
The Top 5 Psychedelic Clinical Trials of 2022
The Top 5 Psychedelic Clinical Trials of 2022

Michael Haichin, PharmD
Psychedelic Alpha Medical Advisor

Part of our Year in Review series
Psychedelic Alpha’s Medical Advisor, Michael Haichin, looks back at five of the most impressive psychedelic clinical trials of 2022. A word of advice: Haichin is a footnote fanatic, so be sure to read them as you go.
Much of the contemporary psychedelic re-emergence is driven by the desire to determine whether psychedelics have medical benefits that outweigh their risks, and if so, how best to use them. Despite critiques1 from diverse stakeholders2, on trying to fit a psychedelic peg in a square hole among others, randomized controlled trials (RCTs) currently remain atop the (hopefully increasingly less rigid) hierarchy of ways to learn more about these drugs. See our Psychedelic Research Methods Panel and an opinion piece from the ‘Reimagining Psychedelic Trials’ working group from the Year In Review for more on this subject.
An observation about this past year: the quality (and volume) of trials improved3, making the selection of a top 5 difficult compared to previous years, and a good reason for cautious optimism as the field grows more sophisticated. While the debate about what falls under the “psychedelic” umbrella rages on4, studies with MDMA and ketamine were considered for this list but none made the cut5.
In no particular order, the top 5 psychedelic clinical trials from last year6…
1. Single-dose psilocybin-assisted therapy in major depressive disorder: A placebo-controlled, double-blind, randomised clinical trial
Robin von Rotz, Eva M Schindowski, Johannes Jungwirth, Anna Schuldt, Nathalie M. Rieser, Katharina Zahoranszky, Erich Seifritz, Albina Nowak, Peter Nowak, Lutz Jäncke, Katrin H. Preller, and Franz X. Vollenweider
EClinicalMedicine, December 2022.
What is the current knowledge on the topic?
Psilocybin garners significant attention as a potential rapid-acting antidepressant when delivered in some form of psilocybin-assisted therapy. The existing evidence is undoubtedly promising, bolstered by the recent publication of COMPASS’ Phase 2b trial7 of participants with treatment-resistant depression, but far from conclusive; previous studies were limited by their design, inconclusive results8, small sample sizes, or were conducted in a depressed population in the context of a life-threatening cancer diagnosis.
What question(s) did this study address?
What is the feasibility, safety, and antidepressant effect of a single administration of psilocybin (0.215 mg/kg)9 compared with placebo, both in conjunction with psychological support from one therapist, in participants with major depressive disorder?
What does this study add to our knowledge?
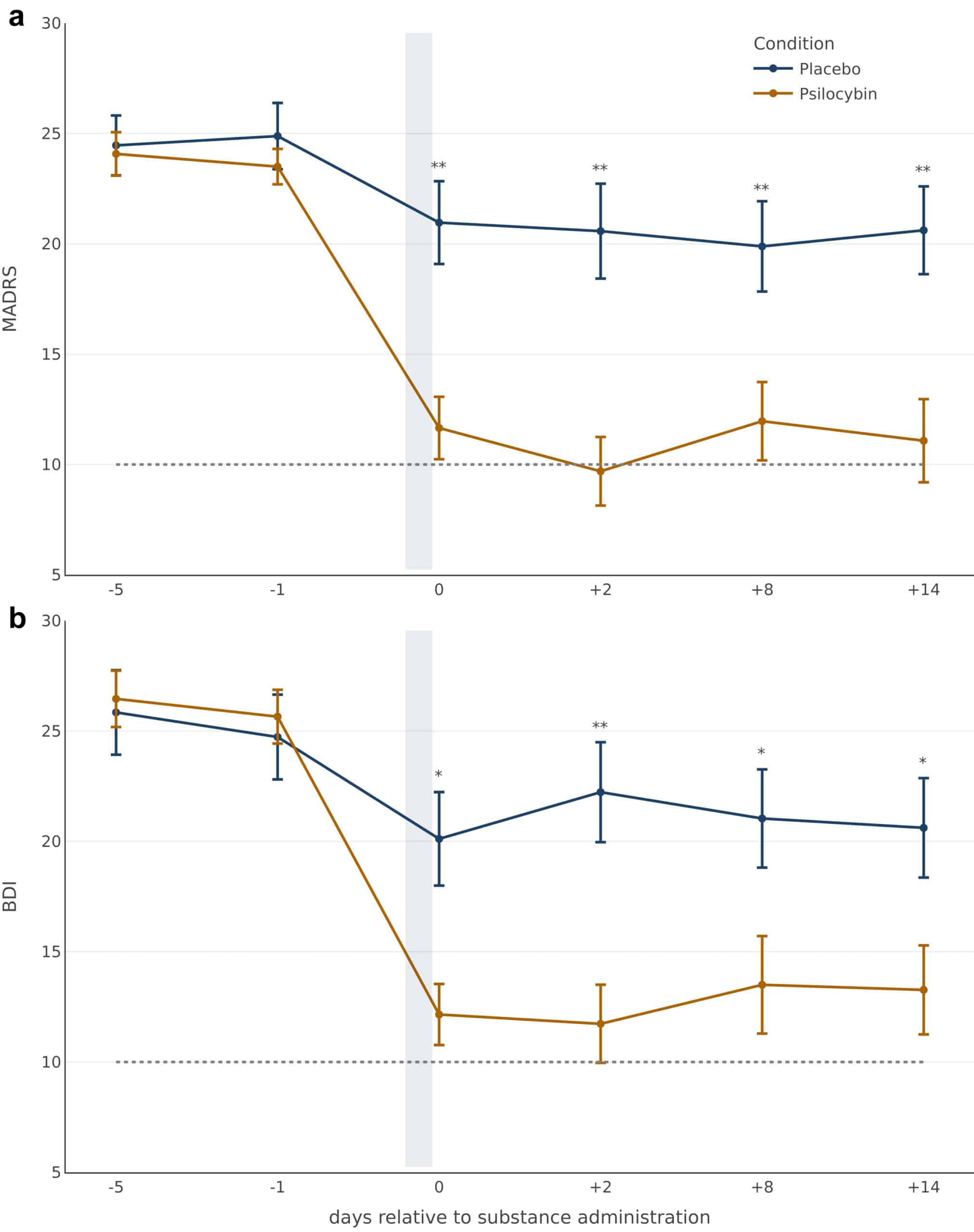
Psilocybin (n=26) produced short-term antidepressant effects (up to 2 weeks) with a 13-point decrease on the MADRS10 compared to baseline, which was significantly different from the placebo group (n=26). As the first modern placebo-controlled trial of psilocybin11, it provides some assurances that the symptomatic improvements in depression cannot be solely attributed to the non-pharmacologic effects of the supportive therapy. Contrary to a purported therapeutic mechanism, the overall intensity of psilocybin’s subjective effects was not strongly associated with its antidepressant effects12. The comparatively lower dose of psilocybin and reduced psychological support was well-tolerated with no serious adverse events.
A few thoughts:
- This is the latest in a line of studies attempting to optimise the delivery of psilocybin-assisted therapy in terms of safety and efficacy by manipulating several notable variables of the intervention: the amount of psychological support13, the therapist:patient ratio14, the number of psilocybin doses15, and the dose itself.
- Despite moderate baseline depression scores (MADRS ~24 in both psilocybin and placebo groups), 73% (19/26) of participants in each group were not taking any antidepressants at baseline16, thus making them markedly different from participants included in the Compass trial17. While cross-trial comparisons should be interpreted with caution, this, alongside the difference in psilocybin dose18, might explain the discrepancy in MADRS scores19.
- Concerns raised by researcher Eiko Freid about outcome switching (that the reported primary outcome was different than the one pre-registered on clinicaltrials.gov) were ultimately unfounded.
- As some have noted, despite the Swiss efficiency in comparison to other psilocybin therapy for depression trials, an intervention as cumbersome as this with only two weeks’ worth of effects20 is not likely to be a reimbursable one21.
2. Lysergic Acid Diethylamide-Assisted Therapy in Patients With Anxiety With and Without a Life-Threatening Illness: A Randomized, Double-Blind, Placebo-Controlled Phase II Study
Friederike Holze, Peter Gasser, Felix Müller, Patrick Dolder, Matthias Liechti
Biological Psychiatry, September 2022
What is the current knowledge on the topic?
During the first wave of psychedelic research in the 1950s to 1970s, LSD-assisted psychotherapy demonstrated promising antidepressant and anxiolytic effects in terminal cancer patients22. A small pilot study23 published in 2014 reported anxiolytic potential in a similar population with life-threatening diseases. However, modern studies have generally focused more on psilocybin24 than LSD25 and none have investigated the impact on anxiety symptoms in the absence of a life-threatening disease.
What question(s) did this study address?
What are the effects of two doses of LSD (200 mcg) or placebo spaced apart by six weeks, interspersed with equal amounts psychotherapy26 from a single therapist in participants with an anxiety disorder or with significant anxiety associated with a life-threatening illness, as part of a two 24-week period crossover design27?
What does this study add to our knowledge?
LSD (n=20) used in combination with psychotherapy produced anxiolytic effects throughout the first treatment period28 compared to placebo (n=22), with the maximum benefit two weeks after the second dosing session and sustained up to 16 weeks after (Week 24)29. At the end of that treatment period, 42% (8/19) of participants in the LSD group showed a clinical response30 compared to only 10% (2/20) in the placebo group. The LSD was generally well-tolerated with nausea (10%), anxiety (7%), and headache (2%) as the most frequent side effects, though one serious adverse event (SAE) of transient anxiety and delusions required the use of rescue medications lorazepam and olanzapine.
A few thoughts31:
- Not surprising: While the integrity of participant and researcher blinding did not appear to be formally assessed, the publication stated only a single participant in the LSD-first group mistook LSD as a placebo, with the revelation of their error following their placebo doses in the second treatment period.
- Somewhat surprising: As observed with other trials of psychedelics, measures of the acute subjective psychedelic effects32 were correlated with the therapeutic outcome in the first treatment period. Interestingly, maximum heart rate during LSD sessions also correlated with the therapeutic effects, though this correlation was weaker.
- Most surprising: Crossover studies introduce the possibility of order effects, whereby the sequence in which treatments are given may impact the outcome. In this trial, the placebo-first group had a weaker response to LSD, which was unexpected given that the acute psychedelic effects were similar in both groups. In an accompanying editorial33 researcher Harriet de Wit noted that extended psychotherapy combined with placebo appeared to attenuate the anxiolytic effects of LSD for the placebo-first group. Additionally, not only did placebo + psychotherapy not reduce anxiety during the first treatment period, it appeared to heighten it34. In her words: “These findings show that we still have much to learn about the psychedelic treatment of psychiatric disorders, and about how the drugs’ direct effects interact with both expectancies and concomitant psychotherapy.”
- Lastly, the authors acknowledged that 200 mcg of LSD is likely too high a dose and presents a heightened risk of acute anxiety for some patients, particularly those who are inexperienced. This was reflected in several ways throughout the trial: the above mentioned SAE requiring use of rescue medications; three participants (including the one who experienced the SAE) reduced their dose to 100 mcg for their second session because the effects were too strong; and a participant drop-out due to being “overwhelmed by LSD”. Their suggestion that a first dose of 100-150 mcg may be more appropriate, with an optional dose escalation for subsequent doses, warrants further exploration35.
3. Percentage of Heavy Drinking Days Following Psilocybin-Assisted Psychotherapy vs Placebo in the Treatment of Adult Patients With Alcohol Use Disorder: A Randomized Clinical Trial
Michael Bogenschutz, Stephen Ross, Snehal Bhatt, Tara Baron, Alyssa Forcehimes, Eugene Laska, Sarah Mennenga, Kelley O’Donnell, Lindsey T Owens, Samantha Podrebarac, John Rotrosen, J Scott Tonigan, Lindsay Worth
JAMA Psychiatry, August 2022
What is the current knowledge on the topic?
During the first wave of psychedelic research in the 1950s to 1970s, LSD demonstrated considerable promise in the treatment of alcohol use disorder (AUD)36. A more recent meta-analysis of six randomized controlled trials from that period revealed LSD-treated participants were almost twice as likely as comparators to reduce their drinking37, although with several limitations and important differences among the underlying studies38. In 2015, Bogenschutz and colleagues published the results of an open-label, proof-of-concept study that showed two doses of psilocybin were safe and led to significant decreases in drinking during the 32-week follow-up period.
What question(s) did this study address?
What is the safety and potential clinical efficacy of two doses of psilocybin or active placebo (diphenhydramine39) spaced one month apart, combined with psychotherapy, in reducing the percentage of heavy drinking days in treatment-seeking individuals with AUD?
What does this study add to our knowledge?
Psilocybin-assisted psychotherapy (n=48) reduced the proportion of heavy drinking days from 56.48% at baseline to 9.71% 32 weeks after the first dosing session (Week 4), a significant decrease compared to the diphenhydramine (n=45) group (48.57% baseline; 23.57% at follow-up). While there were no serious adverse events in the psilocybin group, anxiety was reported more frequently40 after the first administration of medication through Week 36. The integrity of the double-blind was assessed41, and perhaps unsurprisingly, participants and therapists correctly guessed the treatment assignment for both sessions over 90% of the time, with high degrees of certainty.

A few thoughts42:
- As the optimal dose of psilocybin for the treatment of AUD is unknown, the trial employed a dose escalation protocol that attempted to maximize the likelihood of strong subjective effects from psilocybin43 while minimizing the chances of adverse events. If the first session in which participants received 25 mg/70 kg of psilocybin was well tolerated, they were offered an increase firstly dependent on their willingness, with subsequent stratification to 30 mg/70 kg or 40 mg/70 kg44 based on intensity of the first experience and a measure of cardiovascular safety (QTc). In the end, the majority of participants in the psilocybin group opted to increase the dose, with only one staying at the initial dose and five others opting out of a second medication session altogether. Similarly, the diphenhydramine group was offered a dose titration (from 50 mg to 100 mg), though this was based on safety and participant preference.
- The results pertaining to the relationship between subjective effects and treatment outcomes were not included in this publication, but were briefly discussed during the NIH Psychedelic Workshop that took place in January 2022.
- Looking at the above figure, one can see a large reduction in drinking that occurred before any drug administration in Week 4. This can be attributed to the participants’ treatment-seeking nature45, four psychotherapy sessions46 that occurred prior to dosing, and the protocol requirement to attempt to remain abstinent during the week preceding the drug sessions.
- The trial also stands out for its attempt to develop a psychotherapeutic model inspired by an evidence-based behavioural intervention relevant to the disorder being treated, while integrating the psychedelic drug component. The first component was focused on facilitating motivation to change alcohol-related behaviours; known as Motivational Enhancement and Taking Action (META) therapy, it was based in part on motivational enhancement therapy (MET) and cognitive behavioural therapy. The other component was geared towards preparation, support, and integration (PSI) of the drug sessions. Participants were assigned to two therapists to receive PSI and META before and after the drug administration sessions, with each therapist taking the lead on a particular component. The psychotherapy provided was resource intensive and would be difficult to translate into a real-world setting, however to reduce costs, one can imagine the META component being delivered without the second therapist present.
- The considerably high retention rate49, an optional open-label psilocybin session with additional psychotherapy was offered to all participants to incentivise continued participation. These participants were followed for an additional 18 weeks (Week 54), but this means no longer term follow-up data comparing the psilocybin group to diphenhydramine can be expected.
4. Exploratory study of the dose-related safety, tolerability, and efficacy of dimethyltryptamine (DMT) in healthy volunteers and major depressive disorder
Deepak D’Souza, Shariful Syed, L Taylor Flynn, Hamideh Safi-Aghdam, Nicholas Cozzi, Mohini Ranganathan
Neuropsychopharmacology, June 2022
What is the current knowledge on the topic?
Modern psychedelic research in the West involving humans commenced in the early 90s, with work done at the University of New Mexico studying the effects of dimethyltryptamine (DMT) in healthy volunteers. Its therapeutic potential, however, has not been examined as with other psychedelic substances. Given the resource-intensive nature of psychedelic-assisted therapies thus far and an interest in their potential scalability, pertinent questions include what is the minimally effective psychotherapy dose to accompany drug administration, and can a shorter-acting molecule such as DMT50 be therapeutically useful?
What questions did this study address?
What is the safety, tolerability, and preliminary efficacy of two doses of intravenous (IV) DMT51 spaced at least 48 hours apart with minimal psychological support in patients with treatment-resistant depression52 and healthy volunteers?
What does this study add to our knowledge?
Despite participants reporting the experiences as intense and challenging, the two doses of DMT (n=10) were generally well tolerated, with the 0.1 mg/kg dose given a tolerability score of 89.9053 and 0.3 mg/kg with a slightly lower, significantly different rating of 71.11. The most common adverse events were transient anxiety, headaches, and high blood pressure. There was one SAE of asymptomatic low heart rate and blood pressure that required intervening with supportive measures54. Preliminary antidepressant efficacy was demonstrated in the patients with depression (n=7) one day after the 0.3 mg/kg dose, but not one day after 0.1 mg/kg.

A few thoughts55:
- This study is arguably the boldest to date in terms of exploring the boundaries of a suitable but minimalist container for use of a classic psychedelic in a medical setting.
- Setting: Instead of a comfortable and inviting room with a couch surrounded by art, dosing took place in a sterile hospital room in a “medical-grade reclining chair” under fluorescent lighting, with no music throughout. These contextual factors are presumed to play some role in the potential therapeutic effects of psychedelics, in part by enhancing the ritual nature of the experience56.
- Psychological support: Preparatory sessions lasted only 45 minutes, consisting of basic education of DMT’s effects and “flight instructions” for how to navigate the experience, as well as a brief discussion of their mood-related symptoms and history relevant to depression. No psychotherapy was provided by the two psychiatrists present during dosing sessions, however it is reasonable to speculate that their mere presence provided comfort to participants. Participants were simply asked how they felt at regular intervals coinciding with planned physiologic measurements. Integration, described by the authors as a debriefing conducted by one psychiatrist, was almost as sparse as preparation, taking place over approximately two hours as the DMT effects dwindled before participant discharge.
- Given the intensity of the experience associated with DMT, including the possible presence of entity encounters and entering the “DMT world”57, it’s not a stretch to wonder whether the minimal psychological support presents some safety risks and the possibility of destabilization beyond the trial period. However it’s important to note that a prerequisite to participation for those with depression was engagement in treatment including psychotherapy. Perhaps the investigators were relying on this fact for additional integration if required?
- The overall test intervention has some resemblance to how ketamine is often used in the treatment of mood disorders, which without repeated doses typically has rapid antidepressant effects that wear off in 3-14 days. Might this be the same case with DMT and none of the other accoutrements commonly found in psychedelic-assisted therapies? Antidepressant outcomes were only measured out to one day post-dosing, so longer follow-up times will be needed in subsequent, larger studies.
5. Exploratory investigation of a patient-informed low-dose psilocybin pulse regimen in the suppression of cluster headache: Results from a randomized, double-blind, placebo-controlled trial
Emmanuelle Schindler, R Andrew Sewell, Christopher Gottschalk, Christina Luddy, L Taylor Flynn, Yutong Zhu, Hayley Lindsey, Brian Pittman, Nicholas Cozzi, Deepak D’Souza
Headache, November 2022
What is the current knowledge on the topic?
Cluster headache disorder is a rare headache disorder with few effective treatments, considered so painful they are known by the harrowing nickname “suicide headaches”. In the late 1990s, patients with cluster headaches began discussing their use of psychedelic compounds as a treatment option in online forums, later resulting in the formation of Clusterbusters58. Several survey studies59, and more recently an open-label trial of psilocybin60, have corroborated that psychedelics may have therapeutic potential for this condition.
What question(s) did this study address?
What is the safety and preliminary efficacy of a patient-informed61 psilocybin (0.143 mg/kg)62 or placebo pulse regimen, consisting of three doses separated by 5 (± 2) days each, for the treatment of episodic or chronic63 cluster headache?
What does this study add to our knowledge?
The psilocybin (n=8) pulse regimen decreased the number of headache attacks64 by 3.2 attacks/week compared to 0.03 for placebo (n=6) over a 3-week period, but that difference was not statistically significant65. The intensity of the acute psychedelic effects were not correlated with the change in weekly attacks66. The psilocybin was well-tolerated, with no reported SAEs and nausea during dosing sessions as the only significant side effect compared to placebo.

A few thoughts67:
- The duration and pain intensity of cluster attacks also did not exhibit statistically significant decreases in the psilocybin group compared to placebo. In fact, the duration appeared to increase in the psilocybin group by an average of 10 minutes, for both episodic and chronic patients. It is important to note that the attack duration was significantly higher at baseline68 in the psilocybin group, possibly indicating a different level of severity.
- Despite the non-significant findings, the measured efficacy of psilocybin was larger in chronic compared to episodic participants, and somewhat persisted over the extended 8-week period in chronic participants. This effect aligns with the results of the open-label trial from Madsen and colleagues at the University of Copenhagen. Ten chronic cluster headache patients were given a similar pulse regimen of three doses of 0.14 mg/kg psilocybin separated by seven days each and a statistically significant reduction in attack frequency was observed from baseline to follow-up. While chronic patients are generally considered more difficult-to-treat, the inclusion of episodic patients presented a number of additional challenges, including the potential for natural termination of the cluster attack period or regression to the mean69.
- To ensure adequate blinding of researchers and participants, the study reported using an “enhanced blinding procedure”, the details of which were not provided as it might compromise future attempts to reuse it70. Unfortunately, blinding was not formally assessed so the procedure’s effectiveness is unclear. Several data points indicate it was likely insufficient and functional unblinding may still have occurred: the psilocybin group scored significantly higher on ratings of the intensity of the acute psychedelic effects71 and also had numerically more adverse events72 compared to placebo73. Future trials should systematically evaluate the blinding integrity, especially since it is plausibly more achievable with the lower dose of psilocybin used.
- If psilocybin is to be approved for any medical condition, cluster headache disorder may be the least surprising option. Besides the high unmet need and the early evidence generated by Clusterbusters’ citizen science, psilocybin shares chemical and/or pharmacological similarities with several existing treatment options: triptans, as well as ergotamine, dihydroergotamine, and methysergide.
- Who else remembers when the use of psilocybin-containing mushrooms for treating cluster headaches was featured74 on the TV medical drama House M.D. over 15 years ago? Not sure it aged well…
Honourable mentions75:
- Abdallah et al. (2022) – Dose-related effects of ketamine for antidepressant-resistant symptoms of posttraumatic stress disorder in veterans and active duty military: a double-blind, randomized, placebo-controlled multi-center clinical trial76
- Price et al. (2022) – A Novel, Brief, Fully Automated Intervention to Extend the Antidepressant Effect of a Single Ketamine Infusion A Randomized Clinical Trial77
- Shnayder et al. (2022) – Psilocybin-assisted therapy mediates psycho-social-spiritual change in cancer patients as assessed by the NIH-HEALS
- Family et al. (2022) – Safety, tolerability, pharmacokinetics, and subjective effects of 50, 75, and 100 μg LSD in healthy participants within a novel intervention paradigm: A proof-of-concept study78
- Becker et al. (2022) – Ketanserin reverses the acute response to LSD in a randomized, double-blind, placebo-controlled, crossover study in healthy subjects79
- dos Santos et al. (2022) – Effects of ayahuasca on the endocannabinoid system of healthy volunteers and in volunteers with social anxiety disorder: Results from two pilot, proof‐of‐concept, randomized, placebo‐controlled trials
- Burmester et al. (2022) – Subacute effects of a single dose of psilocybin on biomarkers of inflammation in healthy humans: An open-label preliminary investigation
- Wießner et al. (2022) – LSD and creativity: Increased novelty and symbolic thinking, decreased utility and convergent thinking
- Sanz et al. (2022) – Natural language signatures of psilocybin microdosing
- de Wit et al. (2022) – Repeated low doses of LSD in healthy adults: A placebo-controlled, dose–response study
- Cavanna et al. (2022) – Microdosing with psilocybin mushrooms: a double-blind placebo-controlled study
The volume of trials will only continue to increase. This year we can expect the publication of MAPS’ second Phase 3 trial of MDMA-assisted therapy for PTSD, Usona’s Phase 2 study of psilocybin therapy for major depressive disorder, among many others. Before long, trying to keep up will be like trying to drink from a firehose. Here’s to hoping at least the quality of the water continues to improve.
(Editor’s Note: You can follow Michael Haichin on Twitter, where he often shares psychedelic research updates and thoughts.)

Part of our Year in Review series
This content is part of our 2022 Year in Review, which looks back at the past year through commentary and analysis, interviews and guest contributions.
Receive New Sections in Your Inbox
To receive future sections of the Review in your inbox, join our newsletter…
Browse Other Sections of the Review
To explore our Year in Review, visit this webpage.
Join thousands of psychedelics insiders
Get the weekly psychedelic medicine briefing.
A free weekly digest covering trials, regulation, policy, and access.
By signing up, you agree to our privacy policy. You can unsubscribe at any time.
- At the forefront are those about methodology, never put more succinctly than by Aday et al. in Great Expectations: “Although some challenges are shared with psychotherapy and pharmacology trials more broadly, psychedelic clinical trials have to contend with several unique sources of potential bias. The subjective effects of a high-dose psychedelic are often so pronounced that it is difficult to mask participants to their treatment condition; the significant hype from positive media coverage on the clinical potential of psychedelics influences participants’ expectations for treatment benefit; and participant unmasking and treatment expectations can interact in such a way that makes psychedelic therapy highly susceptible to large placebo and nocebo effects.”
- For other noteworthy critiques from this year, see: Ona et al.; Schenberg et al.; Celidwen et al.; Devenot et al.; Ortiz et al.; Cristea et al.; Butler et al.; Bedi et al.
- Other limitations still largely apply, such as limited generalizability of the results to wider populations due to strict trial inclusion/exclusion criteria and relatively small sample sizes.
- Spurred on by the publication of Proposed Consensus Statement on Defining Psychedelic Drugs from the new Psychedelic Medicine journal. I do not have particularly strong feelings on the matter, but will note that media coverage of MDMA and ketamine in recent years will make it hard to disentangle them from the word ‘psychedelic’. The debate will not stop any time soon, and I suspect will only become more convoluted as additional molecules with overlapping pharmacology are explored and the boundaries between psychedelic, entactogenic, and dissociative anesthetic effects are further blurred.
- It was not a particularly active year for MDMA with only a few post-hoc analyses published (see Revisiting MDMA-Assisted Therapy Data), while ketamine just missed the cut. See Honourable Mentions for more.
- Topline results via press release and preprints were excluded as they are subject to change through the peer review process; though a couple stood out as particularly notable: Mason et al. and Madsen et al.
- Goodwin et al., 2022.
- A Bayesian reanalysis by Nayak et al., 2022 of Carhart-Harris et al., 2021 provided some confidence that psilocybin with psychological support was likely no worse than the SSRI escitalopram combined with the same amount of psychological support.
- Far from a microdose, this corresponds to ~16 mg for a 70 kg patient, albeit a lower amount than the doses assessed in previous psilocybin therapy for depression trials (i.e., ~ 25 mg, up to 30 mg/70 kg).
- Montgomery-Asberg Depression Rating Scale (MADRS) is a commonly used, clinician-rated depression severity score. Beck’s Depression Inventory (BDI), a self-report questionnaire, was also used to assess response to treatment. The primary outcome of the study was the change in MADRS and BDI score from baseline (~5 days before psilocybin administration) to 14 days after dosing compared to placebo.
- Other trial designs included low dose psilocybin (e.g. 1 mg), escitalopram, niacin, or a wait-list control as comparator groups.
- As researcher Max Wolff noted, the methods used to obtain this result factored in the overall intensity of the subjective effects by using the sum score of the 5-Dimensional Altered States of Consciousness (5D-ASC) scale, rather than the “psychotherapeutically relevant acute effects” captured by certain 5D-ASC subscales.
- Each participant had seven visits with a therapist for psychological support: two preparatory and three integration sessions (1 hour each) surrounding the 6-hour drug administration period, totalling 11 hours of therapist time.
- Each participant was assigned a single therapist for psychological support throughout the trial (See “Reducing the Costs of PAT” for more on this).
- The trial employed a single administration of psilocybin instead of two, as seen in other modern studies such as Davis et al. 2020 and Carhart-Harris et al. 2021. The use of two doses was previously considered more advisable, allowing patients to become acclimated with the psychedelic effects, especially if the first resulted in a psychologically distressing experience.
- No information about previous or ongoing psychotherapy was provided, indicating this may have been a largely treatment-naive population.
- Goodwin et al., 2022 focused on a population with treatment-resistant depression, requiring previous treatment failure of two to four antidepressants.
- Can also count a lower-than-normal placebo response as a factor, likely driven by the nocebo effect of not receiving psilocybin.
- A 12-point MADRS score decline in the 25 mg group compared with a 5.4-point decline in the 1 mg group at 3 weeks (difference of -6.6).
- The Supplementary Materials of von Rotz et al., 2022 indicate longer-term follow-up data will be published at a later date. Also published in this past year was the long-term follow-up of Davis et al. 2020, demonstrating psilocybin therapy’s antidepressant efficacy may be durable for up to 1 year (Gukasyan et al., 2022).
- A single dose of ketamine, delivered without any adjunctive therapy, produces comparable antidepressant effects that may last 3-14 days.
- e.g., Grof et al., 1973.
- Gasser et al., 2014.
- Grob et al., 2011; Griffiths et al., 2016; Ross et al., 2016.
- A trial directly comparing the effects psilocybin and LSD (Holze et al., 2022) was published by the same research group last year, but was received with some skepticism after reporting similar subjective effects that differed only by LSD’s longer duration of action; phenomenological descriptions found in trip reports often note more distinguishing features.
- Five study visits of approximately 1 hour each, conducted at baseline, between dosing sessions, and 2, 8, and 16 weeks after the second dose, per treatment period.
- Meaning all participants received two doses of both LSD and placebo in a random order, divided by a “washout” period of 2 weeks.
- A crossover analysis can involve using each participant as their own respective control (i.e. within subjects, comparing the first treatment period vs. second). This was reflected in the predefined primary outcome, which was the change in State-Trait Anxiety Inventory-Global (STAI-G) score from baseline to 16 weeks after LSD compared to placebo within subjects. However, due to the LSD-first group’s therapeutic effects persisting into the second treatment period, known as a carryover effect, the primary outcome was changed to compare LSD and placebo in the first period (i.e. between subjects) 16 weeks after dosing. It is unclear to me why within subjects was chosen as the initial primary outcome, when previous crossover trials of psilocybin-assisted therapy (Griffiths et al., 2016; Ross et al., 2016) showed similar carryover effects that limit therapeutic interpretations post-crossover.
- This length of therapeutic effect is considerable, and those in the LSD-first group maintained similar benefits until the end of the second treatment period (up to 50 weeks, but without a control in the second period).
- Defined as ≥30% reduction in STAI-G scores
- For a few more, see Bulletin #102 from back in May regarding the topline results, which predictably painted a less nuanced picture than the final publication.
- Oceanic Boundlessness and the Mystical Experience Questionnaire (MEQ30), but not Anxious Ego Dissolution.
- Progress and Puzzles in Psychedelic Therapy.
- A result of the nocebo effect, due to not receiving the desired LSD? Possible, but also counterbalanced by the fact participants knew they would receive LSD eventually within the crossover design. One can speculate that those with life-threatening illnesses had a nocebo response regardless, given potential mortality concerns, however the increase in anxiety occurred for placebo-first group participants both with and without a life-threatening illness (see Figures S4 and S7).
- MindMed, a collaborator of University Hospital Basel’s Liechti Lab who ran this trial, are running their own multi-site, dose-finding Phase 2b study of LSD (LSD D-Tartrate a.k.a MM-120) for anxiety.
- For a detailed review of this period of research, see Mangini, 1998.
- Treatment follow-up ranged from 1-12 months.
- Krebs et al., 2012.
- More widely known as Benadryl. Chosen for a multitude of reasons, including but not limited to: lack of evidence as a treatment for AUD, low abuse potential at the doses used in the trial, and noticeable subjective effects such as drowsiness and confusion that might enhance the blinding effectiveness (especially in those who are psychedelic-naive, though exclusion criteria allowed for up to 25 lifetime uses of any hallucinogen).
- Two participants required the use of rescue medication (diazepam) due to anxiety during their second psilocybin session.
- A rare occurrence in psychedelic trials (and in medicine more generally). See Schenberg, 2021.
- For a few more, see Bulletin #117 from back in September.
- Both mystical-type experiences (as measured by the MEQ) and more general measures of intensity of psilocybin’s effects predicted drinking-related improvements in the aforementioned proof-of-concept AUD trial (Bogenschutz et al., 2015).
- This dose is significantly higher than what has been trialed in depression studies, in part because there is evidence (both historical and from the proof-of-concept study) that alcohol-dependent individuals may require higher doses to achieve an equivalent subjective effect. Similar logic is being applied to other psilocybin trials for the treatment of substance use disorders (e.g., opioid (NCT05242029), methamphetamine (NCT05322954)).
- See a more detailed explanation from researcher Matthew Baggott.
- Out of a total of 12; Four others occurred between the two dosing sessions, and four in the month following the second dose.
- Only three participants in the diphenhydramine group and only two in the psilocybin group failed to complete follow-up at Week 36.47 for an AUD trial is likely driven by one reason: out of concern for a larger drop-out rate in the diphenhydramine group48Related to the likelihood of unblinding as a result of psilocybin’s strong subjective effects
- The onset of DMT’s psychedelic effects when administered via IV is within two minutes and typically ends after 30 minutes.
- Delivered as part of an open-label, fixed-order dose escalation starting with 0.1 mg/kg, and if well tolerated and the participant agreed, was followed by a second session of 0.3 mg/kg. The psychedelic “threshold” for IV DMT was established in healthy volunteers at 0.2 mg/kg (Strassman et al., 1994).
- Patients with major depressive disorder were defined as treatment-resistant if they previously failed treatment with at least two antidepressants, and at least one during the current depressive episode.
- On a scale of 0 to 100, where 0 = intolerable and 100 = well tolerated.
- After the incident, the subject revealed they had a history of many (>30) episodes of low blood pressure. The authors determined the event was related to DMT’s effects combined with the participant’s undisclosed medical history.
- For a few more, see Bulletin #106 from back in June.
- Demonstrated in Olson et al., 2020 where psychedelic-like effects are induced without any drug administration.
- For example, see Davis et al., 2020 and Lawrence et al., 2022 for more about the phenomenology and content of the DMT experience.
- Before registering as a nonprofit organization in 2006, Robert “Bob” Wold started Clusterbusters as an online forum/patient support group in 2002 specifically to discuss the use of psychedelics for the treatment of cluster headaches.
- e.g., Sewell et al., 2006; Schindler et al., 2015.
- Aforementioned preprint from Madsen et al., published last year.
- “Patient-informed” refers to the fact that the pulse dosing regimen was derived from over two decades of patient experimentation and experience. However, the fixed design of dosing in the study differs from what is commonly practiced by patients with cluster headache, where adjustments to the dose or regimen during the pulse may occur. To learn more about the history of this citizen science, see “Unauthorized Research on Cluster Headache” and Bailey et al., 2022.
- Meant to approximate one of the more commonly used regimens by patients, consisting of 1-2 grams of dried Psilocybe cubensis mushroom (roughly equivalent to 6-12 mg pure psilocybin) taken three times, ~5 days apart. 0.143 mg/kg = 10 mg/70 kg.
- Cluster headache disorder can be episodic, where attacks occur daily for 2-8 weeks seasonally, or the more uncommon and difficult-to-treat chronic form, defined as less than three months without attacks/year.
- Measured using a headache diary to record the number of cluster attacks, the duration of the attack, and the pain intensity, which is the gold standard in headache research.
- The authors state that this negative finding is owing in part to the small number of participants. This is not unusual in exploratory studies of this type, and even happened with the first modern psilocybin study in patients with advanced-stage cancer (Grob et al., 2011).
- This is somewhat expected given that the patient-informed pulse regimen is often composed of low or sub-perceptual doses of psychedelics. Additionally, a non-hallucinogenic analog of LSD known as BOL-148 (2-bromo-LSD) demonstrated some therapeutic potential for cluster headaches in an open-label study (Karst et al., 2010) and is currently under further investigation. These both point to a potentially unrelated mechanism of action than what is suggested with the use of psychedelics in mood or substance use disorders thus far. A similar lack of association was found in a trial of psilocybin for migraine run by the same group of researchers (Schindler et al., 2020).
- For more on the legal and patent side of this work, see Matt Zorn’s On Drugs Psychedelic Patent Wars 2: Lost at the Patent Office (Part 1).
- The authors wrote that alternative methods of analysis including adjusting for baseline differences did not affect the results.
- Note that in the placebo group, episodic patients reported a decrease in attack frequency, contrasted with the observed increase for chronic patients at both 3 and 8 weeks.
- Interested parties are encouraged to reach out to the authors for further details about the enhanced blinding procedures.
- Particularly on the vigilance reduction (VIR) dimension of the 5D-ASC scale, which is related to fatigue. Therefore, the authors suggested an active control with sedating effects such as a benzodiazepine or antihistamine would serve as a more appropriate comparator.
- As with the negative primary outcome, had there been more participants, there may have been significant differences other than solely nausea.
- More speculatively: out of two participants in the placebo group excluded from the final analysis due to protocol violations, one was because they started using “psilocybin-containing mushrooms on their own shortly after the dosing sessions completed while they were still in cycle and the headache diary was still being kept” – might this indicate they were aware of their placebo assignment?
- Watch on YouTube.
- If you’ve made it this far, you may be wondering why the COMPASS Phase 2b publication (Goodwin et al.) was excluded from this top 5 list. Despite being an obvious choice due to sheer size and multi-site nature alone, it was omitted because of the already extensive coverage provided earlier in our “Late- Stage Psychedelic Trials” section of the Year in Review.
- Important negative finding; While there were issues with the dosing protocol, it is a good reminder that the proposed transdiagnostic efficacy of these substances needs to be systematically evaluated.
- For bonus justified praise and criticism, see Schatzberg 2022 and Cristea et al., 2022, respectively.
- For the accompanying qualitative results, see Hendricks et al., 2022 – more trials should publish these results concurrently.
- See our Research Themes section for more.