
Psychedelic Policy Reform & Legal Markets in 2023
Part of our Year in Review series
2023 was another significant year for psychedelic-related policy reforms and developments. For the purposes of this section of our 2023 Year in Review, we take a broad view of reforms and include not only bills introduced in legislative bodies but also ballot initiatives, attempts to carve out funding from state agencies, and notable discussions in public arenas.
We then look at how the growing crop of legal psychedelics markets fared in 2023. These include the state-legal psilocybin market in Oregon and Australia’s limited rescheduling of MDMA and psilocybin which allows their prescription in certain cases. Colorado, meanwhile, is gearing up to launch its own state-legal psychedelics program.
Notable Psychedelic Policy Reforms and Related Developments
U.S. State-Level Psychedelic Policy Reform
According to research by Calyx Law, more than half of U.S. states considered some form of psychedelics-related legislation in 2023.
While the majority of these efforts failed, that’s not necessarily a bad omen: according to LexisNexis, 20% of bills introduced are eventually passed, when averaging across all 50 state legislatures1.
Here, we review a handful of notable 2023 stories in state-level psychedelic policy reform and related developments.
California’s Double Fault
Perhaps the most significant psychedelics-related bill to pass a state legislature in 2023 was California’s SB 58, which would have effectively decriminalised the personal possession and use of small quantities of psilocybin (and psilocin), DMT and mescaline.
While the bill passed through the legislature by a narrow margin, California voters appeared to be generally on board with the measure. August 2023 polling by FM3 indicated that 60% of likely 2024 voters expressed that Governor Newsom’s endorsement of the bill would have a positive or neutral impact on their opinion of the Governor.
But Newsom was not willing to autograph the decrim. bill, instead opting to return it to the Senate unsigned on Saturday October 7th, effectively vetoing it. We speculated on his reasons for doing so at the time, which likely included national political sensitivities (which we first flagged in September).
For advocates of the bill, it’s not all doom and gloom. In his veto letter, Newsom left the door ajar by inviting legislators to introduce a new bill in the next session “that includes therapeutic guidelines”. He also acknowledged the “exciting frontier” represented by psychedelic research, adding that “California will be on the front-edge of leading it”. (See Analysis: California Governor Vetoes Psychedelic Decrim. Bill)
Senator Scott Wiener, who introduced SB 58 following two prior failed attempts at psychedelic decrim. in the state, heeded Newsom’s invitation. Along with a Republican colleague, Marie Waldron, he is forwarding an amended version of AB 941. (See Wiener Regroups and Fourth Time Lucky for California Psychedelic Policy Reform?)
Aside from Wiener’s legislative efforts, a group called TREAT California sought to get an ambitious plan to fund a $5 billion psychedelic R&D agency on the 2024 ballot. Ultimately, the initiative was withdrawn following unfavourable polling, which suggested voters in the Golden State did not endorse the creation of a state agency to fund the program. (See TREAT Retreats)
A Turf War in Massachusetts
The Bay State has been a hotbed of psychedelic policy reform for some time, with at least a half-dozen local jurisdictions in the state having decriminalised psychedelics since 2021. Now, advocates believe they’re nearing success at the state-wide level.
But, 2023 saw tense debate between a local “grassroots community group”, Bay Staters for Natural Medicine, and the national New Approach PAC. Bay Staters were behind successes in securing decrim. in all of the aforementioned localities, as well as others such as Portland, Maine. New Approach PAC, meanwhile, was behind successful state-wide initiatives like those seen in Oregon and Colorado, as well as cannabis ballot initiatives. Bay Staters describe New Approach as a “DC-based PAC” that “rigged the rules for psychedelics in Oregon”. In Massachusetts, New Approach supports a campaign committee called Massachusetts for Mental Health Options.
Massachusetts for Mental Health Options floated two ballot measures in the state. Both looked substantially similar to those passed on Oregon and Colorado, but there was one major difference: Version A would allow for home cultivation of named psychedelic substances, while Version B would not.
According to a 2023 year-end report, Massachusetts for Mental Health Options raised nearly $4m in donations, with familiar faces like Dr. Bronner’s and Blake Mycoskie acting as keystone donors. Some lesser-spotted psychedelic philanthropists, such as HubSpot co-founder and CTO Dharmesh Shah, also supported the committee with substantial donations. In 2023, the committee spent $3.8m on activities related to the ballot initiative, the vast majority of which went to The Outreach Team. Interestingly, the committee made a $35,000 donation to Bay Staters for Natural Medicine on August 1st.
Bay Staters for Natural Medicine has taken a dim view of Massachusetts for Mental Health Options in general, but particularly their B-side ballot initiative that would not permit home-growing.
And that August donation certainly didn’t placate the grassroots group. Later in the year, Bay Staters claimed to have video footage of “canvassers for New Approach PAC … lying to voters” and sought to substitute the ballot question for “a more affordable alternative”.
After both versions of the ballot were cleared by the state’s Attorney General, Massachusetts for Mental Health Options decided to forward Version A, which includes home growing. The ballot initiative, titled Massachusetts Regulated Access to Psychedelic Substances Initiative, has been transmitted to the legislature. It could then be passed on to voters, subject to additional signature collection, who would decide its fate in the forthcoming November 5th, 2024 election.
The ins and outs of the situation are far more complex and nuanced than the present review allows for. Those interested in diving deeper may find Mason Marks’ coverage in Psychedelic Week useful, though apparently partisan.
One clear thread that emerges is a tussle between local actors and what are perceived as out-of-state influences, such as New Approach PAC. There also appears to be a fundamental difference in approach, with New Approach supporting regulated use models which—at least in early iterations—might be associated with significant barriers to entry such as fees and licensing hurdles; and groups like Bay Staters forwarding more fundamental, straightforward drug policy reform like decriminalisation.
While New Approach’s, well, approach, resembles2 something closer to respectability politics, Bay Staters’ is much more grassroots and unapologetic.
Arizona Funds Whole-Mushroom Psilocybin Studies
The passage of HB 2486 saw a $5m carve-out for grants to support clinical studies into psilocybin added to Arizona’s 2024 fiscal year budget. The program, overseen by the state’s Department of Health Services, was specifically designed to support studies of “whole mushroom psilocybin”, as opposed to synthetic or isolated forms.
Unfortunately, a drafting error meant the bill did not include text allowing the Department of Health Services to disperse the money over a series of years. Instead, any unused funds would be absorbed back into Arizona’s general fund at the end of the 2024 fiscal year (June 30th, 2024); which is a very short timeline for studies.
Efforts to extend the funding period through the 2026 fiscal year appear to be succeeding, thus far.
Washington’s Watered-down Bill Shows ‘Task Forces’ Are a Popular Middle-Ground
In May, Washington’s Governor Inslee signed the Washington Psilocybin Services Act (SB 5263), which established a Psilocybin Task Force and a modestly-funded ($1.4m) pilot program at the University of Washington.
But Inslee vetoed a number of the bill’s more progressive elements, opting instead for the task force and pilot program approach. Establishing advisory boards, working groups, or task forces has become a popular strategy in other states, too, including Nevada.
Some might view the creations of such entities as Governors and legislators kicking the can down the road, while others see them as a healthy compromise and an opportunity to build further support within the halls of government.
Kentucky’s Ibogaine ‘Manhattan Project’ Flops
Those who supported an ambitious plan to cleave off $42 million in opioid settlement funds to support ibogaine research and drug development lost a key champion in December 2023 as the Kentucky Opioid Abatement Advisory Commission (KYOAAC) saw a changing of the guard.
As we reported at the time, it looks like the project will be shelved following new leadership. (See Kentucky’s $42m Ibogaine Funding Appears Dead in the Water)
Local Decriminalisation Continues in the U.S.
In 2023, more than a half-dozen cities, countries, or other local jurisdictions passed actions that decriminalised or otherwise liberalised the possession and use of certain psychedelics within their limits.
These include Minneapolis (Minnesota), Eureka (California), Ferndale (Michigan), Jefferson County (Washington), Portland (Maine), Provincetown (Massachusetts), and Salem (Massachusetts).
While the majority of these actions took place via votes by local councils, Minneapolis’ came via an executive order from the mayor.
Psychedelics at the Federal Level
At the federal level, we saw plenty of psychedelics-related discussions in 2023. These included, but were certainly not limited to, the following (some of our contemporary coverage is linked at the end of each bullet point):
- DoD to Fund Psychedelic Trials in Active-Duty Service Members: The must-pass 2024 National Defense Authorization Act (NDAA)—which stipulates the annual budget for the U.S. Department of Defense (DoD)—contained a curious easter egg when it was finalised last year: an amendment that instructs the DoD to provide grant funding for research into the use of psychedelics to treat PTSD, traumatic brain injury (TBI) or chronic traumatic encephalopathy (CTE) in active-duty service members. The Act carved out $15m in grants per fiscal year from 2024 through 2028. (See Psychedelic Trials for Active-Duty Service Members Included in Passed NDAA)
- VA Subcommittee on Health Discusses Psychedelic Therapies: In November, the U.S. House Committee on Veterans’ Affairs’ Subcommittee on Health held a hearing titled Emerging Therapies: Breakthroughs in the Battle Against Suicide? The meeting marked the first time the House has substantially discussed psychedelic therapies, a milestone that the Chair, Mariannette Miller-Meeks (R-IA), made sure to highlight in her opening remarks. (See Veterans’ Affairs Subcommittee on Health Discusses Psychedelics)
- FDA Issues Draft Guidance on Psychedelic Trials: In June, the FDA issued its first draft guidance on clinical trials with psychedelic drugs. By late August, over 200 comments were made on the draft guidance—we read all of them to summarise key themes. (See Psychedelic Industry and Community Responds to FDA Draft Guidance; FDA Issues Draft Guidance on Psychedelic Trials)
- Federal Appeals Court Asks DEA to Re-Assess Psilocybin Rescheduling Petition: In November, a federal appeals court threw a lifeline to a psilocybin rescheduling effort forwarded by Dr. Sunil Aggarwal of the AIMS Institute. Aggarwal—who wishes to offer psilocybin to cancer patients he worked with—had his request for psilocybin to be placed in Schedule II denied by DEA in September 2022. (See Federal Appeals Court Throws Lifeline to Psilocybin Rescheduling Effort)
- Legislators Introduce the VISIONS Act: In September, Earl Blumenauer teamed up with Robert Garcia to introduce the Validating Independence for State Initiatives on Organic Natural Substances Act of 2023; also known as the VISIONS Act. It’s quite simple in that it would seek to prevent federal intervention in states with liberalised psilocybin laws by prohibiting “the use of Federal funds from preventing a State from implementing their own laws with respect to psilocybin.” (See A Cole Memo for Psilocybin? Not Quite, but It Could Be a Start…)
- DOI and DOC Fall Into DEA’s Crosshairs Once Again: In December, The U.S. Drug Enforcement Administration (DEA) published a notice of proposed rulemaking that would see two psychedelics, 2,5-dimethoxy-4-iodoamphetamine (DOI) and 2,5-dimethoxy-4-chloroamphetamine (DOC), placed in Schedule I of the Controlled Substances Act (CSA). The DEA previously published almost identical proposed rulemaking in spring 2022, but withdrew in late August 2022. (See DEA Relaunches Attempt to Place Two Psychedelics—DOI and DOC—in Schedule I)
- FDA Asked to Review First Psychedelic-Assisted Therapy New Drug Application: In December we learned that Lykos Therapeutics (fka MAPS PBC) submitted a New Drug Application (NDA) to the FDA for MDMA-assisted therapy for PTSD. (See MAPS PBC Files New Drug Application for MDMA-Assisted Therapy for PTSD)
A Slow Year for Psychedelic Reform in Canada
Compared to the years prior, Canada’s psychedelic policy reform and advancements were somewhat muted in 2023.
Psychedelic dispensaries continued to appear on the streets of Canada’s metropolitan areas. As observed in 2022, these outfits were often raided by local law enforcement, though many quickly re-opened. (See Whac-A-Shroom: Psychedelic Dispensaries Pop Up, Slapped Down, Across Canada from our 2022 review)
Elsewhere, the Federal Court of Canada refused attempts to overturn Health Canada’s decision to deny Section 56 exemptions sought by nearly one hundred healthcare professionals. They had hoped to benefit from these exemptions to legally undergo experiential training with psilocybin via the non-profit TheraPsil. Another case before the Federal Court, Thomas Hartle’s charter challenge that claims the government is failing to provide fair access to psilocybin for patients where their doctor is willing to support them, remains ongoing; albeit very slowly.
In November, Canada’s Senate Subcommittee on Veterans Affairs published a report calling for a robust research program into the potential of psychedelic-assisted therapy. (See Canadian Senate’s Subcommittee on Veterans Affairs: “The Time is Now” to Fund Psychedelic-Assisted Therapy Research)
Readers with a particular interest in Canada’s legal landscape concerning psychedelics might find Green Economy Law’s 2024 State of Canadian Psychedelic Law report helpful.
UK Parliament Hears Debate on Psilocybin
While policy reform progress is much slower in the UK than its North American peers, there was some progress in 2023.
In May, the House of Commons debated access to psilocybin treatments and issued a Research Briefing on the topic. The transcript of the debate is available via Hansard.

The Advisory Council on the Misuse of Drugs (ACMD) has been reviewing barriers to research involving Schedule I substances since 2017. In late December 2023, it finally published Part 2 of its report which included a number of recommendations. We await a response from the Government.
In August, the Parliamentary Office of Science and Technology (POST) published a Rapid response review titled, Psychedelic drugs to treat depression. More detailed notes are expected to publish in 2024.
Psychedelics-Related Discussions in the European Union
At the European Union level, several promising developments emerged in 2023.
In March, members of the European College of Neuropsychopharmacology (ECNP), along with the European Medicines Agency (EMA), penned a regulatory perspective piece in The Lancet marking the first time EU regulators had officially spoken in support of research into psychedelic medicines, according to PAREA.
A month prior to that publication, a cross-party group of Members of the European Parliament (MEPs) wrote to the EMA to argue that “the time is right” for EU agencies to act in a coordinated fashion to provide policymakers with the requisite information and direction to lead the way in psychedelic research and roll-out.
In September, EMA issued its latest draft Guideline on the clinical investigation of medicinal products for the treatment of depression, which includes a section on psychedelics. The draft guidance—which is concise, at around a page long—then detailed a half-dozen challenges that may be encountered in psychedelic clinical trials. Public consultation on the draft Guideline closes on the 31st of March, 2024. (See EMA’s New Draft Guideline for the Clinical Investigation of Depression Medicines Includes Section on Psychedelics)
In November, the inaugural meeting of the MEP Action Group for the Medical Use of Psychedelics took place at the European Parliament in Brussels. Attendees sought to explore how a revised EU pharma package could support this field, with speakers including Tadeusz Hawrot of PAREA, psilocybin trial participant Maryam Jabir, researcher Tomáš Pálenícek, Viktor Chvátal of PsychedelicsEUROPE, Damian Kettlewell of Clairvoyant Therapeutics, and this author (Josh Hardman). (See MEP Action Group Meets in Brussels to Discuss Regulatory Pathways for Psychedelics)
Legal Psychedelics Markets in 2023
In 2023, two new legal jurisdictions for psychedelic services emerged, and one progressed toward launch.
Oregon Launches First State-Legal Psilocybin Services Program
In January, Oregon began accepting licence applications for the first state-legal psychedelic program in the country. Things got off to a somewhat slow start, with a trickle of applications and eventual license issuances.
By May, Oregon Health Authority had issued the first psilocybin service centre licence, just two and a half years after Oregonians passed Measure 109 that mandated the creation of Oregon Psilocybin Services.
That first licence was awarded to EPIC Healing Eugene. EPIC’s founder, Cathy Rosewell Jonas, told Psychedelic Alpha about the challenging and expensive path to licensure, but also the “incredible” demand she had seen for services. (See Oregon Approves First Psilocybin Service Centre)
As the reality of nested levels of state, county and local policies became clear, not all would-be service centres were as well-positioned as EPIC. In March, we covered Synthesis Institute’s ill-fated bet on a large parcel of land that ultimately saw the company implode.
According to Oregon Psilocybin Services’ Year in Review, by year-end the agency had issued licences to:
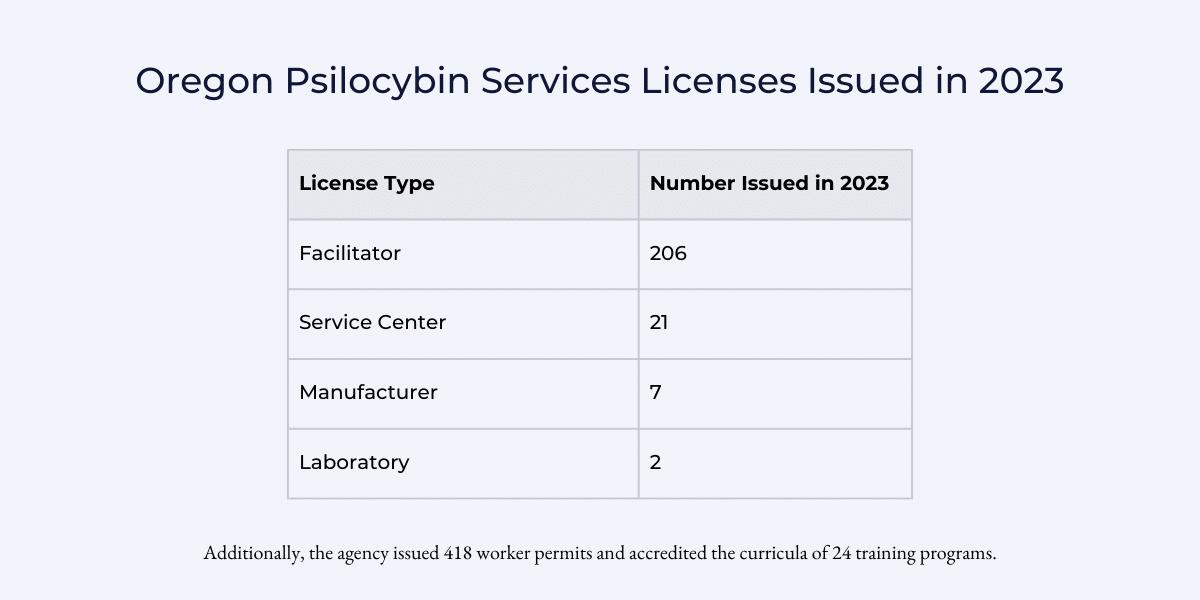
As demonstrated by these numbers, there is a significant over-supply of facilitators who have few places to offer their services. And there are likely many more individuals who have been through facilitator training programs that have not yet applied for, or received, a licence.
During the year, the agency re-entered the rulemaking process to institute “technical fixes and clarifications” to the rules adopted in 2022, as well as rules related to data collection and reporting (to implement SB 303) and amended rules to clarify curriculum approval requirements for facilitator training programs.
Tweaks to the program are expected to continue in 2024.
Colorado Prepares for Natural Medicine Roll-Out
In November 2022, Coloradans voted to pass Proposition 122, the Natural Medicine Health Act. This made Colorado the second state to establish a framework for providing adults access to certain psychedelics. At launch, the program will allow for psilocybin and psilocin, but in time the state will consider adding DMT, ibogaine and mescaline (excluding that which is derived from peyote). The Proposition also decriminalised the possession and use of certain psychedelics.
In terms of statute, SB 23-290 stipulated how the Act would be implemented, and the roles of state agencies like the Department of Revenue and Department of Public Health and Environment. But it left much of the responsibility to recommend rules to the Natural Medicine Advisory Board (NMAB).
During 2023, the NMAB and its subcommittees worked through fundamental and technical questions regarding the program’s implementation. While there is debate among the diverse set of stakeholders, a common goal appears to be improving upon Oregon’s program.
Visit our Colorado Natural Medicine Health Act Tracker and Bulletin, produced with Vicente LLP, for more.
Development will continue this year before the program launches on January 1st, 2025.
Australia Recognises MDMA and Psilocybin as Medicines
In February, Australia’s Therapeutic Goods Administration (TGA) surprised many by announcing a limited rescheduling of MDMA and psilocybin, albeit with plenty of strings attached. (See Australia Recognises MDMA and Psilocybin as Medicines)
In doing so, Australia became the world’s first country to recognise MDMA and psilocybin as prescribable medicines, at least in the Western paradigm of medicine.
But real-world impact appears to be limited due to regulatory, logistical and payor barriers. In fact, Psychedelic Alpha understands that not a single MDMA or psilocybin prescription was filled in 2023, though we await data from the TGA.
Pα: As mentioned in Josh’s reflections on 2023 in psychedelics, it was arguably the year that governments and regulators came to the table in earnest. We saw access to psilocybin debated on the Parliament floor in the UK, Australia enacting a limited rescheduling of MDMA and psilocybin, the European Medicines Agency offering its perspective on psychedelic drug development, and plenty of activity at the local, state and federal level in the U.S. While state-legal markets like Oregon’s got off to a relatively slow start, they appear to have avoided mainstream backlash or high-profile scandals, for now. We should expect these models to be iterated on in 2024, with all eyes on Colorado as the next state to roll-out such a program. ∎
Stay Informed in 2024 with Pα+
This article, which is part of our 2023 Year in Review, is free to read. Subscribe to Pα+ to receive all of our Bulletins, deep dives and analysis, as well as access to the Pα+ Library and other subscriber-only content.
Your subscription goes directly toward supporting our coverage, not glossy graphic design or marketing budgets. Learn more and subscribe here, or get in touch to discuss team plans.

Join thousands of psychedelics insiders
Get the weekly psychedelic medicine briefing.
A free weekly digest covering trials, regulation, policy, and access.
By signing up, you agree to our privacy policy. You can unsubscribe at any time.
- There is an enormous amount of variance between states. LexisNexis cites Minnesota and Missouri’s passage rates as <5%, while Utah and Idaho’s are >60%.
- Psychedelic Science Funders Collaborative President and co-founder Joe Green appeared on the podcast, On with Kara Swisher to discuss psychedelics. During the episode, he discussed his organisation’s approach to psychedelics policy reform initiatives, which is well worth a listen for those looking to better understand this.