
Psychedelic Policy & Law in 2022
Psychedelic Policy & Law in 2022
Psychedelic drug policy reform initiatives continued to accelerate in 2022, with North America remaining the most active locale for such efforts. Given the fast-paced nature of such changes, this section briefly reviews a number of key initiatives and themes across the U.S., Canada and Europe.

Part of our Year in Review series
The United States
In December 2022, an article published in JAMA projected that a majority of states will legalise psychedelics by around 2034-2037. While the model used by the researchers placed a lot of credence in the idea that psychedelic drug policy reform will follow a similar temporal and geographic trajectory as marijuana reforms did, it’s nonetheless reflective of a broader feeling that psychedelic drug policy reform is predestined; at least in a majority of the fifty states.
As we noted in a November Bulletin, the state-by-state medical and recreational marijuana timeline does demonstrate some translational utility: California, Oregon and DC, for example, were among the first states to pass medical marijuana legislation in the mid-to-late 1990s. Today, they’re hotbeds of psychedelic drug policy reform initiatives; and successes.
Here, we look at two of the most substantive state-level initiatives that emerged or progressed in 2022, before turning to nascent efforts at the federal level. We then review some themes in psychedelic drug policy reform as well as other legal matters such as litigation and increasing scrutiny of ketamine clinics.
Colorado’s Natural Medicine Health Act Wins Voter Approval
In November 2022, Colorado became the second state to vote for the implementation of a legal psilocybin services program when voters endorsed Proposition 122, the Natural Medicine Health Act (NMHA). Arguably the most substantive psychedelic policy reform measure passed in the U.S. to date, the NMHA creates a state-regulated supervised psilocybin therapy program that should be ready for 2024, and decriminalises a number of psychedelics.
It was a tense run-up to the elections for proponents and opponents of the Proposition alike, with a poll conducted in the last week of October suggesting a very close call. And close it was:
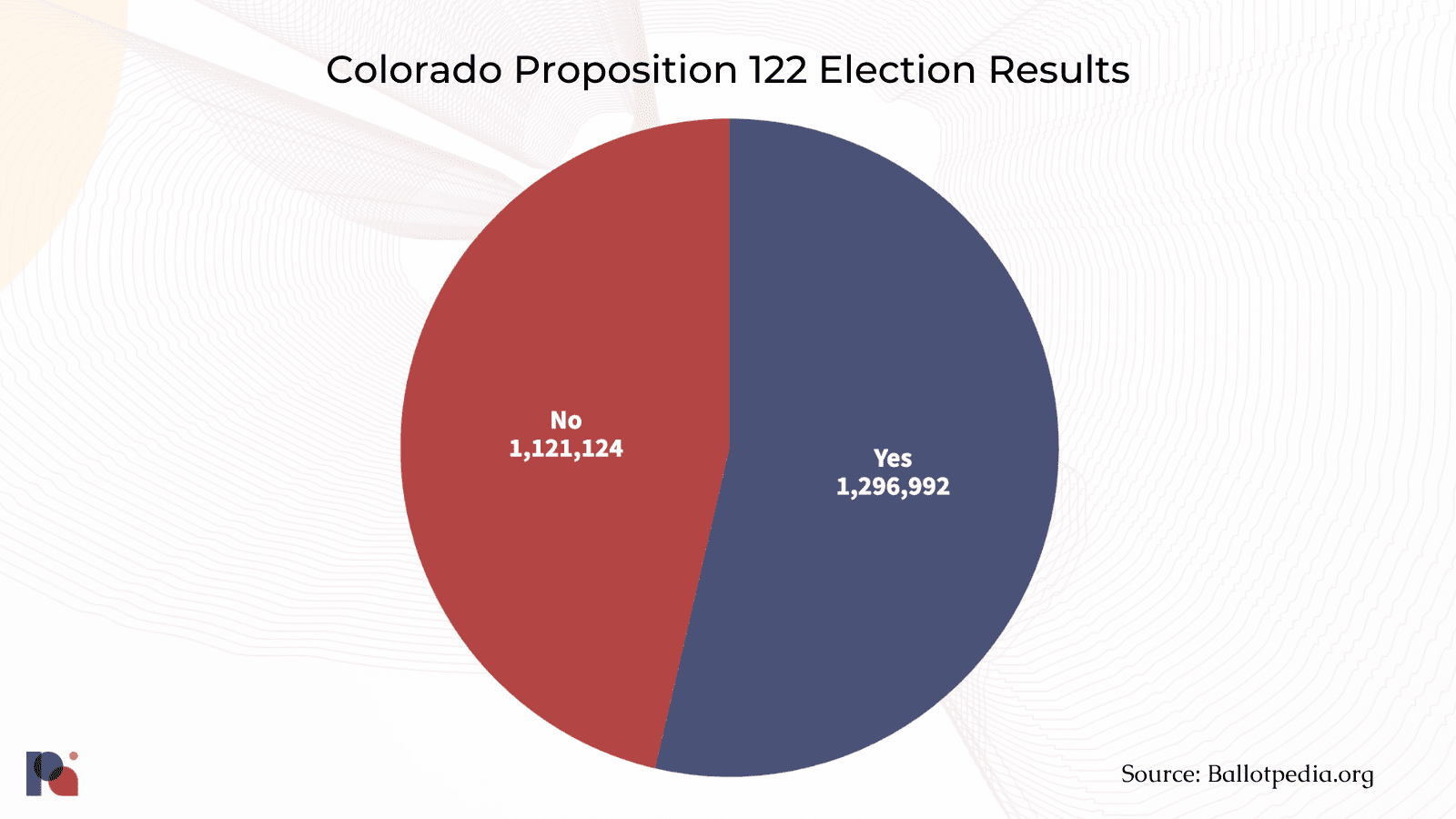
Now, NMHA’s advisory board will stew over the nuts and bolts of the program’s implementation for 18 months.
Oregon’s Psilocybin Services Face a Challenging Start
Oregon’s efforts to establish a regulated Psilocybin Services program by the beginning of 2023 reached a crescendo in 2022, as the realities of the undertaking became apparent to insiders and observers alike.
The most basic of these realities is the ability of Oregon Psilocybin Services (OPS) – the dedicated section housed within the Oregon Health Authority (OHA) to oversee the implementation of the psilocybin services program endorsed by voters in Measure 109 – to remain financially solvent. Once psilocybin services are up-and-running in 2023, the section appeared to have expected to sustain itself via a fee-based budget (i.e., license fees paid by service providers and associated taxes on psilocybin).
But, late in 2022, OPS felt compelled to request additional state general funds (around $6.6 million) to sustain its work through 2023, bridging an anticipated shortfall in funding. Given that the licensing fees are already substantial ($10,000 per year for service centres, manufacturers, and testing labs; $2,000 per year for facilitators), OPS presumably felt that it was not possible to increase them further.
OPS told Psychedelic Alpha that, for the 2023-25 biennium, it has estimated the following number of licensees:
- Facilitators: 750
- Service Centers: 28
- Manufacturers: 26
- Testing Labs: 13
Even if these lofty targets are reached (all signs point to the contrary, at present), it still leaves OPS in a precarious situation and a present shortfall.
To be sure, it’s standard procedure for state agencies to request funds from state governments, with Oregon Health Authority reporting around 50 policy packages in its 2023-25 requests. Nevertheless, it will be interesting to see if and when OPS becomes self-sustaining in the future; as the original Measure had suggested to voters.
New efforts to implement Oregon-esque regulated access programs will likely seek to learn from the Beaver state’s self-imposed restrictions. Colorado, for example, will allow its program’s funds to come from a broader range of sources, including donations and grants1.
Another political reality was rural Oregon’s distaste for psilocybin services, which manifested via an Opt-Out option baked into Measure 109, which allows for local jurisdictions to opt-out of allowing psilocybin services within their borders.
During the November 8, 2022 elections, many Oregonians used this option, with large swathes of Oregon’s landmass turning red on our dedicated Opt-Out Tracker produced in collaboration with Emerge Law Group, Calyx Law, and Healing Advocacy Fund.
Circles represent cities, conforming to the same color-coding as counties.
[display-map id=’15875′]
But, as we explained prior to the elections2, when viewed through the lens of population density the situation is less concerning for proponents of psilocybin services in Oregon.
Take the five most populous counties in Oregon, for example. At the time of our reporting in late August, just two of them put opt-out ordinances on the ballot:
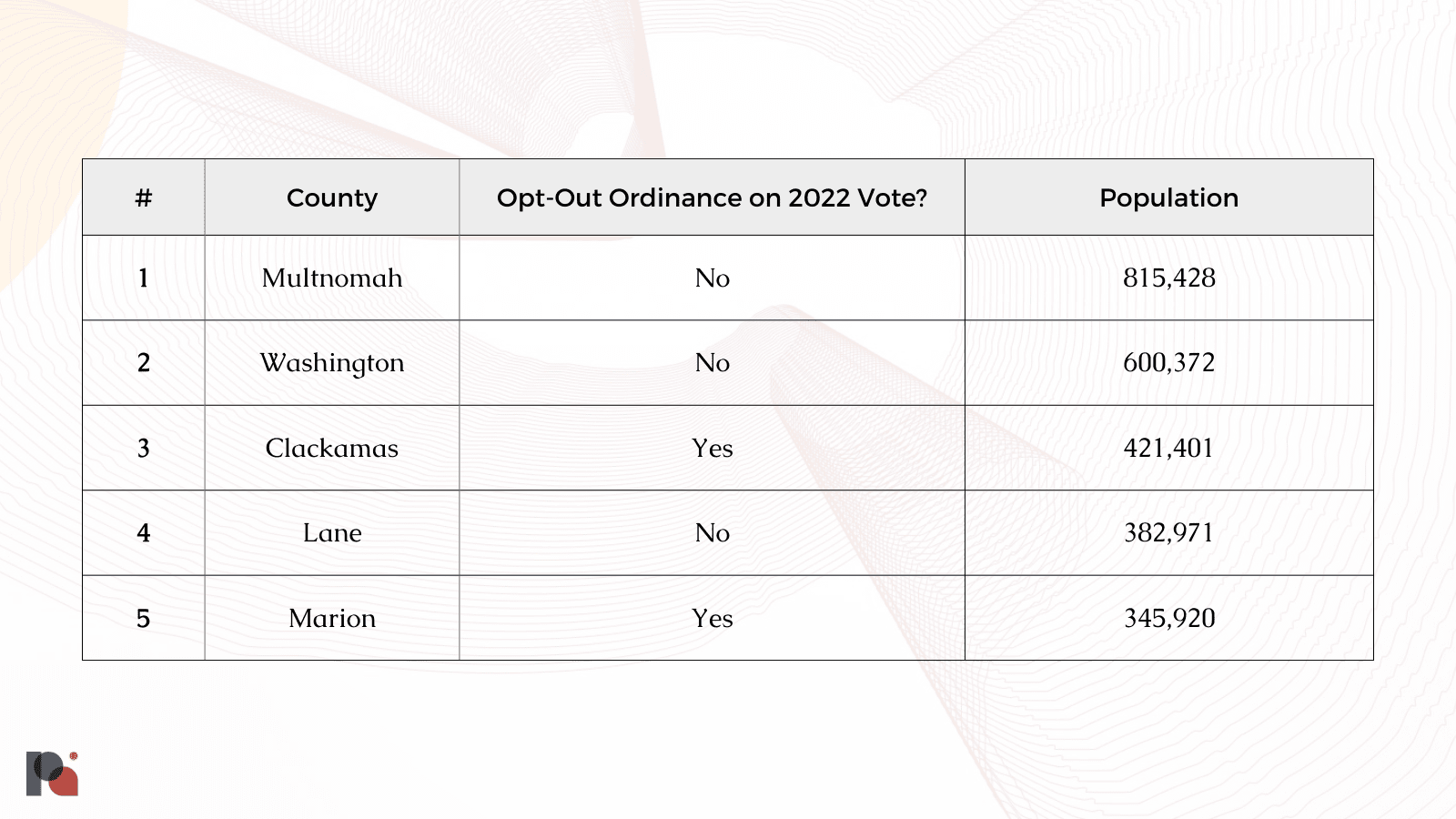
Indeed, both Clackamas and Marion opted-out in the November elections.
With a combined population of around 2.5 million, these five counties alone represent just over 60% of Oregonians. The seven Eastern-most counties – all of which are staunchly Republican, rural, and have low population densities – all voted to opt-out. But, despite representing around a third of the landmass of Oregon, they represent a combined population of around 175,000 people: just over a fifth of Multnomah.
As such, our map is visually quite misleading in showing such large swathes of red, or counties and cities that ‘Opted-Out’ in November 2022. If we adjusted the size of counties on the map by their population, the picture would be much different.
This point was picked up by The Oregonian’s coverage, which noted:
“A map from Psychedelic Alpha shows where opt-outs passed. While it shows nearly two-thirds of the state will not allow psilocybin centers and manufacturing, the places where the majority of Oregon’s population live, the Portland metro area and the majority of the Willamette Valley, are all opted-in.”
It’s even more misleading when you take into account the fact that city land is subject to city regulation: it is only unincorporated land outside of city limits that is subject to county control. As such, cities within opt-out counties could choose to remain opted in, and become ‘safe haven’ cities. These are represented as green blobs amidst red counties on the map.
The population of these cities is often substantial, meaning that even in counties like Clackamas and Marion, the most populated cities like Portland and Salem, respectively, are “safe haven” cities for Oregon Psilocybin Services (i.e., they’re not opted-out).
Such is the importance of delving deeper into the data.
***
As we noted in the days after the elections, there are also a few other points to keep in mind:
- Not all of these opt-outs are permanent; some are temporary moratoriums.
- Public education and engagement appears to work: voters in Deschutes County convincingly rejected the opt-out ordinance. Deschutes was home to one of a handful of campaign groups formed specifically to oppose opt-outs. On this point Sam Chapman, Executive Director of the Healing Advocacy Fund, told Psychedelic Alpha that this shows that “when people hear about the potential of psilocybin therapy from their own community members, they understand why it’s important”.
- It’s important to appreciate the equity and access implications of the new geography of psilocybin services in Oregon.
If Oregon’s psilocybin program proves to be a success, there’s no reason that wider adoption won’t emerge among the presently ‘opted-out’ counties. Fluence’s Victor Cabral told us that, “the hope is that over time, successful implementation and improvement of the Measure 109 program in counties that opt-in will lead to wider adoption across the state.” Chapman agreed, predicting that “as the therapy gets implemented in Oregon communities in 2023 and beyond, voters will continue to learn about how it can help address our state’s mental health crisis, and over time access will only continue to expand.”
Beyond these more existential threats to OPS and the local contours of psilocybin services availability, there are a bevy of logistical issues that would-be psilocybin service centres and other operators are facing.
Harris Bricken’s Vince Sliwoski has been covering these challenges via the firm’s Psychedelics Law Blog, and identifies his ‘top five’ challenges as:
- “The lack of a retail sales or an off-site use allowance, to supplement the on-site, supported adult use model
- The high cost of licensing for facilitators in particular (up to $8K for certification, plus $2K in OHA fees annually)
- The length of administration sessions, which can be six hours minimum at the highest doses (and not reimbursable by insurance)
- The decision by 137 towns and counties to opt out of psilocybin services
- Intractable issues caused by federal prohibition, especially around taxation and lack of banking”
These more mundane issues, which also include zoning and land use rules, will likely further slow or stunt the establishment of psilocybin services in Oregon.
Federal Psychedelic Drug Policy Reform Efforts Emerge
To date, many psychedelic drug policy reform efforts have taken place at the local or state level. However, 2022 saw the emergence of a number of efforts pushing for psychedelic policy reform at the federal level, with varying degrees of coordination. In brief, these include:
- Federal Task Force: In May, a letter from Assistant Secretary of Health and Human Services for Mental Health and Substance Use Miriam E. Delphin-Rittmon to Representative Madeleine Dean explained that SAMHSA is, “exploring the prospect of establishing a Federal Task Force to monitor and address numerous complex issues associated with emerging substances”, with reference to psilocybin and MDMA. Psychedelic Alpha understands that the establishment of such a Task Force is underway, with the involvement of groups like Reason for Hope and The Veterans Mental Health Coalition (VMHC).
- Breakthrough Therapies Act: Introduced by Cory Booker and Rand Paul, this bipartisan effort would reduce barriers to psychedelic research and access3. We provided further details in our December analysis of the act.
- NDAA amendments: In perhaps the most bipartisan pairing of representatives possible4, Dan Crenshaw and Alexandria Ocasio-Cortez forwarded amendments to the National Defense Authorization Act (NDAA) for 2023, which would modestly reduce federal restrictions on certain psychedelic research. The House of Representatives adopted the amendments, which were likely more of a political win than a substantive one. Our commentary from July provides more colour.
- The Congressional Psychedelics Advancing Clinical Treatments (PACT) Caucus: with involvement from a wide array of groups, this new congressional caucus has been celebrated by the likes of MAPS. Its goals include increasing awareness of PATs among members of congress and their staffs, supporting increased federal funding for psychedelic science, and examining regulatory impediments to psychedelic research. The Caucus will not offer recommendations on the decrim., legalisation, or re/de-scheduling of psychedelics.
Other Themes in U.S. Psychedelic Policy and Law in 2022
Local Decriminalisation Efforts Continue
- As you can see from the non-exhaustive timeline at the very bottom of this webpage, a number of decriminalisation efforts made headway in 2022.
- Take California, for example. While a state-wide psychedelic decrim. effort was resuscitated in late 2022, local decriminalisation efforts continued in the Golden State, with San Francisco passing something akin to decriminalisation of psychedelics (though, not exactly) and the city of Berkeley looking to decriminalise a number of psychedelics (including LSD) via a resolution that passed the Berkeley Health Commission in late 2022.
At-Home Ketamine Comes Under Scrutiny
- As a COVID-era waiver to the Ryan Haight Act allowing remote prescribing of scheduled substances looks set to sunset in 2023, where does this leave ketamine telehealth companies? When viewed in the context of a growing number of unfavourable media reports of these at-home ketamine offerings, some companies are bracing for impact.
- As we covered in May 2022, the fate of San Francisco-based mental health startup, Cerebral, may be a canary in the coalmine5. The company, which became known for prescribing Adderall (a Schedule II substance) without in-person consultations, is under investigation by the U.S. Department of Justice for potential violations of the Controlled Substances Act.
Psychedelics-Related Litigation Ramps Up
In 2022, we saw a number of psychedelics-related litigation headlines. While it’s still a small crop, it’s interesting to see a variety of cases emerge, from inter-company complaints right through to challenges to the DEA. Here are a handful of examples:
- DEA Ditches Plan to Schedule 5 Tryptamines: In the Summer of 2022, the DEA scrapped a proposal to place five tryptamines on Schedule I after a group of researchers, psychedelic startups and lawyers challenged the agency. We sifted through around two hundred documents related to the case to produce our most in-depth Special Report of 2022, Inside the Challenge to DEA’s Proposed Scheduling of 5 Psychedelic Tryptamines.
- DOI and DOC Escape Scheduling: Later in 2022, the DEA backpedalled on another attempt to schedule psychedelics. While both instances of the DEA bowing to public pressure and legal challenges are promising, many expect the agency to return with fresh plans to schedule such substances.
- Terran Biosciences files lawsuit alleging that COMPASS “milked” researcher for trade secrets before “secretly” filing patent on same technology: This complaint, which is understood to constitute the first trade secret lawsuit in the psychedelics space, was filed on the 5th of August, 2022. We covered it in an August Bulletin.
- Two Post-Grant Reviews Denied: In June, COMPASS Pathways survived two challenges to its Polymorph A patents, which are understood to cover its crystalline forms of psilocybin including COMP360. The challenges were brought by Freedom to Operate, a non-profit group founded by Carey Turnbull dedicated to “protecting psychedelic science and medical development for public benefit”. At the time we described this as, “an unreserved win for COMPASS”, explaining that the Patent Trial and Appeal Board (PTAB) had sided with the company on both of the claim constructions, finding each of FTO’s invalidity arguments unsupported and unpersuasive. Requests for rehearing and further review, filed in July and August, remain pending.
- AIMS v. DEA: see Kathryn Tucker’s description of events. Though, as with all contributions to this Review series, do note that new developments may have emerged since the time the contribution was authored.
The Emerging Role of the VA
- Given the politically and culturally salient nature of veterans in the U.S., it’s unsurprising that many policy reform and advocacy efforts are foregrounding the plight of many in this community. In October, the Department of Veterans Affairs published an Evidence Brief, Psychedelic Medications for Mental Health and Substance Use Disorders. “Evidence is therefore very preliminary and several critical gaps need to be addressed by future research”, the report concludes.
Canada
Canada has been a hotbed for psychedelic business and advocacy for a number of years, from its Special Access Program amendments through to successful localised efforts in the (psychedelic) drug policy reform arena. Here, we look at a handful of developments from 2022.
Special Access Program (SAP) Amendments Provides Very Modest Access to PATs
Near the end of December 2020, Canada’s leading health agency, Health Canada, notified the general public of its intent to pursue regulatory amendments which would restore access to “restricted drugs”6 through the agency’s Special Access Program (SAP). Over the course of a two-month public consultation period, Health Canada received 392 responses from individuals and organisations. Interestingly, expanded access to psychedelics was a leading theme in responses gathered by Health Canada. As the agency described:
“80% of all respondents associated Health Canada’s proposal with increasing access to psychedelic restricted drugs (e.g. MDMA, psilocybin, LSD, DMT), often for the treatment of various conditions, most notably mental health disorders.”
On January 5, 2022, Health Canada’s amendments were announced, allowing the Minister of Health to authorise, “the sale of an unapproved drug from a manufacturer to a practitioner for the emergency treatment of a patient under certain circumstances.” However, SAP requests for psychedelics are limited to only psilocybin and MDMA (Pazdan & Goldgrub, 2022).
In its Gazette notice, Health Canada noted how many public consultation respondents erroneously believed that these regulatory changes would allow for a, “guaranteed access to psychedelic restricted drugs.” In light of these frequent misinterpretations, the agency emphasised that its amendments would not support such widespread access to restricted drugs, nor should they be interpreted as an indication of Health Canada’s desire to move towards broader decriminalisation or legalisation.
Instead, access requests are to be submitted on behalf of qualifying patients by eligible practitioners through the SAP. Submitted requests will be considered “on a case-by-case basis” and, in doing so, the agency will take into account the existing body of clinical evidence that supports the use of the drug(s) for a patient’s condition. Interestingly, Health Canada described how requests “are normally only considered when positive results of Phase II or Phase III clinical trials are already available.”
Furthermore, as Pazdan & Goldgrub (2022) explain:
“SAP requests, however, will only be granted for patients with a ‘serious or life-threatening condition’ in cases where other conventional treatments have failed, are unsuitable for the patient, or are not available in Canada.”
Prior to Health Canada’s recent SAP amendments, patients hoping to access psychedelics required the Minister of Health to grant an exemption to the Controlled Drugs and Substances Act (CDSA) under Section 56 of the act. While dozens of patients have successfully gained access to psilocybin using the section 56 exemption pathway, wait times can be notoriously long. Furthermore, patients granted these exemptions are required to source their own drugs.
Conversely, patients who obtain access to psilocybin or MDMA using the recently-amended SAP are not required to source their own supply. Instead, Health Canada-approved dealers provide their manufactured drugs to the patient’s health care provider.
While the new process was widely viewed as a step in the right direction, shortly after the amendments came into effect applicants, many of whom had sought access exemptions through the section 56 pathway, began to have their applications denied by Health Canada.
In one such case, Health Canada informed a patient that they “had not exhausted all possible legal routes available” to obtain access to psilocybin, and as a result were not being approved for an exemption under section 56 of the CDSA. In fact, as a CBC article published on February 28, 2022 described, zero section 56 exemptions had been granted since psilocybin was added to the SAP list earlier in 2022.
Thus, it appears that in the eyes of Health Canada patients seeking medical access to certain psychedelics should first apply through the amended SAP, and rely on section 56 of the CDSA as a secondary option.
However, these rejections applied not only to patients seeking medical access, but also to health care providers who sought section 56 exemptions to access psilocybin for training reasons. In fact, near the beginning of 2022, Health Canada rejected the applications of roughly 80 Canadian healthcare professionals. In response, Health Canada suggested that practitioners interested in training should do so by participating in an approved clinical trial.
These rejections and limits on access to psychedelics prompted seven patients and one practitioner to file a lawsuit against the Canadian Government and Minister of Health. The plaintiffs, with support from TheraPsil, are “arguing for a constitutional right to individualised psilocybin-assisted therapy” (Pazdan & Goldgrub, 2022).
The charter challenge cites a number of important deficiencies (prohibitions) that unjustly limit existing options available to patients and providers who wish to access psilocybin. Accordingly, the group claims that the prohibitions referenced in their challenge violate section 7 of the Canadian Charter of Rights and Freedoms, which guarantees “the right to life, liberty and security of the person”.
As Pazdan & Goldgrub (2022) describe in their review of Canadian psychedelic law, the challenge does not appear to be without strong precedent:
“The patients’ argument is a strong one, clearly echoing that of the successful 2000 Parker case. In Parker, the Ontario Court of Appeal held that the law broadly prohibiting access to medical cannabis violated the epileptic plaintiff’s Charter rights. Notably, the court regarded the possibility of the plaintiff accessing medical cannabis by means of s. 56 exemption as insufficient to save the government’s legal stance. As a remedy, the court mandated that the federal government create Canada’s first regulated, individualized medical cannabis regime within a year.”
As we move further into 2023, it will be interesting to see how this Charter challenge unfolds, and whether it might have broad impacts on medical access to psilocybin in Canada.
While the amended SAP certainly failed to meet the needs or expectations of many, on March 21, 2022, Health Canada authorised Dr. Valorie Masuda to treat six patients for their end-of-life distress using psilocybin. Subsequently, in May 2022, Numinus Wellness and Braxia Scientific both announced that they had received SAP approval from Health Canada to treat patients suffering from depressive disorders using psilocybin.
More positive news emerged near the end of the year, when TheraPsil announced that two physicians had successfully billed for and were paid by the province of Quebec “after completing psilocybin-assisted psychotherapy for a patient with legal access”, making the province of Quebec the first to cover the costs of psilocybin-assisted therapy.
Alberta Becomes the First Province to Regulate PATs
On October 5, 2022 the government of Alberta announced that it would be amending province’s Mental Health Services Protection Regulation to, “introduce new requirements for psychedelic assisted therapy.” The amendments, which came into effect in January 2023, made the province the first to regulate psychedelic-assisted therapies in Canada
Pursuant to the new regulations, only a “psychiatrist or a physician in consultation with a psychiatrist” will be permitted to prescribe psychedelics to patients. Furthermore, service providers (medical facilities) who wish to deliver psychedelic therapies must, “apply and hold a licence under the Mental Health Services Protection Act”, in addition to appointing “a psychiatrist as a medical director” who is charged with overseeing “all clinically related aspects of the licensed services.”
Psychedelic therapies in Alberta must take place only at a medical facility or clinic, except in cases of end-of-life care. Patients are also not permitted to self-administer the drugs: psychedelics can only be administered by an eligible health professional, who is then required to “monitor, treat and care for the patients while in an altered state of consciousness.” These care providers include psychiatrists, clinical psychologists, and other authorised health care professionals such as occupational therapists, nurses, and social workers.
While a number of pro-psychedelics groups heralded the announcement as a success, it’s not so simple. These new regulations could be used as a way to bear down on unlicensed providers, as opposed to a signal that the province intends to institute pre-approval access to PATs in any meaningful way. It’s important to remember that the regulation of drugs and controlled substances falls under the remit of the Government of Canada, not provincial governments.
While these changes demonstrate an awareness of psychedelic therapy, and some level of expectation that PAT is coming down the pike, it’s unlikely that they represent any meaningful increase in the accessibility of psychedelic therapy for now.
BC Decriminalises Possession of Certain Amounts of Controlled Substances, Including MDMA
In November 2021, the Canadian province of British Columbia (BC) submitted an “exemption request” under Section 56(1) of the Canadian Controlled Drugs and Substances Act (CDSA) to the country’s federal health agency. In its submission, the province described how the “criminalization of simple possession remains a significant impediment to BC’s ability to implement a comprehensive public health response to the illicit drug poisoning crisis.” Accordingly, the province sought to decriminalise personal possession of certain amounts of controlled substances.
On May 31, 2022, representatives from Health Canada announced that BC’s request had been granted, and that the exemption would come into effect later in the new year. As of January 31, 2023, adults in BC (18 years and older) will no longer face criminalization for the possession of less than 2.5g of an opioid, crack and powder cocaine, methamphetamine, or MDMA. Instead, individuals found to be in possession of less than 2.5g of these substances will be “offered information about health and social supports.”
Whac-A-Shroom: Psychedelic Dispensaries Pop Up, Slapped Down, Across Canada
Many Canadians might remember the surge in transient, unlicensed cannabis dispensaries that began to pop up well in advance of the federal government’s move to legalise and regulate the drug for recreational use. While many of these establishments were (repeatedly) shut down by local police action, the unregulated industry proved to be a rather resilient thorn in the side of municipalities and politicians who sought to put an end to illegal dispensaries operating in their jurisdictions.
Now, a few years after licensed cannabis dispensaries were permitted to open their doors across the country, a new group of speciality shops have begun to appear. While not yet as pervasive as their cannabis predecessors, magic mushroom dispensaries are growing in popularity in many of Canada’s most populous cities. Over the course of 2022, cities like Vancouver, Toronto, Ottawa, and Hamilton all saw new brick and mortar establishments open dedicated to selling magic mushroom products, in spite of the drug’s status under Canada’s Controlled Drugs and Substances Act.
Some of these new shops have successfully evaded municipal or police action for periods of time. However others, like the Mushroom Cabinet in Hamilton, have not been so fortunate. In late December it was announced that, just one day after opening its doors, Hamilton’s first dispensary of its kind was shut down by police.
In light of the absence of any federal commitment to psychedelic decriminalisation or legalisation, it will be interesting to see how this unregulated, public-facing industry develops, and how different jurisdictions across the country respond to their presence.
Health Canada Issues Guidance for Psychedelic Clinical Trial Sponsors
Near the end of 2022, Health Canada issued guidance outlining its “expectations regarding the implementation of risk-management measures” by sponsors of clinical trials investigating psychedelics. As we discussed in a twitter thread on the announcement, Health Canada recognized the growing interest in psychedelic-assisted psychotherapy, and due to the potential “psychological and physical risks” trial participants face, the federal agency felt the need to establish best practice guidelines for clinical trials.
In its notice, Health Canada described a number of “risk-management measures” that are to be addressed and included in any clinical trial application. Highlights of these measures include:
- Proper training for therapists on “evidence-informed protocols” for PAT.
- Therapists should be licensed to provide psychotherapy, or, in cases where therapists teams are not regulated, members “should be under the direct supervision of a licensed therapist.”
- “Rapport between the clinical trial participant and the therapist is critical”.
- “During the phase in which the drug is being administered to the participants, there should be a minimum of two therapists present.”
- “The therapists should remain the same through the clinical trial for each patient in order to maintain trust.”
- “Lodging should be available on the day of the administration of the drug in cases where it is preferable to keep participants overnight to help ensure their safety.”
- “Throughout the clinical trial, sponsors are required to report any serious unexpected adverse drug reactions to Health Canada.”
- “Consent for touch should be obtained prior to the administration of the investigational product, as well as in the therapeutic moment.”
- “Any physical contact between the therapist(s) and the participant must be of a non-intimate and non-sexual nature.”
Europe
European policymakers and psychedelic drug policy reform advocates might have got off to a slower start than those ‘across the pond’, but there are promising developments at the Member State and EU-wide levels.
A Member of the European Parliament, Robert Biedroń, told Psychedelic Alpha: (from Poland and representing political group of the Progressive Alliance of Socialists and Democrats):
“In Europe, over 100 million citizens are affected by mental health conditions and substance use disorders, and treatment options for these diseases are often suboptimal. Many members of the European Parliament – including myself – recognize a tremendous potential of psychedelic-assisted therapies in addressing huge unmet needs in those therapeutic areas. This year, we will work with PAREA to establish in the European Parliament an alliance of supportive parliamentarians to raise awareness in the Parliament about those novel treatments and call upon the European Commission and EU member states to already start preparing for their likely approval by the European Medicines Agency in the coming years.”
Tadeusz Hawrot, founder of the Psychedelic Access and Research European Alliance (PAREA) which emerged in 2022, is in an excellent position to provide an update on EU-level discussions regarding psychedelics.
Editor’s note: remember that the remit of this Year in Review is limited to events that happened in 2022, so we (nor our contributors) have not mentioned promising indications from early 2023 in this report, which include a related publication in the Lancet co-authored with European Medicines Agency representatives.
Beyond supranational efforts, individual Member States are looking to better understand, and engage with, the emerging psychedelic medicine landscape. One such country is Czechia (the Czech Republic), which has a fascinating history with psychedelics and related research; especially LSD. Today, there are a number of instances of psychedelics entering into discussions among policymakers in the central European country.
We are grateful to Tereza Dleštíková, Assistant Professor and Researcher at the Police Academy of the Czech Republic in Prague, who has provided us with a brief dispatch covering these developments:
Appendix: A Non-Exhaustive Timeline of Psychedelic Bills and Resolutions in 2022

Part of our Year in Review series
This content is part of our 2022 Year in Review, which looks back at the past year through commentary and analysis, interviews and guest contributions.
Receive New Sections in Your Inbox
To receive future sections of the Review in your inbox, join our newsletter…
Browse Other Sections of the Review
To explore our Year in Review, visit this webpage.
Join thousands of psychedelics insiders
Get the weekly psychedelic medicine briefing.
A free weekly digest covering trials, regulation, policy, and access.
By signing up, you agree to our privacy policy. You can unsubscribe at any time.
- Though, this poses an entirely different set of issues: predominantly on the ethics and conflicts front.
- At a time when we were tracking states and localities at risk of opting out.
- The act effectively replaces the Right to Try Clarification Act.
- Though, it’s likely that the pair did not collaborate on their separate amendments.
- Interestingly, a couple of months before the DOJ investigation came to light, Cerebral partnered with Field Trip Health to “provide end-to-end mental health care”.
- “Restricted drugs are controlled substances regulated under Part J of the Food and Drug Regulations. They generally have no approved medical uses and can only be used for scientific or research purposes, such as clinical trials.”