticity might not depend on psilocybin’s hallucinatory effects, Shao et al. maintained that due to the differences between humans and mice, further studies will be needed to evaluate this relationship in humans. Nonetheless, their discoveries add to an exciting conversation around psilocybin’s therapeutic mechanisms.
Studies being conducted by researchers and companies alike continue to contribute to our understanding of the relationship between psychedelics, neuroplasticity, and derived therapeutic benefits. Recent results from an in vitro preclinical trial by Algernon Pharmaceuticals (which, it should be noted, are neither published nor peer-reviewed) suggested that even a sub-perceptual (non-hallucinogenic) dose of N, N-Dimethyltryptamine (DMT) could result in a rapid growth of neural connections in the brain. The company hopes to one day leverage these effects to treat patients recovering from the neurodegenerative impacts of strokes.
Another 2021 study found that the in vivo administration of the non-hallucinogenic (according to animal studies) ibogaine analogue tabernanthalog led to dendritic spine formation as well. This work is being commercialised by Delix Therapeutics in partnership with the National Institute on Drug Abuse (NIDA).
So what might some of the implications of these findings be? As the researchers iterated, these neural modifying effects may help explain some of psilocybin’s purported antidepressant benefits. Like others in the field, including Delix Therapeutics’ David Olson, Kwan sees the promotion of neural plasticity as a key mechanism of action behind psychedelics’ apparent therapeutic effects.
“We are now seeing hundreds, if not thousands, of novel psychedelic-like compounds. For me, a key question is how to screen them and prioritize the most promising candidates for human studies? I believe neural plasticity is key, and a step towards identifying changes in the brain that are unique to psychedelics and that drive the beneficial actions.”
A number of popular 2021 publications have proposed that psychedelics could one day be used as treatments for Alzheimer’s and brain injuries (perhaps working via other mechanisms beyond neuroplasticity: see Szabó et al., 2021, for example). Should the emerging evidence continue to support the theory of psychedelic-induced neuroplasticity in humans, these compounds may be applied to a much broader range of psychiatric and neurodegenerative treatment indications.
Selected psychedelics and neuroplasticity/neurogenesis publications from 2021
- Psilocybin induces rapid and persistent growth of dendritic spines in frontal cortex in vivo
- DMT Increases Growth of Rat Neurons by 40 Percent, New Data Shows (Algernon)
- An analog of psychedelics restores functional neural circuits disrupted by unpredictable stress
- Psychedelics for Brain Injury: A Mini-Review
- A Single Dose of Psilocybin Increases Synaptic Density and Decreases 5-HT2A Receptor Density in the Pig Brain
- Psychedelics and Neuroplasticity: A Systematic Review Unraveling the Biological Underpinnings of Psychedelics
- Therapies to Restore Consciousness in Patients with Severe Brain Injuries: A Gap Analysis and Future Directions
- Critical Period Plasticity as a Framework for Psychedelic-Assisted Psychotherapy
- From Psychiatry to Neurology: Psychedelics as Prospective Therapeutics for Neurodegenerative Disorders
- Psychedelics as Novel Therapeutics in Alzheimer’s Disease: Rationale and Potential Mechanisms
- Neuroplasticity as a Convergent Mechanism of Ketamine and Classical Psychedelics
- Psychedelic-inspired Approaches for Treating Neurodegenerative Disorders
- Psilocybin Therapy Increases Cognitive and Neural Flexibility in Patients with Major Depressive Disorder
Further Clarity on Interactions Between SSRIs and Psilocybin
Given that many of the populations targeted by psychedelic-assisted therapies are attempting to find relief via other pharmacological agents, such as SSRI antidepressants, it’s important that we understand the interaction between these common drugs and psychedelics.
Beyond avoiding potential issues with safety or reduced efficacy, deepening our understanding of drug interactions of this kind may help increase the accessibility of psychedelic-assisted therapies. If it’s found that SSRIs and psychedelics do not generate adverse effects when co-administered, this could allow psychedelic-assisted therapies to be provided as an adjunct therapy as opposed to a monotherapy. This wouldn’t require participants in the therapy to taper off SSRIs, which can be a daunting and tumultuous process for those who have been chronically taking antidepressants.
It’s not surprising, then, that for-profit companies are keen to produce such data.
In December, COMPASS Pathways shared unpublished results of a single-arm open label study of 19 patients taking SSRI antidepressants alongside its COMP360 psilocybin therapy. The company found that a single 25 mg dose of COMP360 resulted in comparable treatment outcomes to patients in the company’s Phase 2b trial (mentioned above), in which patients were required to taper off their SSRIs. These findings will likely inform the company’s Phase 3 trial design, for which it is set to meet with the FDA in the new year with an eye to commencing the trial in Q3 2022.
MindMed, via its collaboration with the Liechti Lab, also published results from an SSRI and psilocybin interaction study. The research also appears to refute received wisdom that chronic administration of serotonergic antidepressants (such as SSRIs) dampen the subjective effects of psychedelics (e.g. Bonson and Murphy, 1996, in the case of LSD).
The MindMed sponsored study pretreated participants with the SSRI escitalopram for 14 days (7 days at a 10 mg dose, followed by 7 days at a 20 mg dose), or placebo pretreatment, and then administered 25 mg of psilocybin.
The study found that pretreatment with escitalopram had “no relevant effect on positive mood effects of psilocybin but significantly reduced bad drug effects, anxiety, adverse cardiovascular effects, and other adverse effects of psilocybin compared with placebo pretreatment.”
But, is a 14 day stepped pretreatment representative of real-world SSRI usage? The Psychedelic Pharmacists Association asked, “Is 14 days of pretreatment sufficient to draw conclusions about the need for antidepressant tapering and cessation before psilocybin administration?”
The authors of the study noted this limitation themselves, explaining that, “escitalopram pretreatment lasted only 14 days, which may have been too short to produce more chronic neuroadaptations and changes in receptor expression that can alter the response to psilocybin.”
Appreciating Methodological Issues
Throughout the course of recapping some of these central studies of 2021, it should have become apparent that psychedelic research is fraught with methodological challenges. Some of these are inherent to the broader field of psychotherapy and pharmacology, but others are unique to psychedelics. These include outsized expectancy effects (due, in part, to increasing ‘hype’) and, perhaps most obviously, the difficulty (impossibility?) of designing a genuinely placebo-controlled trial, given that it’s pretty obvious to most people if they’re having a ‘trip’.
Those interested in reading more about these methodological issues will likely enjoy a recent preprint publication by Aday et al., which provides recommendations for improving the methodological rigor of psychedelic clinical trials.
Newly-Approved and Announced Studies
As interest continued to grow throughout 2021, investigations into the effects, mechanisms, and therapeutic potential of psychedelics proliferated. Companies at various stages of development announced the approval and initiation of dozens of new trials studying psychedelics for an increasingly wide range of treatment indications, and investigators at academic institutions and research centres across the globe helped push the number of ongoing investigations to new heights. Researchers are working diligently to bolster and challenge our current understanding of psychedelic science.
Below you will find a sample of sponsor or investigator led trials that were announced, approved, or initiated in 2021.
A Sample of Company-Sponsored Trials That Were Approved or Began in 2021
TRYP Initiates a Phase 2 Trial of Psilocybin for Binge Eating Disorder [December 23, 2021]
- In October of 2021, TRYP Therapeutics announced that the FDA had placed a clinical hold on its Phase 2 trial of psilocybin in the treatment of binge eating disorder. The company subsequently amended its IND application with the FDA. On December 23, TRYP announced that it had received notice from the FDA that the clinical hold had been lifted. The study will evaluate both the safety and feasibility of treating patients diagnosed with binge eating disorder using psilocybin. [NCT05035927]
MindMed Initiates a Phase 2a Proof-of-Concept Trial of LSD for ADHD [December 17, 2021]
- On December 17, 2021, MindMedicine announced that it had begun enrollment for a Phase 2a trial evaluating repeated low doses of LSD as a treatment for ADHD. The company will collaborate with both Maastricht University and the University Hospital at Basel. Through the study, researchers will gather information on changes in ADHD symptoms, dosing regime, therapeutic mechanisms, and other effects of low doses of LSD.
TRYP Receives FDA Approval for a Phase 2a Trial of Psilocybin for Fibromyalgia [December 6, 2021]
- In December of 2021, TRYP Therapeutics announced that it had received FDA approval to study its TRP-8802 psilocybin candidates as a potential treatment for fibromyalgia. The trial, which is slated to begin in 2022, will evaluate whether or not psilocybin-assisted psychotherapy can help treat chronic pain symptoms in patients diagnosed with fibromyalgia. [NCT05128162]
Cybin and the University of Washington Initiate a Phase 2 Trial of Psilocybin for COVID-Related Distress [December 1, 2021]
- On November 30, 2021, Cybin announced that the FDA had authorized advancement of an investigator-initiated Phase 2 trial studying psilocybin-assisted psychotherapy in the treatment of frontline clinicians suffering from COVID-related distress. The co-funded trial will occur at the University of Washington in Seattle. Investigators will use Cybin’s EMBARK model of psychedelic-assisted psychotherapy to inform its use in future company-sponsored trials. [NCT05163496]
COMPASS Pathways Announces a Phase 2 Trial of Psilocybin for PTSD [November 3, 2021]
- On November 3, 2021, COMPASS Pathways announced a new Phase 2 trial investigating COMP360 psilocybin therapy as a potential treatment for PTSD. The company intends to enroll 20 patients to assess the safety, tolerability, and efficacy of its psilocybin therapy protocol. This announcement followed the completion of the company’s flagship Phase 2b trial of psilocybin therapy for treatment-resistant depression (TRD).
DemeRx (atai) Initiated Part 1 of a Phase 1/2a Trial of DMX-1002 for OUD [September 21, 2021]
- On September 21, 2021, atai life Sciences announced that its platform company DemeRx had dosed its first patients in the first part of a Phase 1/2a trial investigating DMX-1002 (ibogaine) as a treatment for opioid withdrawal syndrome. The company announced that this Phase 1 part of the trial will evaluate the safety, tolerability, pharmacokinetics, and efficacy of its DMX-1002 oral ibogaine formulation. [NCT05029401]
Small Pharma Initiates Part 2 of a Phase 1/2 Trial of DMT for MDD [September 21, 2021]
- On September 21, 2021, Small Pharma announced that it had completed the first phase of its Phase 1/2a clinical trial studying SPL026 (DMT) as a treatment for major depressive disorder (MDD). The company subsequently initiated the second Phase 2a proof-of-concept segment of the trial to assess the efficacy, safety, and tolerability of its intravenous DMT drug candidate in conjunction with psychotherapy. [NCT04673383]
Beckley Psytech Initiates a Phase 1b of Psilocybin for SUNHA [September 14, 2021]
- On September 14, 2021, Beckley Psytech announced that it had begun dosing patients in its previously approved Phase 1b trial of low-dose psilocybin for Short-lasting Unilateral Neuralgiform Headache Attacks (SUNHA). The trial will investigate the safety, tolerability, and efficacy of different ascending doses of psilocybin in patients diagnosed with SUNHA. [NCT04905121]
Mydecine Announces a Phase 2/3 Trial of MYCO-001 for Smoking Cessation [September 7, 2021]
- On September 7, 2021, Mydecine announced its intent to initiate a new Phase 2/3 studying its MYCO-001 psilocybin drug candidates for nicotine dependence. The company will collaborate on the trial with Johns Hopkins researcher Dr. Matthew Johnson. Through the study, researchers will assess the efficacy of using MYCO-001 in conjunction with a smoking cessation treatment program to treat nicotine dependent patients.
Braxia Initiates a Phase 2 Trial of Psilocybin for TRD [August 27, 2021]
- In August of 2021, Braxia Scientific announced the initiation of a Phase 2 trial studying psilocybin for treatment-resistant depression (TRD). Braxia will collaborate on the study with the Usona Institute to assess the safety, feasibility, and efficacy of varying doses of psilocybin in patients diagnosed with treatment-resistant depression. [NCT05029466]
A Sample of Investigator-Initiated Trials That Were Approved or Began in 2021
Trial of LSD Microdosing for Creativity and Brain Activity at the University of Auckland
- This randomised controlled trial will study the effects of LSD microdosing on healthy adult males. The study will assess the effects that LSD microdosing has on a number of personality and creativity measures. Researchers will also measure brain activity before and after the administration of LSD. [ACTRN12621000436875]
Phase II Trial of Psilocybin for Co-occurring MDD and AUD at Johns Hopkins University
- This double blind, placebo controlled study will evaluate the therapeutic effects of psilocybin in patients suffering from both major depressive disorder (MDD) and alcohol use disorder (AUD). Primary measures include reductions in depressive symptoms and amount of alcohol consumption. [NCT04620759]
Phase I Trial of Psilocybin for Headache Disorders at Yale University
- This Phase I trial will study repeated dosing of psilocybin as a potential treatment for migraines. The trial will recruit 24 participants who will receive varying dose combinations of psilocybin and placebo. Primary measures in the trial include migraine frequency, intensity, and duration. [NCT04218539]
Phase I/II Trial of Psilocybin for Severe OCD at Beersheva Mental Health Center
- This open label, Phase I study will evaluate the safety, efficacy, and feasibility of psilocybin-assisted psychotherapy for the treatment of severe obsessive compulsive-disorder (OCD). 15 patients suffering from OCD who had failed at least one prior treatment will be enrolled. [NCT04882839]
Phase II Trial of Psilocybin for AUD at Copenhagen University Hospital Rigshospitalet
- This trial, sponsored by Anders Fink-Jensen, MD, DMSci, will evaluate safety of using psilocybin in patients suffering from alcohol use disorder (AUD). Secondary measures will focus on feasibility, pharmacokinetics, several subjective effects, and changes in alcohol cravings, self-efficacy, and mindfulness. [NCT04718792]
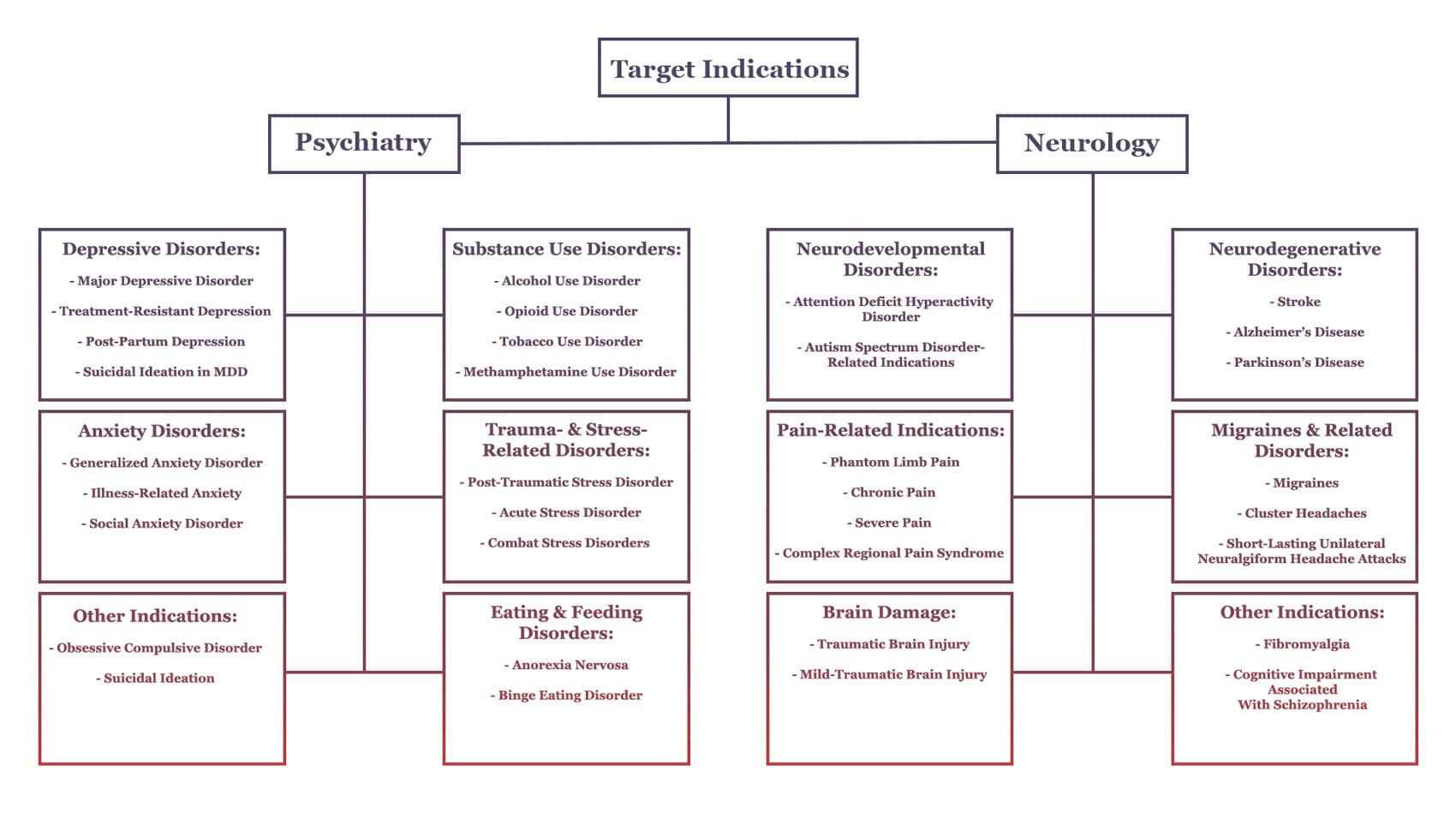
An Increasingly Warm Regulatory Environment for Psychedelic Research
Following a near half-century nadir in psychedelic research, the field is booming. This is helped, in part, by an increasingly warm regulatory environment: from federal funding and increases in production quotas for research purposes, right through to further fast-track designations and research partnerships with federal agencies.
Below, we highlight some of the key signals of bureaucratic amenability toward psychedelic research.
DEA Increases Psychedelics Production Quotas
The U.S. Drug Enforcement Administration (DEA) sets annual quotas for the legal production of controlled substances for research purposes, including psychedelics like psilocybin, DMT and MDMA.
The federal agency has increased these quotas—both intra-year quotas for 2021, and for the current year, 2022—multiple times in the latter half of 2021, with drugs like psilocybin seeing enormous boosts to production levels.
In an early September 2021 bulletin we reported on the DEA’s proposal to significantly increase the production quota for research psilocybin for 2021. Then, in mid-October, we covered the Administration’s proposal to greatly increase psychedelic production quotas for the present year, 2022.
Take psilocybin, for example. In its earlier proposed increases to the 2021 quotas, the DEA expressed plans to increase psilocybin production to 1,500 grams (1.5 kg), which represented a fifty-fold increase from the 30 g federal quota. On Monday 15th November, a notice published in the Federal Register revealed that the agency has boosted this in-year quota increase yet again, with the final adjusted aggregate production quotas allowing for 6,000 grams (6 kg) of psilocybin and 3,500 grams (3 kg) of psilocin to be produced in 2021. In December, that number was increased even further to 8,000 grams (8 kg).
For those looking for a deeper diver, Kyle Jaeger’s reporting in Marijuana Moment provides a fantastic overview of the changes.
Biden-Harris Administration Recommends Reducing Barriers to Research For Schedule I Substances
In a press release published in September the White House explained that Regina LaBelle, Acting Director of National Drug Control Policy, presented to Congress the Biden-Harris Administration’s recommendations that primarily related to reducing the supply and availability of illicitly manufactured fentanyl-related substances (FRS).
However, if one looks beyond recommendations related to FRS, the piece explains the Administration’s ambition to “establish a simplified process that would align research registration for all Schedule I substances […] more closely with the research registration process for Schedule II substances.”
The press release goes on to explain that, “the Biden-Harris Administration strongly supports expanding the research of Schedule I substances to help advance evidence-based public policy.”
Given that psychedelics are Schedule I substances in the United States, these recommendations to Congress could make research into psychedelics such as psilocybin and DMT more straightforward than at present, especially when combined with DEA quota increases.
Federal Grant Awarded to Johns Hopkins Researchers to Study Psilocybin for Smoking Cessation
In September 2021, prominent psychedelic researcher Matthew W. Johnson announced (via Twitter) that he has received a grant from the National Institute on Drug Abuse (NIDA) to study psilocybin for the treatment of tobacco addiction.
According to Johnson, this is the first grant from the U.S. Government dedicated to investigating the therapeutic effects of a classic psychedelic in over a half-century, marking a “new era in legitimacy” for psychedelic science.
We spoke to Sandeep Nayak, Post-Doctoral Research Fellow at Johns Hopkins’ Center for Psychedelic & Consciousness Research, about this development.
“It’s hard to overstate the importance of federal funding for psychedelic research,” Nayak noted. Commenting on the changing research funding landscape, he went on to add that, “this is a huge step towards normalizing this line of work which has been mostly funded by philanthropy and now nascent drug companies.”
Beyond the grant itself, which amounts to nearly $4m, this government funding “legitimizes the research and makes it much more feasible for researchers to build careers working on psychedelics,” Nayak added, noting that, “it’s also a clear sign of how much public opinion has shifted.”
The Johns Hopkins researcher, who is working alongside Matthew W. Johnson on this research, believes this isn’t the last we will see of government funding for psychedelic research: “This funding is significant on its own, but I anticipate it will be the first of many.” Finally, he noted that this trend is not limited to the U.S., “there are now government funded psychedelic trials in Germany and Canada, and likely we’ll see this expand greatly in coming years.” As we mentioned in our first Year in Review section, the Australian government carved out $15m for psychedelic research, which is supporting seven trials.
National Institute on Drug Abuse Partners with Psychedelics Companies
Two psychedelic drug developers, Delix Therapeutics and CaaMTech, are among a growing crop of companies to score cooperative R&D agreements with U.S. federal agencies.
In the case of CaaMTech, the company has entered into a Cooperative Research and Development Agreement (CRADA) with the Designer Drug Research Unit (DDRU) at the National Institute on Drug Abuse (NIDA) Intramural Research Program (IRP). That’s a lot of jargon, but the company told Psilocybin Alpha that the purpose of this agreement is to screen CaaMTech’s library of tryptamine compounds using a number of in vitro and in vivo assays.
According to a CaaMTech spokesperson, “previous work in this area has been frustrated by a lack of access to pure, well-characterized compounds.” As such, the aim of the CaaMTech-DDRU collaboration is to “supply much-needed data about the fundamental biological activity of tryptamine compounds, making it possible to develop safer and more effective next-generation psychedelic drugs.”
In December, Boston-based Delix Therapeutics announced its own partnership with NIDA. Research will be conducted under NIDA’s Addiction Treatment Discovery Program (ATDP), which works to screen promising therapies that may be more effective than the standard of care for substance use disorders.
Delix’s non-hallucinogenic (in animal models) drug candidates, such as DLX-7, appear to reduce alcohol- and heroin-seeking behaviour in preclinical studies. The company will hope to have these findings validated through further study, with initial data from NIDA’s research on the candidate expected in early 2022.
DMT Therapy Receives Fast-Track Designation in UK
In October, London-based Small Pharma announced that it had been granted a fast-track designation from a UK regulator for its DMT lead candidate. The UK Medicines and Healthcare products Regulatory Agency (MHRA)–the country’s equivalent of the FDA–granted Small Pharma’s SPL026 candidate an Innovation Passport Designation via a relatively new Innovative Licensing and Access Pathway (ILAP) program.
In a similar manner to the FDA’s fast-track designation, the ILAP aims to accelerate time to market and facilitate patient access to emerging novel treatments. The ILAP connects other key stakeholders in the drug development and roll-out process, including the country’s National Health Service (NHS) and the National Institute for Health and Care Excellence (NICE). The program was touted as a way to encourage the development of innovative medicines in the UK post-Brexit.
Psilocybin Alpha understands that Small Pharma’s DMT candidate is the first psychedelic to receive this designation since they began being issued in Spring 2021.
Speaking to Psilocybin Alpha, Small Pharma’s Chief Medical and Scientific Officer Dr. Carol Routledge explained that the designation allowed for the company to receive “specialist advice throughout the drug development process,” and that the Innovation Passport “has the potential to enable a speedier, more efficient development process for SPL026,” the company’s lead candidate.
SLP026, the company’s DMT-assisted therapy, entered clinical trials for major depressive disorder (MDD) at the start of this year, with topline results expected H1 2022.
Investors seemed optimistic, too, with the company’s share price jumping c.60% on the news after months of steady decline. This was short-lived, however, with the stock price slowly bleeding toward pre-news levels.
New Psychedelics Research Centres
2021 saw the number of prestigious institutions with psychedelic research centres continue to swell. Below are just a few of the new centres launched this past year…
Nikean Foundation Provides $5m to Launch Centre for Psychedelic Psychotherapy in Toronto
In September, we helped break the news of the launch of the Psychedelic Psychotherapy Research Centre at Toronto’s University Health Network.
A $5m donation from the Nikean Foundation, founded in 2019 by Canadian tech entrepreneur Sanjay Singhal, provides founding capital for the new centre.
Linda Medeiros, Director of Operations at Nikean Foundation, told Psilocybin Alpha that the creation of this centre “was a giant leap forward in Canadian history.” Medeiros went on to say, “the partnership with the University Health Network validated the need for systemic change in mental healthcare and the urgent need for new tools to meet unmet therapeutic needs.”
Importantly, Nikean supports those organisations conducting research in accordance with an Open Science approach rooted in principles of cooperation and accessibility. This is in contrast to the operating principles of many for-profit actors, especially in drug discovery and development, who tend to focus on confidentiality and defensibility (through various forms of IP, for example).
Medeiros was keen to highlight this point, telling us that, “as we emerge into this psychedelic renaissance, it is crucial that we promote collaboration and Open Science methods to move this field forward together for the greater good of humanity.”
Harvard Law School’s Petrie-Flom Centre Launches Research Initiative on Psychedelics and the Law
In June, the Petrie-Flom Center for Health Law Policy, Biotechnology, and Bioethics at Harvard Law School announced a new research initiative that will promote safety, innovation, equity and access in psychedelics research, commerce, and therapeutics.
The Project on Psychedelics Law and Regulation, or POPLAR, is the first academic initiative offering a clear focus on psychedelics law, regulation and ethics.
In our coverage of the launch we spoke to Mason Marks, Senior Fellow at the Center and Project Lead of POPLAR.
Beyond legislative measures, the debate around intellectual property in the psychedelics space is set to be a “major focus” of POPLAR, which will “analyze the role of biopiracy in psychedelics commercialization and the ethics, validity, and social utility of patents on psychedelics related inventions,” Marks explained.
University of Wisconsin Madison Launches Psychoactive Research Centre and Master’s Program
This year, the University of Wisconsin Madison approved a new Transdisciplinary Center for Research in Psychoactive Substances, and launched the first psychedelic master’s program in psychoactive pharmaceutical investigation, which is led by Dr. Cody Wenthur.
We spoke to the Director of the Center, Dr. Paul Hutson, who explained that the Center will expand its current portfolio of translational and Phase I, II, and III clinical research into psychedelics, as well as “embracing members from across the UW Madison campus” from a variety of disciplines including, “historians, anthropologists, and ethnobotanists, as well as the expected biomedical scientists and clinicians.”
According to Hutson, the Master’s program was well subscribed in the Fall of 2021, “and anticipates expansion in the coming years that will include internships in many of the new global psychedelic pharmaceutical companies.”
Top Publications by Attention
Hungry for more? We pulled the top 16 articles of the year, according to Altmetric. We covered some of these above, but not all…
- Trial of Psilocybin versus Escitalopram for Depression (Altmetric Score: 2,874) [April 15, 2021]
- A “trip” to the ICU: intravenous injection of psilocybin (Altmetric Score: 2554) [January 1, 2021]
- MDMA-assisted therapy for severe PTSD: a randomized, double-blind, placebo-controlled phase 3 study (Altmetric Score: 2216) [May 10, 2021]
- How ecstasy and psilocybin are shaking up psychiatry (Altmetric Score: 1721) [January 27, 2021]
- Psilocybin induces rapid and persistent growth of dendritic spines in frontal cortex in vivo (Altmetric Score: 964) [August 18, 2021]
- Self-blinding citizen science to explore psychedelic microdosing (Altmetric Score: 729) [March 2, 2021]
- Racial Justice Requires Ending the War on Drugs (Altmetric Score: 666) [January 7, 2021]
- Psychedelic-inspired drug discovery using an engineered biosensor (Altmetric Score: 551) [May 13, 2021]
- Ketamine for the treatment of mental health and substance use disorders: comprehensive systematic review (Altmetric Score: 400) [December 23, 2021]
- Adults who microdose psychedelics report health related motivations and lower levels of anxiety and depression compared to non-microdosers (Altmetric Score: 389) [November 18, 2021]
- Lysergic acid diethylamide (LSD) promotes social behavior through mTORC1 in the excitatory neurotransmission (Altmetric Score: 387) [February 2, 2021]
- Acute Effects of Psilocybin After Escitalopram or Placebo Pretreatments in a Randomized, Double-Blind, Placebo-Controlled, Crossover Study in Health Subjects (Altmetric Score: 384) [November 7, 2021]
- Psychedelic Therapy: A Roadmap for Wider Acceptance and Utilizations (Altmetric Score: 348) [October 4, 2021]
- First Study of Safety and Tolerability of 3,4-methylenedioxymethamphetamine-assisted Psychotherapy in Patients with Alcohol Use Disorder (Altmetric Score: 342) [February 18, 2021]
- Psychedelics and Other Psychoplastogens for Treating Mental Illness (Altmetric Score: 340) [October 4, 2021]
- Psychedelics Alter Metaphysical Beliefs (Altmetric Score: 315) [November 23, 2021]
Read the Rest Of Our Year in Review
Join Our Newsletter
Have our multi-part Year in Review, plus our 2022 predictions, delivered to your inbox.
Join thousands of psychedelics insiders
Get the weekly psychedelic medicine briefing.
A free weekly digest covering trials, regulation, policy, and access.
By signing up, you agree to our privacy policy. You can unsubscribe at any time.