
A weekly round-up of the
latest coverage in psychedelics.
Receive the free weekly digest straight to your inbox every week
By signing up, you agree to our privacy policy. You can unsubscribe at any time.
Explore our 🎯 Q1 2026 Psychedelic Pipeline Bullseye Charts.
This week ended with major news for the psychedelic drug development world: the large pharmaceutical company Otsuka is set to acquire Transcend Therapeutics in a transaction valued at up to $1.225 billion, $700 million of which is to be paid upfront. Transcend is developing the MDMA analogue methylone, primarily for PTSD. It is currently initiating a Phase 3 trial that will see patients dosed once weekly for four weeks.
The deal is the latest signal that pharma is willing to run with candidates that have subjective effects that require, in trials at least, close monitoring. The news comes around half a year after AbbVie announced it would acquire Gilgamesh’s bretisilocin, and just weeks after it became known that AtaiBeckley is in discussions around potential partners and/or royalty investors for its lead intranasal 5-MeO-DMT candidate, BPL-003.
Speaking of Gilgamesh, Gilgamesh Pharma, which spun-out of Gilgamesh Pharmaceuticals to house the rest of the company’s pipeline following the AbbVie acquisition, this week announced the close of a $60 million Series A.
In the mainstream media, The Economist published an hour-long podcast about ibogaine, titled The red-state psychedelic, which profiles ibogaine evangelist-in-chief Bryan Hubbard and his efforts to carve out state funds for research into the drug. That effort first started in Kentucky and Ohio, where both states ultimately turned down the idea. It found a home in Texas last year, however, and now Hubbard and co. have national ambitions.
The podcast also revealed that Rick Perry, former Texas Governor and self-proclaimed “right-wing, knuckle-dragging conservative”, is writing a book about ‘psychedelics for Christians’.
Here on Psychedelic Alpha, we published a lengthy interview with Cleveland Clinic staff psychiatrist and psychedelic researcher Dr. Brian Barnett on the key questions around how psychedelic treatments can scale, if approved.
On Friday, after we shared the Otsuka-Transcend news, we also published Bulletin 222, which takes a deep dive into two recently published studies: Germany’s EPIsoDE psilocybin for treatment-resistant depression trial, and a meta-analysis of psychedelics vs. traditional antidepressants that has caused a great deal of discussion over the past couple of weeks. Those were the headline stories, but there’s much, much more inside.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelic business, policy, research and beyond.
Josh Hardman
Founder & Editor

This week, we published my latest Dispatch from Washington, D.C., where conversations I had with those close to agencies, drug developers, and lawmakers suggest that psychedelic approvals remain on track, even as an election year threatens to slow advocacy efforts and ibogaine exceptionalism emerges as a potential fault line.
We also learned that the European Medicines Agency has granted Priority Medicines (PRIME) eligibility to AbbVie’s Bretisilocin for major depressive disorder (MDD), making it the first psychedelic drug to enter the scheme, which is similar to the FDA’s Breakthrough Therapy Designation.
Recent publications paint a murkier picture of psychedelic efficacy, however. A German state-funded 144-patient trial of psilocybin vs. nicotinamide in treatment-resistant depression (TRD) was published in JAMA Psychiatry and features a negative primary endpoint, though the authors note a more favourable slate of secondary outcomes. In the same journal, Balázs Szigeti and colleagues present findings from a meta-analysis of 24 psychedelic trials and conclude that psychedelic-assisted therapy “is no more effective than [traditional antidepressants]”. More on both papers in our next Psychedelic Bulletin.
While almost all of the 106 psychedelics-related bills we’re tracking are looking to in some way catalyse research or access, a new effort introduced in South Carolina this week (S1039) seeks to more clearly criminalise the class by adding “substituted tryptamines” to the state’s list of Schedule I substances.
Elsewhere, we shared an interview with Dr. Charles Nemeroff about Texas’ ibogaine trials and psychedelic research more broadly.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
As you may have noticed, we’re celebrating six years of Psychedelic Alpha with a brand new website and look. As part of that revamp, emails are looking slightly different, too, including your weekly Psychedelic News Feed. Rest assured, the format remains the same: a weekly digest of psychedelics-related coverage from around the world.
At the start of this week, we reported on talks that AtaiBeckley is in with potential pharma partners and royalty investors. Those discussions revolve around its lead candidate, intranasal 5-MeO-DMT (BPL-003) for treatment-resistant depression, which is expected to enter Phase 3 in the second quarter. Sources close to the company told us that it does not intend to sell the asset outright, and hopes to use any fresh capital to explore the drug in other conditions, as well as to progress other candidates in its pipeline.
Later in the week, our 221st Psychedelic Bulletin took a closer look at recent readouts and publications, including Johnson et al.’s psilocybin vs. nicotine patch for smoking cessation study, which published this week. It reports that, at the 6-month follow-up, 40.5% (n=17/42) of those in the psilocybin group achieved prolonged abstinence versus 10% (n=4/40) in the nicotine patch arm.
Elsewhere, UW-Madison’s School of Pharmacy announced a new professorship in psychedelic science, which it describes as “the second of its kind in the United States and the first in a pharmacy school.” Professor Paul Hutson, who led the first Phase I psilocybin trial in the country, is the inaugural recipient.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
There are now around one-hundred psychedelics-related bills in-flight across the U.S., at the state and federal level. Many are making slow progress through their respective legislatures, but others have begun to pass chambers and make it to the desks of Governors. This week, we shared our February 2026 roundup, which provides a one-stop view of new bills introduced last month as well as those that have made substantial progress.
Elsewhere, Bloomberg reported yesterday that AtaiBeckley is exploring partnership or sale options for BPL-003 (intranasal 5-MeO-DMT), its lead candidate that is currently under development for treatment-resistant depression. We will be sharing more on this front next week.
For my part, I have been in New York, Baltimore, and Washington, D.C., this past week, speaking with psychedelic researchers, investors, drug developers, and regulators. Keep an eye out for our next Bulletin to read what I gleaned on this trip.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
I had a great time in New Orleans this week, where I delivered a ‘State of the Psychedelic Industry’ at the Psychedelic Therapeutics and Drug Development Conference and participated in a panel on ‘psychedelic innovation’ at the New Orleans BioInnovation Center.
I’m now in Austin, Texas, where U.S. Health and Human Services (HHS) Secretary RFK Jr. appeared on the Joe Rogan Experience for a two-and-a-half-hour discussion that included around ten minutes on the topic of psychedelics.
“I would say everyone at my agency, as well as over at VA, at Doug Collins’ agency, is very anxious to get a rule out there that will allow these kinds of studies, will allow access in therapeutic settings,” he told Rogan. But Kennedy was broadly noncommittal when discussing how access to psychedelics might manifest in the U.S. medical system.
The Secretary emphasised a need for guidelines to prevent a “Wild West” that could result in “horror stories”. “Some people can have very, very bad experiences on [psychedelics]”, he told Rogan. “We need to move in baby steps with this, because you don’t wanna create a situation where people are getting hurt”, he went on.
He also reiterated his personal interest in the class of drugs, including ibogaine. “I have seen so much overwhelming anecdotal evidence, but also clinical studies, that attest to the effect of this under some circumstances with some people for these medicines”, he told Rogan.
The comments come around eight months after RFK Jr. told Congress that the administration was working to ensure access to psychedelics within a year, though the Secretary appears more restrained than when he made that vow. A more detailed write-up of RFK Jr.’s comments will appear in our next Bulletin.
Elsewhere this week, AtaiBeckley shared a topline readout from its Phase 2a R-MDMA (EMP-01) for social anxiety disorder trial. While the company described the readout as positive, pointing to safety and tolerability findings, shares in the company dropped on the news. Future development plans for the candidate are unclear at present.
Also on the psychedelic drug development front, Reunion Neuroscience shared that FDA has granted its lead candidate, RE104 (4-HO-DiPT), breakthrough therapy designation for postpartum depression.
Here at Psychedelic Alpha, we covered further news stories from the past week in Bulletin 220, including new survey data from RAND that found Americans are divided on psychedelics policy, with 23% open to psilocybin use being legalised.
We also published a discussion I had with Yale’s Dr. Terence Ching about learnings from Yale’s psilocybin for OCD trials.
I’m now heading to New York and Washington, D.C.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
Compass Pathways’ Phase 3 readout dominated this week’s new cycle, as the company shared topline data from the psilocybin for treatment-resistant depression program on Tuesday morning.
It certainly kept us busy! Early on Tuesday morning, we published our analysis of the news, which included discussion with Compass CEO Kabir Nath and CMO Guy Goodwin. Later in the day, we shared our notes from the company’s webcast, during which further data was shared. And, on Friday, we published views from across the field, synthesising what we had heard from researchers, practitioners, and executives.
Elsewhere, two psychedelics trials were published in academic journals this week, providing a more detailed look at methods and outcomes. On Monday, a Phase 2a trial of intravenous DMT for major depressive disorder, carried out by Small Pharma (which has since been acquired by Helus), published in Nature Medicine. Then, on Wednesday, Transcend Therapeutics’ Phase 2 trial of methylone for PTSD published in JAMA Psychiatry.
On Thursday, we shared the latest edition of our Psychedelic Drug Development Pipeline Bullseye Charts, supported by UBC. These visualisations aim to provide a one-stop overview of the pipeline, from discovery through to approval, segmented by molecule and indication.
While our Bullseye Charts show an increase in late-stage trial activity, earlier this week, FDA’s Vinay Prasad and Martin Makary appeared to confirm the agency’s intent to expect just one pivotal trial by default, via a letter in NEJM. We dive deeper in our next Bulletin.
For the next two weeks, Psychedelic Alpha is on the road. I will be speaking at the 6th Annual Psychedelic Therapeutics & Drug Development Conference in New Orleans on Thursday, as well as at an event at the New Orleans BioInnovation Center. I then head to New York and Washington, D.C. If you would like to connect, please do get in touch.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor

BREAKING: Second Positive Phase 3 for Compass’ Psilocybin, Though Modest Magnitude Raises Questions
To dive deeper into the topline readout, we spoke to Compass CEO Kabir Nath and Chief Medical Officer Guy Goodwin.


This week, we published the third Issue of The Psychedelic Practitioner, which focuses on psychedelic dosing sessions or ceremonies. In it, you will hear from practitioners, ethicists, and researchers, who complement our regular programming of practitioner-relevant news and research updates.
Elsewhere, the Oregon Psychedelic Evaluation Nexus (OPEN) announced that it has scored a five-year $3.3 million grant from the National Institute of Drug Abuse (NIDA) to measure the impact of state-legal psychedelic services. It is the first grant of its kind.
Early on in the week, Helus Pharma (formerly Cybin) announced the appointment of a new CEO, Michael Cola, whom we briefly interviewed.
Next week, we launch our Q1 2026 psychedelic pipeline Bullseye Charts, so do keep an eye on your inbox and our socials.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
This week, it became public knowledge that the Trump administration last October vetoed a Commissioner’s National Priority Voucher (CNPV) that FDA had apparently hoped to award to psilocybin drug developer Compass Pathways. STAT broke the story.
As I noted on LinkedIn, the episode appears to be further evidence that MAGA and MAHA remain misaligned on several fronts, with the administration perhaps wary of the optics of accelerating the review of a psychedelic drug further, particularly as the midterms approach.
In our view, the snub is unlikely to materially alter Compass’ timeline. The CNPV aims to cut the review time down to one or two months, the agency has said, but Compass already benefits from numerous regulatory tailwinds, including breakthrough therapy designation and rolling review (it announced the latter last November, when it said it was accelerating its launch timeline by up to a year).
So, there is little reason to believe that FDA’s Division of Psychiatry is resting on its laurels, here. And just as the administration may be sensitive to optics ahead of the midterms, the psychedelic drug development field is wise to be mindful of optics, too. A psychedelic new drug application should (be seen to) succeed on its own merits, with scientific rigour and throgouh review likely to confer greater long-term credibility than the risk of a CNPV review appearing rushed or politicised, particularly as that program becomes increasingly controversial.
Elsewhere, psychedelics featured prominently in the mainstream media this week. The Guardian reported on calls from a former British Army chief for MDMA-assisted therapy to be made available to UK veterans, while Michael Pollan’s new book on consciousness has generated fresh media attention, including a feature interview with The New York Times.
Elsewhere, in our latest Bulletin, we share some observations and updates on America’s psychedelic drug du jour: ibogaine. We also take a closer look at the aforementioned CNPV snub, a Delaware lawsuit that sees Signant Health accuse Definium Therapeutics of trade secret theft, a roundup of the latest psychedelics-related Bills, and much more.
We also covered the latest data from Oregon’s psilocybin system and published an interview with Bruce Damer of the Center for MINDS.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor

Psychedelic Bulletin 219

This week gave us a chance to catch our breaths following a flurry of developments and headlines in the first few weeks of 2026.
Still, there is plenty to review on the psychedelic policy reform front, with psychedelics-related bills introduced or revived in state legislatures across the U.S. At present, our Psychedelic Bill Tracker is tracking 75 bills, and this week we published our January round-up.
There’s activity at the federal level, too, with a new House bill introduced this week that aims to establish a psychedelics research and extended/compassionate access program in the VA.
Here at Psychedelic Alpha, we published an interview with Joost Breeksema on the ‘human side’ of psychedelic therapy, an Op-Ed on psychedelic public health, and our 2026 Psychedelic Alpha Reader Survey.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
The biggest headline in the psychedelics field this week was outgoing New Jersey Governor Phil Murphy’s signing S2283 into law, establishing a $6M psilocybin pilot program in the state. While some outlets hailed this as the fourth state-regulated psilocybin access system in the U.S., it does not create a psilocybin services system like those seen in Oregon and Colorado.
Instead, the legislation creates a two-year pilot program that will see the drug researched at three hospitals in New Jersey, which will each receive $2M, in accordance with FDA and DEA regulations. (Compare that, for example, to Oregon and Colorado, which defy DEA scheduling and are not aligned with FDA.)
The bill did originally envisage the creation of an Oregon-style system featuring service centres and the like. But it was stripped down as it progressed through the state legislature, much to the chagrin of some in the psychedelic policy reform movement.
We may see more state-sponsored psychedelic research bills pass in the coming months, with several introduced already. You can track 55 bills that are in motion in the state and at the federal level via our new Psychedelic Bill Tracker.
While legislators seem increasingly open to psychedelic policy initiatives, Americans remain somewhat divided, according to a new Ipsos/Psychedelic Alpha poll released this week. You can read the topline over at Ipsos and a deeper dive in our latest Bulletin.
Aside from their attitudes, we also got a look at how Americans are using psychedelics and related substances last week, as RAND shared the first cut of data from its 2025 survey. It reports, among other things, that around 10 million U.S. adults microdosed last year.
Elsewhere, our 2025 Year in Review looked at funding, public markets, and M&A activity in the field last year. We reported a relatively strong close to the year in terms of financing activity and a bullish sentiment on the public markets.
Our latest Op-Ed from that same Year in Review series came from Helena Aicher, a researcher and psychotherapist working within Switzerland’s framework for limited medical use. There, Aicher reflects on how Europe’s psychedelics landscape evolved in 2025.
It certainly seems like the pace of developments is not letting up, with plenty to cover on the policy reform side. Pα+ subscribers should keep an eye out for our first roundup of 2026 on that front.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
Pα+ Psychedelic Bill Tracker
Monitor the status of every psychedelics-related bill working its way through both state and federal legislatures in the U.S. with our new Psychedelic Bill Tracker.
Psychedelic Bulletin 218


The psychedelics field’s busy start to 2026 continued this week.
On Monday, MindMed unveiled a full overhaul of its brand, including a new name: Definium Therapeutics. In doing so, it became the second Phase 3 psychedelic drug developer to rebrand this year, after Cybin became Helus Pharma last week.
The companies are presumably hoping to distance themselves from their ‘shroom stocks’ vintage, with names like MindMed and Cybin perhaps indelibly associated with a 2021 ‘shroom boom’ that was driven in part by frothy markets and retail investor excitement. That era was replete with the likes of Kevin O’Leary, r/wallstreetbets analyses and, in the case of Cybin, some questionable stock promotion efforts.
Definium says its name is a combination of the Latin ‘definio’ and ‘infinitum’, literally meaning ‘to bound’ and ‘infinite’, respectively. Perhaps it reflects an ambition to sound equal parts transformative and nondescript, limitless but in a limited, safe kind of way.
Speaking of safety, U.S. National Institute on Drug Abuse (NIDA) Director Nora Volkow said last year that ibogaine is unlikely to ever receive approval for opioid addiction due to its cardiac toxicity, throwing cold water on a surge of interest in the drug that revved up in 2025 and continues to mount today.
But this week, Volkow shared an article on ‘Nora’s Blog’, which lives on the NIDA website, that asks: “Could psychedelics harness neuroplasticity to treat addiction and other mental illness?”
In it, the NIDA Director concludes that “[h]arnessing the brain’s natural plasticity in effecting therapeutic gains is an intuitively obvious avenue for drug development including the development of psychedelics as therapeutics”, adding that basic research might deliver value beyond new treatments, too. “The profoundly meaningful experiences some people report following use of psychedelics could give neuroscientists valuable insights into meaning-making and the ability of the brain to change in a healthy direction after addiction or trauma”, she went on.
Under Volkow, NIDA has funded trials on psilocybin for smoking cessation, psilocybin and ketamine in the treatment of opioid use disorder, and ketamine for stimulant use disorder; so it’s not as though Volkow has blocked such work via the Institute. But it is curious that the Director chose to pen this particular blog entry, which serves as a call for further research into psychedelics as therapeutics and provides a sketch of that research agenda, now.
Also on the research and drug development front, Reunion Neuroscience announced this week that it expects to conduct just one Phase 3 trial to support the approval of its 4-OH-DiPT candidate in postpartum depression. That comes after FDA signalled a shift toward expecting just one Phase 3 by default late last year. We shouldn’t expect every psychedelic drug developer to follow this path, however, with factors like target indications and a drug’s profile coming into play.
On the state front, New Jersey could fund psilocybin research via S2283, which passed the Senate earlier this week. The Act would establish a psilocybin research pilot program and fund it to the tune of $6M. It has now passed both chambers with convincing margins and sits on Governor Phil Murphy’s (D) desk awaiting a signature.
After a busy start to the week, the second half of the work week saw flashy headlines announce that former Senator Kyrsten Sinema is the subject of a lawsuit filed by the ex-wife of one of her staffers, Matthew Ammel. In a complaint that has now been transferred to federal court, Ammel’s ex-wife accuses Sinema of breaking up their marriage.
While, on the face of it, the story has little to do with psychedelics, the suit alleges that Sinema, a licensed clinical social worker, asked Ammel to bring MDMA on a work trip, where she apparently intended to “guide him through a psychedelic experience”.
Indeed, Sinema is an outspoken psychedelics advocate, particularly on the topic of ibogaine. She became interested in advocating for ibogaine, she told me last summer, after Ammel went to Mexico to take the drug and found it beneficial. But, as we have covered in these pages, some in the field have been keen to distance themselves from Sinema, as the former Democrat-turned-Independent politician was already controversial. Some tell me that she inserted herself as a spokesperson for the field, while in other cases, it does appear that she has been welcomed in, especially in some corners of the ibogaine advocacy push.
More: Last summer, I had a somewhat spiky interview with Sinema, who attended Psychedelic Science in Denver with Ammel and a colleague from Hogan Lovells. Later, in a Bulletin, I asked: “What is Kyrsten Sinema Selling?”
Ammel is facing legal troubles, too, after he allegedly threatened his rental property manager last November and was involuntarily committed to a hospital. There, he is accused of attempting to strangle a member of the hospital staff. He was taken to jail on felony charges and released on a $10,000 bond, with a hearing scheduled for the end of January.
It appears that Ammel has been in Gabon, the Central African country, this week taking part in the International Conference on Iboga and Ibogaine, which was co-organised by Americans for Ibogaine.
“Gabon is the ancestral home of iboga”, a statement by Americans for Ibogaine CEO Bryan Hubbard following the closing of the conference reads, “and we deeply value the conversations grounded in cultural respect, environmental stewardship, and science.”
It’s not clear that Sinema shares that view. When I was sitting with Sinema, Ammel, and their law firm colleague in Denver last summer, I asked the former Senator about her thoughts on the role of traditional knowledge-holders like certain groups in Gabon. The Indigenous perspective, she told me bluntly, “is not my problem”.
In that same interview, Sinema told me: “So, because you’re British and you don’t know me, one thing I will tell you is, I am well known for having no drama. I do not engage in drama.” But it certainly looks like the drama has found its way to Sinema, who is facing a fresh bout of media attention following the surfacing of the lawsuit. In Salt Lake City on Friday, hecklers disrupted Sinema’s conversation with Utah Governor Spencer Cox about… disruption.
Tabloids aside, here at Psychedelic Alpha, our 2025 Year in Review shifts to focusing on perspectives from across the field. To that end, we published a Views from the Field piece where several experts share what they’re excited and concerned about for 2026, and an open question they hope to get more clarity on.
Elsewhere, we published an Op-Ed from Healing Advocacy Fund Executive Director Taylor West on ‘what state psychedelics programs are teaching us’, and a Guest Article by Monica Schweickle on how Australia’s ‘psychedelic experiment’ is panning out.
UBC also published a brief interview I did with them before the holidays. We spoke about why I founded Psychedelic Alpha nearly six years ago, how we aim to act as a counterbalance to both stigma and hype, what I’m expecting to see in psychedelics this year, and more.
This was a slightly longer Editor’s note than usual. If you enjoyed it and would like much, much more analysis and news, consider subscribing to Pα+ to receive all our content, including our flagship Bulletins.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
As expected, 2026 is off to a busy start, with the first full week of the new year delivering no shortage of headlines.
On the drug development front, FDA finally accepted GH Research’s inhaled 5-MeO-DMT Investigational New Drug (IND) application after placing it on hold for more than two years, clearing the way for a Phase 3 program that the company hopes to launch this year. AtaiBeckley, meanwhile, expects to launch its own Phase 3 5-MeO-DMT program in Q2, with its intranasal formulation. Both companies are targeting treatment-resistant depression.
FDA also accepted an IND from Compass Pathways, allowing it to progress with a Phase 2b/3 program studying its psilocybin candidate in PTSD. It hopes to begin screening patients in the coming months.
Elsewhere, Cybin officially rebranded to Helus Pharma and listed on the Nasdaq this week, ringing the bell on Monday morning. And Gilgamesh Pharma printed positive topline data from a Phase 2a study of its NMDA receptor antagonist, GM-1020, in major depressive disorder. It’s another win for the company, which sold its lead candidate, bretisilocin, to AbbVie last year in a deal valued at up to $1.2bn.
Here on Psychedelic Alpha, we discussed all of these updates, and much more, in our first Bulletin of 2026. As a reminder, our flagship Psychedelic Bulletins provide one-stop coverage of developments across the psychedelics field. Published multiple times a month, they synthesise news, data, and analysis into a coherent picture of how the field is evolving, helping readers maintain continuity and perspective over time. They’re exclusively for Pα+ members: subscribe today.
We also shared the third and final session from our 2025 Year in Review video series, where I walk you through what happened in 2025 and where the field stands today; what we might expect to see in 2026; and, some potential futures and broader open questions the field faces. Pα+ subscribers can now watch all of the videos in the series.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor

Psychedelic Bulletin 217

Part 3: Future(s) & Open Questions
🎥 Out now: Part 3 of our 3-part 2025 Year in Review video Series
Happy New Year!
As we closed out the year, we highlighted the ten stories that defined psychedelic drug development, policy, and access in 2025. You can read those 10 standout stories now: we lifted the Pα+ paywall for all of the articles mentioned in our rundown for the month of January.
As 2026 gets underway in earnest, our 2025 Year in Review content begins to adopt a more forward-looking tone. This week, we published the second video in a three-part series where I look ahead to 2026 and consider potential FDA approvals, Phase 3 readouts, alternative pathways to access, and much more. Pα+ subscribers can watch the video now.
Things were relatively quiet in mainstream media this week. I hope that you, too, found a little bit of quiet ahead of what looks set to be a very busy year.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor

Part 2: Looking Ahead to Psychedelics in 2026
🎥 Out now: Part 2 of our 3-part 2025 Year in Review video Series
Our Founder & Editor, Josh Hardman, looks ahead to 2026 via a narrated presentation. In this video, Hardman considers potential FDA approvals, Phase 3 readouts, alternative pathways to access, and more. Watch now on our website.

2025 Year in Review: 10 Standout Stories in Psychedelics
We have lifted the paywall on the Pα+ stories featured in this piece for the month of January.
This week, we published our last Bulletin of 2025, which featured interviews, policy and drug development updates, and a closer look at some recent research findings. Our attention now turns to planning for what is sure to be an eventful 2026, but not before we share our 2025 Year in Review.
This year, we’re doing things a little differently. While we will still share a couple of articles from our desk, a selection of global dispatches from guest authors, and Michael Haichin’s Interdisciplinary Annotated Psychedelic Research Bibliography, the core of our 2025 Year in Review coverage is in the form of a three-part narrated presentation that I have pulled together. In it, I share data and updates from around the world, as well as deep dives into specific topics, such as what I learned during a trip to D.C.
The first session, which looks back at 2025 and takes stock of where the field is today, with a primary focus on drug development and policy, will be shared early next week with our Pα+ subscribers. Join today to get access to the videos as they publish.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor

Psychedelic Bulletin 216
As the holiday period approaches, we at Psychedelic Alpha are busy focusing on the launch of several new features and pieces of content to round out 2025 and welcome the new year.
There’s little rest among psychedelics advocates, too. Outside of the traditional model of pharmaceutical approvals, where a regulator like the FDA gives a nod to new drugs, some countries and states continue to pursue psychedelic access pathways with a focus on launching next year.
In Europe, tweaks to Czechia’s criminal code will allow for medical access to psilocybin in 2026, for certain patients in specific contexts, as guidelines for the use of the drug have now been drafted by the country’s psychiatric society.
In the U.S., meanwhile, New Mexico’s Medical Psilocybin Advisory Board has brought forward plans for the launch of its state-regulated program by a year. It now hopes to allow the first patients to access psilocybin in December 2026. That’s around the same time that a pharmaceutical form of psilocybin could be up for FDA approval. It’s certainly going to be an interesting year.
At the federal level, a rumoured executive order to reclassify marijuana has been making headlines. If the supposedly imminent move materialises, psychedelics advocates will be watching closely for how MAGA reacts.
And, back at Psychedelic Alpha, we were pleased to publish the second Issue of The Psychedelic Practitioner earlier this week, which focuses on the topic of Psychedelic Preparation. The next Issue, which will be published in February, will focus on Dosing.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
This week, I shared my Dispatch from D.C., which reflects on my time speaking with lawyers, lobbyists, and advocates, but also agency staffers, drug developers, and well-connected individuals in an attempt to take the temperature on psychedelics among those who might shape the field’s future.
There is significant tumult not only across U.S. politics and the regulatory apparatus, but also within the psychedelics field itself, which does not appear to have landed on a clear or coordinated ‘ask’. But beneath the noise, there appeared to be a quiet confidence among some that multiple FDA approvals of psychedelics are close at hand, with as many as three possible over the next 18 months.
The day after that piece was published, STAT reported that the FDA will only expect one Phase 3 trial before considering a new drug for approval. While many drugs already receive approval on the basis of one pivotal study, psychiatric drugs are often put through two or more Phase 3 trials. In our latest Bulletin, I cover what this could mean for the psychedelics field.
We also cover the Freedom to Heal Act, which was introduced this week by Senators Cory Booker (D) and Rand Paul (R). It aims to ‘fix’ what its backers view as a ‘gap’ in federal Right to Try law that effectively excludes Schedule I substances, including psychedelics. Advocates are flying in to D.C. early next week to drum up support.
Outside of D.C., longevity enthusiast Bryan Johnson’s live-streamed ingestion of psilocybin mushrooms last Sunday generated buzz early in the week. While some applauded the online event as a laudable move to reduce stigma, others poked fun at the spectacle.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor

Psychedelic Bulletin 215
It’s been a very quiet week in the psychedelics field, largely owing to the U.S. holiday.
We’re taking this moment to prepare for our 2025 Year in Review, and this time we’re looking to you, our audience, to contribute. Find out more and pitch a contribution. (The deadline for submissions is Friday 5th.)
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
I just returned from a trip to Washington, D.C., where I found myself immersed in a notably optimistic psychedelics clique, with many expecting several significant developments in the next 18 months.
One of those developments has already come to fruition: ARPA-H’s $100M initiative to develop objective measures of mental and behavioural health. The initiative, which is one of the agency’s largest ever, looks set to feature psychedelic research substantially. (We were the first to cover that program’s reveal last Friday.)
There is plenty more coming down the pike, I am told, with 2026 set to be an incredibly busy year for the field. I will be writing up some of my field notes (a kind of ‘View from D.C.’) in the coming weeks, exclusively for our Pα+ subscribers.
Elsewhere this week, the largest trial of LSD microdosing to date found that it underperformed placebo across all endpoints in a population of 89 patients with major depressive disorder. The null finding is the latest blow to drug developers seeking to prove the efficacy of microdosing, and it becomes the second mid-stage low-dose LSD program to flop.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor

Psychedelic Bulletin 214
I had a great time speaking at the UW-Madison Psychedelic Symposium this week. It’s my second time attending this event, which I appreciate for its remarkably interdisciplinary agenda: there was everything from preclinical studies of psychoplastogens (Katherine Nautiyal) through to the history of touch in psychedelic practice (Zoë Dubus).
For my part, I provided a kind of ‘State of the Union’, presenting on the current state of psychedelic drug development, industry and policy, before considering some potential future directions and open questions. I was also glad to participate in a lively panel discussion. Besides being among my favourite meetings in the field, not least due to the Midwestern hospitality of the hosts and organisers, I was also grateful to have an excuse to spend some time updating my slides and adding some new ones. I hope to share an annotated copy with our Pα+ subscribers soon.
Psychedelics were also on the agenda at the ‘MAHA Summit’ in DC, a closed-door affair with a line-up including Vice President JD Vance and Health and Human Services Secretary RFK Jr. Psychedelics were discussed on stage by HHS lawyer and “psychedelics czar” Matt Zorn, who chatted with AtaiBeckley founder and chairman Christian Angermayer.
Speaking of the HHS Secretary, a new book by journalist Olivia Nuzzi, who says she had a close relationship with RFK Jr., claims that he told her he uses psychedelics and has smoked DMT.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor

We take a deep dive into the readout, which we discussed with AtaiBeckley CEO, Srinivas Rao. ∎
This week got off to a busy start with Compass Pathways announcing on Tuesday morning that it is bringing forward its launch planning by 9-12 months, following a positive meeting with FDA and the completion of enrolment in its second, larger Phase 3 study. The psilocybin developer is expected to be the first to secure FDA approval for the drug, which could come as early as late next year or early 2027.
That same day, we also learned, via an SEC filing, that AbbVie paid $900 million upfront for Gilgamesh Pharmaceuticals’ lead psychedelic candidate, bretisilocin. Until then, there had been no public disclosure of how much of the acquisition deal, which is valued at up to $1.2bn, was paid in cash. The answer, we now know, is 75%: well above what many expected. Some investors and operators in the field view this as evidence of AbbVie’s conviction around the candidate.
Also in the psychedelic drug development world, the merging of atai Life Sciences and Beckley Psytech closed this week, with the former now trading as AtaiBeckley.
Beyond the psychedelics industry, Global Psychedelic Week debuted with online and in-person programming across the world. I was pleased to participate by moderating a panel on funding, as well as having a discussion with psychedelics researcher Robin Carhart-Harris.
I am heading to Wisconsin next week for the UW-Madison Psychedelic Symposium, so please cross your fingers and toes for minimal flight disruptions! I had a great time last year and am looking forward to delivering the final presentation of the meeting on Friday afternoon. Thereafter, I have a short stint on the East Coast. I am excited to again meet with some of our readers in person.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor

Breaking: Compass Pathways Accelerates Psilocybin Launch Timeline by 9-12 Months
We take a deep dive into the news, which we discussed with Compass Pathways CEO, Kabir Nath. ∎

Psychedelic Bulletin 213
It’s been an incredibly active start to the fourth and final quarter of 2025.
In October, funding for psychedelics companies appears to have caught up with the positive sentiment exhibited in our Q3 Psychedelic Investor Survey. More than half a billion dollars was raised across three rounds alone last month (by atai, Cybin, and MindMed), meaning Q4 2025 will see the largest inflows to the space since mid-2021. There are still two months left of the quarter, meaning the heady days of 2021 could still be topped.
On the lobbying front, too, spend is ramping up. Our Psychedelics Federal Lobbying Tracker shows that psychedelic drug developers spent more than ever on U.S. federal lobbying in Q3, though the aggregate reported spend remains relatively modest at just north of $300k over the three-month period. Interestingly, Lykos Therapeutics has ceased its formal spend on federal lobbying, while Compass Pathways has more than doubled its own. What’s more, the Association for Prescription Psychedelics (which represents most of the late-stage developers in the field) began spending cash on lobbying activities in Q3, according to filings. (Pα+ subscribers can read our analysis of the latest data.)
On Friday, the Australian Government’s Department of Veterans’ Affairs announced that it has decided to fund psychedelic assisted therapy for veterans who meet certain criteria. It’s the latest in a string of psychedelics-related headlines from down under, which we will be covering in more detail very soon.
In the 212th Issue of our Psychedelic Bulletin, we looked at several other stories, including Delix Therapeutics’ efficacy signal from a small open-label study of its lead neuroplastogen and NRx Pharmaceuticals’ efforts to have FDA ban its competitors’ ketamine products.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
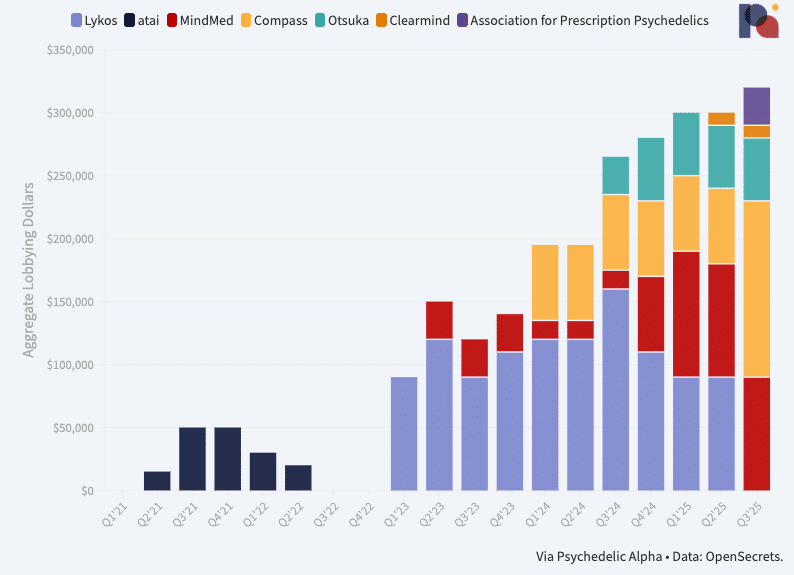
New: Psychedelics Federal Lobbying Tracker
Here, we share several charts that visualise the extent of psychedelics organisations’ lobbying activities at the federal level, at least according to filings. ∎


Psychedelic Bulletin 212
This week was a fine example of how surging optimism among many in the psychedelics field exists alongside calls for a tempering of excitement.
Our analysis of the Q3 2025 Psychedelic Investor Survey revealed record levels of optimism and positive sentiment, yet actual financing data from the same quarter showed a drop in allocation to the field. Taken together, this suggests that funding is lagging sentiment.
But, last week, atai Life Sciences closed a $150M public financing on the back of its receipt of FDA breakthrough therapy status for BPL-003, Beckley Psytech’s intranasal 5-MeO-DMT candidate for treatment-resistant depression. (atai is in the process of acquiring Beckley.) Might this be the beginning of a more active fundraising environment to end the year?
Elsewhere, at least two recent studies have delivered null findings. A study of the longitudinal effects of psilocybin truffle microdosing found no enhancement in self-reported mood or cognition. And, a 62-participant study that saw participants receive IV ketamine or midazolam twice-weekly for four weeks found no significant difference in outcomes between the two groups. (We covered both in our most recent Bulletin.) The latter study, in particular, has sparked plenty of discussion among researchers.
In the mainstream media, BBC Future published an interesting look at the impact of Erowid, “the 30-year-old drug website that transformed psychedelic research”. Women’s Health, meanwhile, explored “why more midlife women are turning to psychedelics to heal their minds”.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor

Psychedelic Bulletin 211

Psychedelic Patent Update: September 2025
Plus: We look at a patent dispute that is brewing around AbbVie’s $1.2bn psychedelic candidate, bretisilocin.
It’s been another busy week in the psychedelics field!
I’m currently in Japan, where psychedelic research is beginning to find its footing. Earlier this week, I met with Dr. Hiroyuki Uchida, a psychiatrist who is leading Asia’s first modern clinical trial of psilocybin therapy for treatment-resistant depression (TRD). I interviewed Dr. Uchida earlier this year (we published it in both English and Japanese), so it was a real pleasure to meet him in person at Keio University Hospital, a sprawling complex in the country’s capital.
I was struck by just how much work it takes to get a psychedelic trial started in a country like Japan. In many Western countries, psychedelic studies are quickly becoming part of the furniture, with entities like Institutional Review Boards increasingly familiar and comfortable with them. But Dr. Uchida and co. were, in many regards, starting from scratch when they set out to launch a study a few years ago. Aside from securing buy-in from the necessary stakeholders, Uchida’s group also had to translate scales like the mystical experience questionnaire (MEQ) and get the study drug into Japan, a country with notoriously strict drug laws. But he persevered, and today the trial has nearly finished dosing the twelve patients it aimed to enrol. I will share some more brief thoughts on Japan’s psychedelic research scene in our next Bulletin.
Outside of Japan, plenty has happened this week, including:
We will cover all this, and more, in Bulletin 211 next week, which is exclusively for our Pα+ subscribers.
Here on Psychedelic Alpha, we published a discussion I had with trauma and memory researcher Samuli Kangaslampi, as well as the Q3 update to our Psychedelic Financing Tracker, plus a summary of the latest funding developments for our Pα+ subscribers.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
It was a very busy week here at Psychedelic Alpha. On Thursday, we launched The Psychedelic Practitioner, a new publication designed specifically for therapists, clinicians, facilitators, and other professionals working with—or preparing to work with—psychedelic therapies and related treatments. Issue 1 is now available to read, and we welcome your feedback via the survey.
Friday was World Mental Health Day, which saw media outlets across the world spotlighting psychedelics’ potential in this realm. I was very pleased that my interview with journalist Dominique Nora was featured in French magazine Le Nouvel Obs’ Friday special. There, I discussed how the psychedelics field has changed in the past half-decade, what the pipeline looks like today, the challenges that psychedelic studies and rollout are facing, or might face, and a brief outlook on the future of the field.
Elsewhere, CBS News featured our U.S. psychedelic laws tracker in a segment on “The rise of psychedelic-assisted therapy for mental health treatment”, which saw Dr. Rachel Yehuda interviewed by the morning show’s anchors.
But the week ended on a sombre note, as we learned that Dr. Nolan Williams had died by suicide. Williams was a Stanford researcher whose work reshaped modern brain stimulation treatments for depression and suicidality. More recently, he became a prominent voice in the psychedelics research field, especially on the ibogaine front. This shocking news underscores the enormity of the struggle he fought to tackle through his own work. But those close to Williams have already expressed that his vision and compassion will continue to shape the field he redefined. Yesterday, psychiatrist Owen Scott Muir, who described Williams as both a friend and a hero, published a eulogy.
Below, you will find your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
If you’re a podcast or radio fan, it’s a great time to be interested in psychedelics. Right now, you can listen to season 2 of the Altered States podcast, catch an episode on the evidence base for psychedelic medicine over at Undark, and enjoy a BBC Radio 4 segment on psychedelics for mental health hosted by Chris and Xand van Tulleken.
If you prefer to consume your media with your eyeballs, psychedelic scientist Manoj Doss and colleagues published a feature on ‘what we know, and what we think we know’ regarding how psychedelics affect the brain in The Scientist. Elsewhere, we published our latest Bulletin, which included our Dispatch from the Borealis Psychedelic Science Summit as well as updates from across the field.
On Wednesday, we unveiled our latest resource: The Psychedelic Perceptions Tracker. We combed through opinion polls, surveys, government reports, and other datasets to produce a curated series of headline figures, charts, and data visualisations on how various groups perceive and interact with psychedelics.
This coming week, we again have something new to share: The Psychedelic Practitioner, a publication designed specifically for therapists, clinicians, facilitators, and other professionals working with—or preparing to work with—psychedelic therapies and psychedelic-based treatments. Keep an eye out in your inbox for that first Issue next week.
Anyhow, without further ado: Here’s your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
Psychedelic Bulletin #210
I write this short note from Stockholm, Sweden, where the fantastic Borealis Summit just wrapped.
I gave a talk that provided an update of sorts on the psychedelic drug development pipeline as well as policy developments from around the world. It was among the driest topics, truth be told, with others exploring themes as rich as consciousness and meaning-making, challenging experiences, and gender and psychedelics.
As a Brit myself, I am very pleased to have reason to attend events and cover stories much closer to home, with Europe catching up with its North American counterparts in terms of psychedelic policy reform efforts, government-funded research, and so on. The continent, and the Nordics, are making their mark on the so-called psychedelic renaissance.
Elsewhere this week, we published a very long Issue of the Psychedelic Bulletin, which covers a whole host of topics: from atai Life Sciences’ $11.4M NIDA grant and Reunion Neuroscience’s $133M upsized Series A financing to a payor-backed psilocybin pilot program in Australia and real-world outcomes data from Oregon’s psilocybin services.
In the press, a New York Times feature has caused quite a stir. It details the story of venture capitalist Amy Griffin, whose immensely popular book, “The Tell”, recounts her story of recovering what she describes as a memory of sexual assault by a teacher during her childhood. The apparent memory was recovered, Griffin says, during an underground MDMA therapy experience. (Griffin was connected to the MDMA therapists via MAPS founder Rick Doblin. Her husband is a MAPS donor, and the pair invested in Lykos Therapeutics via their foundation.) The Times piece probes Griffin’s account and presents several questions that the book, and its popularity, raise. The case also draws focus on questions around psychedelic-induced recovered memories, which is quite a sensitive matter. During my time in Stockholm, I spoke to an expert on this topic; the interview will be published soon.
Anyhow, without further ado: Here’s your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
Psychedelic Bulletin #209
Welcome to the hundreds of new subscribers who have joined us after exploring the latest versions of our signature Psychedelic Drug Development Tracker visualisations, which were published last week.
The timing of those fresh Bullseye Charts was, in hindsight, quite opportune, as it was a relatively quiet week… for psychedelics, at least.
Other than a few media mentions, the only real news that broke was the UK Royal College of Psychiatrists’ new position paper and guidance booklet on the use of ‘psychedelic and related substances’, as they choose to refer to them. We covered both publications when they were made available on Friday morning.
Early next week, we will publish another bumper Issue of the Psychedelic Bulletin and a look at MindMed’s Phase 2b study of LSD for generalised anxiety disorder. Subscribe to Pα+ today to make sure you receive all of our Bulletins and articles. (More info / join here; or, reply to this email to discuss group/corporate plans.)
Anyhow, without further ado: Here’s your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
This week has been a little quieter, as the dust began to settle following the FDA’s publication of the Complete Response Letter it sent to Lykos Therapeutics last August, when it declined to approve its MDMA for PTSD new drug application. On Monday, we published a deep dive into the CRL’s contents, reactions from the field, and what it might mean for Lykos’ path to approval.
On Thursday, we shared a conversation I had with former EMA scientific expert Dr. Florence Butlen-Ducuing, where we discussed Europe’s psychedelic renaissance.
Elsewhere, both the LA Times and Wired ran stories on how athletes are turning to psychedelics, including ibogaine, to address brain injuries. And, STAT reports that psychedelics “are suddenly drawing interest from big drugmakers”, in a story that I was pleased to appear in.
Anyhow, without further ado: Here’s your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
This week looked set to be much quieter, with no real news to speak of for the first half, aside from Cybin’s ousting of its CEO, Doug Drysdale.
Then, on Thursday, our eagle-eyed Medical Advisor, Michael Haichin, noticed the FDA had released 89 previously unpublished complete response letters (CRLs) that it has issued to sponsors since 2024.
Among them was the letter it sent to Lykos Therapeutics last August, when it rejected its MDMA for PTSD new drug application. We broke the news on Thursday morning and will share a little more detail with our Pα+ subscribers on Monday.
Given that we were anticipating a quieter week (read: we tempted fate!), we used Bulletin 208 to look back at two conversations that took place at Psychedelic Science in June. One was an on-stage discussion between psychedelic drug development execs, while the other was a chat that I had with APA Division 56 President Jessica Punzo.
Anyhow, without further ado: Here’s your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
Psychedelic Bulletin #208
Gosh, what a busy week! Monday saw two breaking news stories: AbbVie acquires Gilgamesh Pharmaceuticals’ lead psychedelic candidate in a transaction valued at up to $1.2 billion, and Norwegian regulators approved the use of public funds to cover generic ketamine for treatment-resistant depression. While the first of those two stories signals a growing appetite from ‘big pharma’ to dabble with true psychedelics, the latter story has upset some pharmaceutical firms, especially Johnson & Johnson (more on that here).
Later in the week, we learned that Lykos Therapeutics is renaming itself Resilient Pharmaceuticals, presumably in a nod to the fact that, despite decades of work and last summer’s FDA rejection, the MDMA drug developer keeps pushing on.
On Thursday and Friday, I took part in the Hopkins-Oxford Psychedelics Ethics (HOPE) workshop at The Oxford Union, which was a fantastic, intimate gathering of scholars and scientists (and, well, me!). I look forward to keeping tabs on HOPE’s future outputs.
Anyhow, without further ado: Here’s your Psychedelic News Feed, a one-stop digest for the latest coverage of psychedelics business, policy, research and beyond.
Josh Hardman
Founder & Editor
Psychedelic Bulletin #207
Participate: Microdosing Self-Blinding Study
Imperial College London is seeking participants for a self-blinding microdosing study.
Event: Borealis Psychedelic Science Summit
Join our Editor Josh Hardman in Stockholm next month.
Event: Psychedelic Therapy: From Evidence to Equity
On the 1st of October, the Champalimaud Foundation in Lisbon, Portugal, will host a one-day symposium.
Psychedelic Bulletin #206
Psychedelic Alpha’s Josh Hardman spoke with Forbes about the program and what it could mean for Europe more broadly.
Psychedelic Bulletin #205
BREAKING: Germany Establishes EU’s First Psilocybin Compassionate Access Program
Psychedelic Bulletin #204

Psychedelic Bulletin #203
Participate: Post-Psychedelic Challenges Study
If you have experienced difficulties after taking a psychedelic, you may be eligible to participate in this study from Emory University.
Q1 2025: Oregon Psilocybin Services Tracker
As the first cut of data from Oregon’s psilocybin program becomes available, we take a deep dive into emerging trends through various data visualisations and analysis. This marks the first instalment of our new Oregon Psilocybin Services Tracker, which will produce analysis of each cut of data as it is released.
Psychedelic Bulletin #202
Psychedelic Bulletin #201
Psychedelic Bulletin #200

BREAKING: atai Moves to Acquire Beckley, If Phase 2b 5-MeO-DMT Data Delivers
We spoke with sources close to the companies to learn more about the proposed merger and financing.
The Backyard Psychedelic Renaissance: How Bottom-Up Change Is Delivering Psychedelic Access
In this Guest Article, policy analyst Kody Zalewski calls for a greater focus on local reforms as a key driving force behind future reforms over larger geographies, and foregrounds some of the voices who are joining him in that call.
Pα+ Psychedelic Bulletin #199
REMEMBERING AMANDA FEILDING
Lady Amanda Feilding, a pioneer funder of psychedelic and consciousness research, passed away last week.
“[W]e are deeply grateful for Amanda’s contributions and remain committed to carrying forward her mission of bringing the potential benefits of psychedelic-based treatments into mainstream medical practice.”
“Her legacy is one of courage, curiosity, and an unshakable belief in the power of love over fear.”
Pα+ Psychedelic Bulletin #197

Pα+ Psychedelic Bulletin #196

Pα+ Psychedelic Bulletin #195

Pα+ Psychedelic Bulletin #194
Pα+ Psychedelic Bulletin #193

Psychedelic Policy Update: Medical Access, Industry Influence, and Voter Suppression Shape 2025 Landscape

Transcend Therapeutics has shared topline results from its Phase 2 study of the MDMA analog methylone (TSND-201) in PTSD. While the readout is certainly positive, questions remain about the intervention’s scalability, specifics around the protocol, and the company’s IP moat. Read more…
See also a Research Briefing on the article.

Yesterday, Representatives Lou Correa (D) and Jack Bergman (R) introduced the Innovative Therapies Centers of Excellence Act. The bipartisan bill—co-sponsored by Reps. Morgan Luttrell (R), Ro Khanna (D), and Dan Crenshaw (R)—would see the VA designate at least five such centres, which would focus on researching and delivering certain psychedelic therapies. Here, we speak to two of the bill’s architects…
Pα+ Psychedelic Bulletin #192
Pα+ Psychedelic Bulletin #198



Pα+ Psychedelic Bulletin #191
See our Interview with Maj. Aaron Wolfgang, Chief of Inpatient Psychiatry at the US Army, for more.
The latest report from BrainFutures presents a healthcare provider survey on opportunities and barriers to the roll-out of psychedelic therapies.
Read or listen to Part 1 here.

Pα+ Psychedelic Bulletin #190

A short, simple guide to CO’s regulated psychedelics program’s key rules and structure by Tasia Poinsatte.
“Topline data from the core, randomised, 8-week study are expected in mid-2025.”
See also: Psychedelic Science’s press release with further names, including Psychedelic Alpha’s Editor Josh Hardman.

PSFC has publicly released its roadmap for the future of psychedelic philanthropy. We originally covered the report in December: SCOOP: PSFC’s $130M Roadmap to Shape the Future of Psychedelic Philanthropy.
“The scholarship partnership opens financial support for any interested undergraduate for the BA psychedelic studies minor.”

Opinions: PrEP’s Rollout Offers a Roadmap for Prescription Psychedelics
Hailey Gilmore, MPH explores how PrEP’s rollout offers key learnings for the medicalization of psychedelics, particularly in overcoming stigma, building provider awareness, and integrating new treatments into existing healthcare structures.
Psychedelic Bulletin 188


An LA Times short documentary will premiere on February 18th. It follows two first responders who travel to Mexico to undergo psychedelic therapy in the hopes it will help their PTSD and depression.
Pα+ Psychedelic Bulletin #187
See also an event, Cambridge Psychedelic Research Day, March 12th, 2025.
Johns Hopkins opens the first clinic of its kind in the U.S. which aims to support individuals experiencing difficulties following a psychedelic experience.
Q1’25 Bullseye: The Psychedelic Drug Development Pipeline
Explore the latest edition of our Bullseye Chart, which displays psychedelic candidates by phase of development and molecule type.
Updated: The Psychedelic Drug Development Tracker
We have also updated the Tracker webpage, where you can explore psychedelic candidates by stage of development.

2025’s Psychedelic Policy Surge: A State-by-State, Bill-by-Bill Analysis
Here, we parse through the influx of new bills. First, Psychedelic Alpha Editor Josh Hardman provides a high-level thematic overview of the fresh set of bills. Then, Jack Gorsline runs through the bills one by one.
With Shayla Love of the Atlantic, Josh Hardman of Psychedelic Alpha, Mattha Busby of Vice and Rolling Stone, and Jack Gorsline.
Beckley’s open-label study (n=12) of its intranasal 5-MeO-DMT candidate in AUD reports 50% of participants completely abstinent at 3 months.
Ft. Psychedelic Alpha Editor-at-Large Graham Pechenik.
Pα+ Psychedelic Bulletin 186

HELP WANTED
Psychedelic Alpha is looking for someone to help us share our content on social media and other platforms. The right person is familiar with the psychedelics field, is able to summarise our content and pull out key themes or quotes, and has graphic design skills. If you are interested in this position, please contact hello@psychedelicalpha.com with one or two sentences about yourself and any samples of your work.
Pα+ Psychedelic Bulletin #185
This latest twist in the Lykos Therapeutics saga threatens a major overhaul of the company’s strategy and direction, with Musk ally and billionaire investor Antonio Gracias appearing to want to take the company back toward its nonprofit drug development roots and a more Doblinite philosophy. Now, the company’s board must decide whether to stick with its current lead investor, Helena, or accept Gracias’ proposal.
Pα+ Psychedelic Bulletin #184
Our Editor Josh Hardman will appear on a panel alongside journalists Shayla Love and Mattha Busby to discuss their experiences reporting on the psychedelics renaissance. Hosted by Jules Evans.
Pα+ Psychedelic Bulletin #183
“The main theme of the companies it wants to build? No tripping.”

Bullish on Chaos: Matt Zorn on How Psychedelics Could Benefit from Trump’s Second Term
After years of challenging the U.S. government over its drug laws and their application, lawyer Matt Zorn is bullish about the potential regulatory openings that an incoming Trump administration might present. Here, he discusses the types of ‘deeper questions’ we might ask about the U.S. system of drug and medicines regulation and how we might begin to reimagine it.

Interview: Inside the FDA’s Psychedelics Journey: Javier Muniz, MD on Breakthroughs, Challenges, and the Future of the Field
Psychedelic Alpha’s Josh Hardman speaks with Javier Muniz, MD, who recently left his post as Associate Director at the FDA’s Division of Psychiatry. In that role, Muniz was at the heart of the agency’s psychedelics learning journey and helped formulate its view on thorny topics in psychedelic drug development like functional unblinding and the role of psychotherapy. This interview is far-reaching and provides a unique look into both the mind of the regulator, but also Muniz’s own views on psychedelics, now that he is unencumbered by his position in the agency.

The memorial will take place on Thursday, December 19, from 5:30-8:00 pm. More info and RSVP.
The trailer for a Netflix documentary which follows NFL star Aaron Rodgers has been released, with the film set to drop on December 17th. It prominently features his use of ayahuasca.

Interviews: Meet the Team Who Challenged the DEA on DOI and DOC
Dr. Alaina M. Jaster profiles the scientists, advocates and lawyers challenging the agency’s proposed scheduling of two phenethylamine psychedelics.
Founding members include Compass Pathways, B. More Inc., Lykos Therapeutics and MindMed. The group’s Scientific Advisors include Peter Hendricks, Paul Hutson, Sandeep Nayak and Jennifer Jones.
PsyDAO, a decentralised autonomous organisation that aims to fund psychedelics research and art, has raised around $2m following its initial mint of PSY tokens.
The company reports 12-mo efficacy data from a Phase 2 study of its deuterated psilocin candidate, CYB003, in major depressive disorder (MDD). The results are impressive, with 100% of patients responders and 71% in remission at the 12-month mark. The sample is small, however, with just 8 participants represented in the cut of data, 7 of which received two 16 mg doses (the remaining participant received just one dose). The company announced the commencement of its Phase 3 program last week.

Jack Gorsline shares the latest view from Massachusetts, where psychedelics advocates and detractors are hoping to sway voters to mark a ‘yes’ or ‘no’ next to Question 4 next week. The Yes campaign feels that coverage by The Harvard Crimson was unfair, a key anti-Question 4 campaigner appears to have misrepresented his group’s potential participation in a debate, the No campaign receives its first major donation, and more…

This morning, Compass Pathways announced delays to both of its Phase 3 studies, as well as a major reduction in force that sees its preclinical programs and digital tools effectively shelved. Here, we provide our analysis along with coverage of the company’s earnings call and associated discussion.

We speak to Carey Turnbull and Michael Bogenschutz about an ambitious new study.

We share the first in a series of dispatches from Jack Gorsline, a journalist who is quite literally ‘on the ground’ in the heart of this election season’s psychedelic policy reform battleground: Massachusetts.
BioSpace speaks to three experts, including Cybin CEO Doug Drysdale, about Mass. Question 4.
TOOLS FOR TEACHERS
Our friends at BrainFutures have published a toolkit for academic leaders who are interested in integrating psychedelic coursework into higher education.
A new named professorship supports Deepak Cyril D’Souza’s research on the potential for psychedelic drugs to treat post-traumatic stress disorder and depression.
UCLA’s Ecological Medicine & Psychedelic Studies Initiative is hosting a one-day hybrid event that aims to bridge mental health, Indigenous perspectives, legal rights, city planning, ethnobotany, conservation, ecology, and psychedelic therapies… gosh! Free to attend.
Vicente LLP looks at big-picture questions as the program hurtles towards launch. First up are fees and rule changes, before the firm reviews Natural Medicine-related meetings since our last Bulletin in late August.
Shayla Love explores how “new, non-hallucinogenic versions of psychedelics are blurring the boundaries of the drug trip. (* Included here as it just missed the cut-off for last week’s Feed.)
SEE YOU THERE?
Psychedelic Alpha is pleased to be a sponsor of this year’s UW Madison Psychedelic Symposium. We hope to see you there.
Next year’s track includes around twenty psychedelics-related panels or presentations, including a panel discussion titled ‘Behind the Scenes of the “Psychedelic Renaissance”‘ that sees our Editor Josh Hardman joined by The Microdose’s Jane Hu, The Atlantic’s Shayla Love, and Calyx Law’s Graham Pechenik (who is also an Editor-at-Large here at Psychedelic Alpha).
Our friends at PsyMed Ventures have teamed up with KdT Ventures to offer a virtual workshop for scientists who want to build a neuroscience startup.
If you’ve had personal experience with classic psychedelics—such as LSD, psilocybin, ayahuasca, DMT, or mescaline—you are invited to take part. The survey takes approximately 30 minutes to complete and is best experienced on a computer or in landscape mode on a mobile device.
Bessel van der Kolk encourages Massachusetts voters to get approve the psychedelics question this November.
Lykos Therapeutics has provided an update on a recent meeting with the FDA re: its MDMA for PTSD New Drug Application. The very brief 100-word statement reveals that the ‘path forward’ for the program will include an additional Phase 3 trial, but could also entail an independent, third-party review of the two existing Phase 3 studies (MAPP1 and MAPP2).
Host Regina G. Barber talks with Rosalyn LaPier about ethnobotany–what it is and how traditional plant knowledge is frequently misunderstood in the era of COVID and psychedelics.
“To develop the new drugs, Castrén and his coworkers have established a company called Kasvu Therapeutics. He has received financial support from University of Helsinki Funds, which has also helped him develop the company.”

THE FIGHT FOR QUESTION 4 IN MASSACHUSETTS
As the U.S. elections draw closer coverage of Massachusetts’ Question 4 is heating up, too. If voters endorse the ballot question the state would be tasked with decriminalising certain naturally-occurring psychedelics and establishing a regulated program similar to Oregon’s. But polling shows that the vote is currently too close to call, with donors pouring last-minute funding into the Yes campaign (See Bulletin 176 for more).
We spoke with Massachusetts Psychiatry Society president Nassir Ghaemi about why his group is opposing Question 4, as well as his broader views on psychedelics.
The Boston Globe’s editorial board shares their reasons for opposing the initiative.
The Globe also ran more of an explainer piece, which includes the testimony of a retired school teacher who found psilocybin to be beneficial to her trauma and depression.
The Herald speaks to the initiative’s supporters.
Some local councils in Mass. are taking positions on the Question, too, or at least hosting hearings.
CIIS to launch the first-ever undergraduate Bachelor of Science in Psychedelic Studies offering, planned to begin in fall 2025.
This free, hybrid (in-person and virtual) conference will feature multidisciplinary discussions from the likes of Eduardo Schenberg, Katherine Hendy, Claudia Schwartz and Franklin King.
Ft. Mindstate Design Labs, Beckley Psytech, atai Life Sciences, MindMed, Gilgamesh Pharmaceuticals, Enveric Biosciences.
Earlier this week, Psychedelic Industry UK (PsyIndUK) announced its launch. The new association says that it aims to represent the country’s ‘psychedelic medicine and wellness sector’, with founding members including Heroic Hearts Project UK, Onaya, and Imperial College London’s Centre for Psychedelic Research.
Robin Carhart-Harris; Christian Angermayer; Ekaterina Malievskaia and George Goldsmith; Srinivas Rao; Rick Doblin; Matthew Baggott; David Nutt; Robert Barrow; Doug Drysdale; Michael Mullette.
A final decision on fees for healing centres, cultivators, and manufacturers has been pushed back, but Colorado remains on track to begin accepting license applications at year-end. A public hearing will take place on October 21st at 1-3:30pm Mountain Time via Zoom.
While Janssen’s Spravato is on-track to become a blockbuster drug, access to the esketamine nasal spray product continues to be a key Acchiles heel for patients and their providers. Here, through study of two recent publications and interviews with their authors, we look at variability in access to, and real-world usage of, the therapy that psychedelic drug developers are increasingly hoping to imitate.
Cochrane Library publishes its first systematic review of psychedelic-assisted therapy, which focuses on its uses in mental health challenges among people with life-threatening diseases. Read the plain English summary here.
A small nonprofit clinic in Waynesville, North Carolina has been part of a national movement to broaden the use of MDMA to treat post-traumatic stress disorder.

Exclusive: David Hough on Lykos’ Path to Resubmission
Josh sits down with the man drafted in by Lykos Therapeutics to plot a path to resubmitting its MDMA application. They talk about Hough’s experience in developing Spravato to eventual approval and how that might inform any future clinical development of MDMA, Hough’s thoughts about the FDA’s verdict and the AdComm that preceded it, what a future Phase 3 program might look like, and more…
“Drug Science and UCL are undertaking novel research using a bold alternative approach to understanding how MDMA works. The researchers aim to begin recruiting study participants in 2025.”
Our Editor, Josh Hardman, spoke to Labiotech about how Lykos’ FDA rejection impacts the broader field of psychedelic drug developers.
WaPo covers Mindstate Design Labs’ news that FDA and EMA have green-lit a Netherlands-based Phase I study of its 5-MeO-MiPT-based candidate.
Psyence scoops up fellow psilocybin drug developer for just $500k upfront (paid in shares, no less). Clairvoyant, which is currently trialling a psilocybin-based candidate for alcohol use disorder in a Phase 2 study, also has the chance to earn two $250k milestones as part of the deal… but this is still a punishingly low valuation for a company that raised $3M in the heady days of 2021.
Around $10M in funding is available via the Defense Medical Research and Development Program (DMRDP). Application intent deadline is 20th September.
A must-read for anyone considering operating a Natural Medicine Business, or becoming a Facilitator, in Colorado’s state-regulated psychedelics program.

Endpoints puts some tough questions to Lykos CEO Amy Emerson.
In a fiery interview, Jonathan Lubecky goes toe-to-toe with former Psymposia Managing Editor David Nickles.

Josh delves into FDA’s appeal process with regard to Lykos Therapeutics’ development program, and covers the trio of journal article retractions that have cast another shadow over the company’s published studies. Plus: We assess the fallout beyond Lykos, as psychedelic drug developers wrestle over the role of psychotherapy in their protocols, fast followers assess their strategies, and would-be psychedelic therapy delivery companies face a substantial delay.
A prosecutor says five people have been charged in connection with Matthew Perry’s death from a ketamine overdose last year, including the actor’s assistant and two doctors.
If you have 3-5 lifetime psychedelic experiences, minimal meditation experience, and can commit to a 21-day prep course with visits to UCL over a 4-week period, researchers would like to hear from you.

FDA has issued a Complete Response Letter (CRL) to Lykos Therapeutics regarding its New Drug Application (NDA) for MDMA-assisted therapy in PTSD, meaning the agency is not prepared to approve it at this time.
Read our breaking coverage for more.

With a target PDUFA date of August 11th, FDA is expected to make a decision on Lykos Therapeutics’ MDMA-assisted therapy for PTSD New Drug Application (NDA) at some point this week.
As we brace for a deluge of mainstream media coverage and insider reactions, Josh takes the opportunity to provide a brief assortment of notes and thoughts.
The story has been widely covered in major outlets, including:
The journal Psychopharmacology has issued three retraction notices for MAPS/Lykos’ meta-analysis of phase 2 studies of MDMA-assisted therapy for PTSD, which constituted the rationale for a Phase 3 program. The first retraction pertains to the pooled analysis of the half-dozen phase 2 studies (Mithoefer et al., 2019), the second relates to the long-term follow-up of those six trials (Jerome et al., 2020), and the third applies to an analysis of four of the phase 2 studies to explore the effect of tapering antidepressants (Feduccia et al., 2020).
Prop. 122, which called for the creation of a regulated natural medicine (psychedelics) system in Colorado, didn’t permit localities to opt-out, unlike Oregon’s Measure 109. But, locals are finding a way by setting time, place and manner restrictions.
J&J has filed a supplemental NDA (sNDA) with FDA, Seeking Approval of Spravato as a monotherapy for Treatment-Resistant Depression. If approved, the esketamine nasal spray would become the first monotherapy for adults with the depressive disorder.
See also DEA’s Move To Ban Two Psychedelics Is Challenged As A ‘Disservice To Science’ In Legal Filing From Student Group (July 24) ↗ Marijuana Moment.
Coverage of Siegel et al.’s new study, Psilocybin desynchronizes the human brain, published in Nature on Wednesday. The paper was widely covered elsewhere, including in The New York Times.
“…the top deck of the Semel Institute is being reimagined as a state-of-the-art living laboratory to study Ecological Medicine and psychedelic therapies. It will be the first of its kind in a biomedical setting.”
See the CDC’s study of such products here.
Vicente LLP walk us through more than a dozen hearings, meetings and (proposed) rule updates since our last Bulletin. As we head into a month that features final public hearings on a number of matters, now is the time to make your voice heard.
The BBC looks at what happened to Awakn’s Bristol clinic. TL;DR: There weren’t enough patients able or willing to afford the out-of-pocket costs for treatment courses.
In this Issue:
Pα+ Psychedelic Bulletin #165:
and more…
RAND has published a 161-page report on the state of non-medical psychedelic use in an effort to inform policymakers in the U.S. and beyond. Psychedelic Alpha is pleased to be cited as a source of data and analysis in the piece.
“The plaintiffs are challenging OHA’s “failure to ensure” that the psilocybin program doesn’t discriminate against physically disabled individuals.”

“Canada now joins Switzerland as one of the few countries where cluster headache patients have been explicitly authorised to use psychedelics outside of clinical trials”, said the Organisation for the Prevention of Intense Suffering (OPIS).
“We are going to have to regulate ourselves, and our professional organizations have a role to play in developing guidelines to help clinicians,” Paul Appelbaum said.

“Greg and Matias interview Josh Hardman, founder and editor of Psychedelic Alpha, to discuss the recent FDA advisory committee meeting on Lykos Therapeutics’ MDMA-assisted therapy for PTSD.”
Laurel Kilgour’s 6-week online summer course via Stanford Continuing Studies returns to provide an overview of topics like psychedelic patents, monopolisation risks, ethical implications of commercialising plant medicine, and so on.


Pα+ Psychedelic Bulletin #164:
“The prevalence of post-traumatic stress disorder among college students rose to 7.5 percent in 2022, more than double the rate five years earlier, researchers found.”
MDMA-AT COVERAGE RAMPS UP AHEAD OF FDA ADCOMM
A variety of mainstream outlets are running stories on Lykos Therapeutics’ upcoming FDA advisory committee, slated for next Tuesday.
Pα+ Psychedelic Patent Analysis: April 2024
This month:

“The staff of Lucid News is deeply saddened by the death on Sunday of our friend and colleague Ken Jordan. In addition to being our Editorial Director and co-founder of Lucid News, Ken was a true pioneer and leader in the psychedelic movement and a dear friend to many of us. He had a clear vision of the future of psychedelic communities and dedicated his life to the transformative power of psychedelics for worldwide healing.”
We at Psychedelic Alpha share in this deep sadness. If you would like to honour Ken’s legacy, please consider donating to Lucid News.

MAPS (the nonprofit) issued a statement in response.
“A psychoactive brew prepared from vines by the people of the western Amazon basin, ayahuasca is seen, depending on the version, as a miracle cure, a tool for inner exploration and personal development, a recreational hallucinogen, or a dangerous psychotropic drug.”


Pα+: FDA Advisory Committee to Review Lykos Therapeutics’ MDMA-Assisted Therapy in Early June
Next month, the first psychedelic-assisted therapy will be put before an FDA Advisory Committee as Lykos Therapeutics’ MDMA-AT gets a June 4th slot.

“Psychedelic-assisted therapies have been hailed as the wave of the future. They’re also becoming big business. What if most people can’t afford them?”

“I am in favor of legalization of all drugs, primarily psychedelics. If it takes having psychedelics legalized for therapeutic use, then I’m all in for that also.”
“The idea that all of a sudden, wow, instead of having to take a pill every day, a single dose of psilocybin or MDMA could have this profound effect on a chronic illness—it’s pretty cool,” said former NIMH Director Thomas Insel.


See NCT06309277 for more.
“I feel incredibly lucky to see the change that I’ve been working for happen in my own lifetime,” Doblin says. “I can die happy, whenever that comes around.”
“The [Australian] federal government will spend $3.8 million on the trial involving about 200 survivors of the record-breaking floods that hit the region in 2022.”
“Ernesto Londoño’s memoir ‘Trippy’ explores psychiatry’s renewed flirtation with drugs such as LSD. He’s a believer but not a zealot.”
For more, read Josh’s interview with Londoño.
“A rare condition called hallucinogen persisting perception disorder has puzzled researchers and raised alarms as psychedelics go mainstream.”
“The stepped care treatments in this clinical trial will include an arts-based group compassion program and group-based MDMA-assisted therapy. It is hoped the findings will provide additional treatment options for those impacted by future disasters.”
“Psychedelics are included, a field that Nelsen said ARCH has an interest in. He said that there’s more scientific rigor needed to really dig into how psychedelics work but that the mechanisms of action are intriguing.”
BrainFutures’ latest report “focuses on the critical intersection between the Mental Health Parity and Addiction Equity Act (MHPAEA) and psychedelic-assisted therapy (PAT), offering insights into how this law can help pave the way for comprehensive insurance coverage and access to PAT for those who stand to benefit.”
“She was an enthusiastic supporter of the counterculture. And when she suggested that her brothers rent Mr. Leary a mansion, she made psychedelic history.”

“By pursuing FDA approval of MDMA-assisted therapy first, we believe the learnings will be beneficial as we determine our regulatory strategy in the United Kingdom and Europe”, said Lykos CEO Amy Emerson.
“Hibadulin said his condition impeded his relationships and education, and that the only treatment that proved effective for him was psychedelic-assisted psychotherapy – a controversial method to cure PTSD illegal in Ukraine but used to a limited extent in some Western countries.”

Insights from the European Medicines Agency’s Psychedelics Workshop: A Pα+ Dispatch
Josh explores some of the key themes discussed at the workshop: from regulatory remits and rescheduling through to disentangling pharmacotherapy from psychotherapy, standardisation and its discontents, blinding, and much more.
“CEC is the first non-Christian church to receive protection for its spiritual practices regarding Ayahuasca.”
“Readers respond to a letter which said that MDMA is not helpful in mental health care”
“We respectfully request that you produce VA’s implementation plan that would be deployable as soon as this novel treatment option is approved for clinical use”, the letter reads.
Two new Rapid Response briefings from the UK Parliament’s POST look at the state of the evidence for psychedelics’ use in treating PTSD and eating disorders.

“A lobbying effort led in part by religious groups has state lawmakers considering two different measures that would legalize psilocybin, a drug known as “magic mushrooms.””
GUEST WRITERS
Are you interested in writing for Psychedelic Alpha? Please reach out to hello@psychedelicalpha.com with a short blurb about yourself, topics you might be interested in writing about, and any published work samples.
This six-part series will see a new article published each weekday through next Tuesday.
“[Jules] Evans doesn’t advocate against psychedelics. But by exposing the hazards that tripping could involve, Evans hopes to temper the prevalent ethos of breezy optimism with a dash of caution.”
“From kava to “sleepy girl mocktails,” can anything ever take the place of booze?”

Josh sits down with Jaskaran (‘Jaz’) Singh, Vice President Psychiatry Development at Neurocrine Biosciences. For the purposes of this conversation, however, we focus on Singh’s role in spearheading the development of esketamine for treatment-resistant depression, now marketed as Spravato.
Here, we discuss how Singh’s early work assessing ketamine’s antidepressant effects at the National Institute of Mental Health (NIMH) encouraged him to move into industry; how his team decided to pursue esketamine; lessons learned that might be relevant for psychedelic drug developers; and his broader outlook on the field.
“Arguably the buzziest area of research are psychedelic compounds. Long dismissed because of thorny legal and scientific issues, psychedelics are being taken more seriously as possible treatments for anxiety, depression and psychosis.”
The company’s pipeline includes SPT-348, “a prodrug of a non-hallucinogenic neuroplastogen in development for the treatment of mood and other neuropsychiatric disorders, [which] leverages Glyph to create a potential first-in-class treatment with improved pharmacokinetics and tolerability compared to conventional psychedelics.”
Jane C. Hu asks: Will legal psychedelics be any better?
Hu also appeared on Slate’s ‘TBD’ podcast on Sunday in an episode titled, Is America Ready for Legal Psychedelics?
A day-long LSD-assisted cycle event, Bicycle Day, will replace the ill-fated KAP discipline.
After two rounds of redundancies, a crack team of McKinsey consultants has made a somewhat unorthodox recommendation to federal agencies: LSD-assisted bureaucracy. A pilot program will be carried out at DMV offices.
Initial reports describe the experience, though lacking psychedelics’ characteristic subjective effects, as “heavy” and “somewhat unstable”, but “noticeably patentable”.
The company announced the claims cover its lead candidate, Polymorph Eau.
Fresh from a trip to Beijing, Yellen shares some of her favourite magical meals.
Fresh from a trip to the continent, Pollan takes on the final frontier of ineffability: mime.
The complaint alleges repeated failures to both capitalise and append ® to SPRAVATO®.
Sling darts at your favourite psychedelic drug candidates!
All over America, youths have been heard uttering “nar” with their mates on the way to brekkie at Macca’s.
In March, MindMed scored an FDA Breakthrough Therapy Designation for its LSD-D-tartrate candidate (MM120) for generalised anxiety disorder (GAD). In this piece, Josh takes a closer look at the company’s claim to have ‘eliminated’ psychotherapy from its Phase 2b study of MM120 and explores how a broader ‘Spravato-isation’ of psychedelic therapies is stoking debate around the contribution of psychotherapy (or lack thereof) in these interventions.
“Charlotte Nichols, who has PTSD, says there is enough evidence to prescribe psilocybin therapy as mental health treatment”
Vicente LLP reviews the March 15th Colorado Natural Medicine Advisory Board meeting, where draft rules for Facilitator licensing and training were discussed.
Owing to some bizarre headlines from across the psychedelics space, we have compiled a special edition of our Psychedelic News Feed.
FEATURED PSYCHEDELIC JOBS
Explore more roles and join our Talent Network: ↗ Psychedelic Alpha Job Board
FREE EVENT
Read more about the symposium, at which our Editor-at-Large Graham Pechenik will be speaking, via this American University article. Free registration is available.


We review ICER’s draft evidence report on Lykos Therapeutics’ MDMA-AP, which finds that the publicly-available clinical evidence on the intervention is “insufficient” to inform a comparative clinical effectiveness analysis. The report also highlights clinical and ethical concerns surrounding the studies.
According to the press release, a single dose of BPL-003 “demonstrated a rapid and durable antidepressant effect in TRD patients, with 45% of patients in clinical remission at week 12”.
It’s worth noting that the initial topline results represent 12 participants.
Beckley Psytech also published its own press release.
Tickets are priced at £20 / $25. Get 15% off, using code PsychedelicAlphaPD15 here.

Visit ClinicalTrials.gov for more information on the protocol (NCT06308653).

An excerpt from Molds, Mushrooms, and Medicines: Our Lifelong Relationship with Fungi by Nicholas P. Money.


POLITICO covers Mindstate Design Labs’ IND submission for a Phase I study of 5-MeO-MiPT.
“The remarks from Nora Volkow, the longtime director of the National Institute on Drug Abuse, serve as a cautionary note amid widespread enthusiasm about ibogaine, a naturally occurring substance that drug companies and researchers have increasingly cast as a potential paradigm-shifting addiction treatment.”

We have been tracking psychedelic policy reform across the United States for a number of years now. Visit the updated map today to see the latest in your state.
New Scientist also produced a video on the topic (15 mins).
Mindstate has submitted an IND to the FDA to study 5-MeO-MiPT, aka moxy, co-administered with an approved generic. The Phase I trial aims to dose five cohorts of ten healthy volunteers.
Psychedelic drug developer MindMed has announced that the FDA has granted breakthrough therapy designation to its LSD-D-tartrate candidate (MM120) for the treatment of generalised anxiety disorder (GAD). The company also shared topline 12-week data from the same program, as well as results of a pharmacokinetic bridging study and further details on phase 3 study design.
Pα+ subscribers can read our coverage here.
One of the at-home ketamine industry’s biggest boosters has turned bearish, calling for caution among patients and providers and withdrawing his endorsement of the practice. Here, we take a closer look at the case study that changed Cappello’s mind as well as broader concerns around at-home ketamine operators, and we speak with Cappello himself.
Readers might remember that atai shared topline results from a Phase 1 study of the candidate back in October 2023, finding the oral transmucosal film (OTF) formulation to be “well-tolerated with a favorable safety profile”. However, the company announced it would “further optimize” the candidate in preparation for a Phase 2 study in treatment-resistant depression.
As such, this Phase 1b study will compare an improved OTF formulation of the candidate with IV. The improvements include taste masking, a backing layer, “and enhancements to further increase permeability.”
For reference, atai dosed the very first subject with VLS-01 in Q4 2022.
“At a business licence hearing Tuesday, a three-person panel approved a motion asking staff to reinstate the licence with terms that clarify the business is involved in education and advocacy of medicinal psychoactive substances.”
“We opened the door now, just a crack, but it’s only going to get bigger”, said Dana Larsen.
On March 7th, Vancouver Sun columnist Dan Fumano wrote a piece titled, “Dan Fumano: Psychedelics are illegal — and Vancouver police say criminal charges could be coming”.
CALL FOR SURVEY PARTICIPANTS
Do you have experience conducting psychedelic-assisted therapy outside of the research setting? You may be eligible to participate in a new study from Imperial College London’s Centre for Psychedelic Research. Learn more and participate.
As the first set of draft rules for Colorado’s regulated psychedelics market emerge, the folks at Vicente LLP have penned two explainers:
The Phase 2a study, which saw 73 GAD patients assigned to receive psilocybin-assisted therapy or placebo with psychotherapy, achieved its primary endpoint.
The sponsor, Incannex, is now preparing to test the intervention in a Phase 2b study. More coverage is available in Bulletin 157.
The study is featured in a new SBS documentary, too.
Separately, Incannex’s Chief Scientific Officer, Mark Bleackley, spoke with Pharmacy Times about psilocybin’s potential.
In ketamine therapy and other contexts, dissociation is seen as an unwanted side-effect. But what if there’s more to it?
The loose coalition of ibogaine funding advocates—led by former Kentucky Opioid Abatement Advisory Commission Chairman Bryan Hubbard—has landed on a new state to house their project: Ohio.
In this article, we explore the magnitude of opioid settlement funds available in Ohio; the funding mechanisms we expect ibogaine advocates to tap in support of their renewed initiative; why advocates believe Ohio makes sense as a potential champion of this work; and, the challenges ahead for such an ambitious project.
PSYCHEDELIC BULLETIN #156: Canada’s First Published Psilocybin Trial; University of Maryland Joins Terran’s Complaint Against Compass
Pα+ PSYCHEDELIC PATENT ANALYSIS A Mebufotenin Melee (Dec ’23 & Jan ’24)

Two weeks ago, the Reagan-Udall Foundation for the FDA hosted a two-day meeting, Advancing Psychedelic Clinical Study Design. It offered great insight into how academics, industry representatives, and—crucially—the FDA are viewing psychedelic drug development programs.
In this piece, Josh shares some key themes, analysis and take aways.
The Day 2 Dispatch is now available, too. It covers Set and Setting, FDA’s regulatory authority, and real-world use of psychedelics.
As Colorado’s Natural Medicine Advisory Board (NMAB) officially adopts an initial set of 123 recommendations, with many having dozens of sub-parts, Vicente LLP review the January 2024 meetings of the NMAB and its subcommittees.
Vicente LLP reviews the 123 recommendations adopted by the Natural Medicine Advisory Board, which will inform the first round of rule-making for the state’s regulated psychedelics program.
A PhD candidate at CIIS is looking for English-speaking adults who have had 2+ psilocybin experiences, at least one of which being a ‘highly meaningful experience, to participate in a survey. More info.
Featured content in this Issue:

MAPS Public Benefit Corporation (PBC) has submitted its New Drug Application (NDA) to the FDA in order to seek marketing approval of MDMA-assisted therapy (MDMA-AT) for PTSD.
The organisation and its parent non-profit, MAPS, has been conducting clinical studies of MDMA-AT for over twenty years, making this a significant milestone.
And it’s not just a milestone for MAPS: this is the first psychedelic-assisted therapy submitted to the FDA for approval.
“The filing of our NDA is the culmination of more than 30 years of clinical research, advocacy, collaboration and dedication to bring a potential new option to adults living with PTSD,” said MAPS PBC’s CEO, Amy Emerson.
“If approved, MDMA-assisted therapy would be the first psychedelic-assisted therapy, which we hope will drive additional investment into new research in mental health”, she continued.
More Coverage:
“The measure would allow supervised clinical studies with active duty members. Psychedelic treatments for PTSD have found conservative supporters in the state for years.”
The agency withdrew its earlier effort to do so in August 2022, but noted it would be back with an amended procedure.
“Perhaps this withdrawal is the product of a procedural quirk as opposed to a change in attitude from the agency”, we wrote in Bulletin 116 back in August 2022.
It certainly seems so.
Comments close on January 12th, 2024.
One has to wonder whether proposing this rule over the holidays might reduce the likelihood of significant public and industry engagement, as seen in DEA’s first attempt to schedule these substances.
“Use of ecstasy and nitrous oxide falls overall and drug use of all kinds falls among younger people”.
“More than 100 companies are focused on psychoactive drugs, according to patent attorney Graham Pechenik of the San Francisco-based Calyx Law. Five years ago, only a few dozen patent applications had been submitted for psychoactive-related products, he said. Now his Psychedelic Alpha patent tracker counts more than 1,000.”
“The state is considering funding research on ibogaine, a psychedelic that’s growing in popularity as potential treatment for opioid use disorder.”
“Griffiths lived a remarkable and consequential life. As one young physician scientist said upon hearing of his death: he changed the world for the better.”
“Trial met its primary endpoint with MM-120 demonstrating a statistically significant dose-dependent improvement in HAM-A scores four weeks after a single-dose”.
Delix’s ibogaine analog, DLX-007, will benefit from NIDA funding. “DLX-007 has completed IND-enabling studies and is slated for clinical testing in 2024.”
“Bottom line: if you are a medical professional flirting with these new systems, be careful. Certainly, dial down blasé echoing from the purveyors of the medical marijuana establishment. Get a robust and unbiased legal opinion. Don’t let someone shoot from the hip.”
“Under the plan brokered by Gov. Kotek, a Democrat, state lawmakers would be asked to consider a ban on public drug use and police would be given greater resources to deter the distribution of drugs.”
“The scientists plan to spend another year or so slipping the VR headset on patients dosed with psilocybin to learn what settings might dial up the awe of a psychedelic experience.”
Clinical Leader interviews Enveric Biosciences CEO, Joseph Tucker.
OPEN ACCESS BOOK
Expanding Mindscapes: A Global History of Psychedelics
Erika Dyck and Chris Elcock’s edited volume is available via Open Access.
“The scale of a $42 million investment in ibogaine research raises serious concerns, especially considering that Kentucky has only spent $8 million on evidence-based practices that are proven to work.”
Attempted murder charges against a former pilot have been dropped. Instead, he now faces 83 reckless endangerment charges.
The off-duty pilot had consumed psilocybin mushrooms around 48 hours prior to attempting to cut the engines off during an Alaska Airlines flight.
“The attempted murder charges were never appropriate in this case because Captain Emerson never intended to hurt another person or put anyone at risk – he just wanted to return home to his wife and children,” his defense lawyers Ethan Levi, Noah Horst and Norah Van Dusen said in a statement. “Simply put: Captain Emerson thought he was in a dream.”
“The release conditions include that Emerson undergo mental health services, stay away from drugs and alcohol, and not come within 30 feet (9 meters) of an operable aircraft.”
Written by Graham Pechenik and Rachel Mount.
EVENT RECAP
reMind’s Psychedelics Business Forum
We had a great time in Las Vegas at reMind’s second Psychedelics Business Forum, which brought together a diverse crowd to discuss the present and future of the psychedelics industry.
I (Josh) really enjoyed the focus on sharing practical tips for operators and the focus on what entrepreneurs and practitioners can do right now, or in the near future. This was a nice change from my usual beat, which is often focused on things like drug development that have long timelines.
It was great to moderate a couple of panels, too. I spoke with Courtney Barnes and Sam Chapman about psychedelic policy reforms, as well as Nykol Bailey Rice and Chris Walden about ketamine clinics. Having written quite a bit about both of these topics over the past few years, it was refreshing to hear from those with intimate, first-hand experience.

Having attented many psychedelic conferences in the past few years, I appreciated reMind’s focus on making plenty of space for networking in the program. Providing this scaffolding and drop-in networking space was especially helpful for people like me who simultaenously suffer from networking FOMO and networking burnout at conferences.

This Compass Pathways-funded study evaluated a single dose of psilocybin with psychotherapy in 15 individuals with bipolar II depression.
“…most participants met remission criteria on the Montgomery-Åsberg Depression Rating Scale 3 weeks after a single 25-mg psilocybin dose, and most remained in remission 12 weeks postdose with no increase in mania/hypomania symptoms or suicidality.”
“The findings suggest efficacy and safety of psilocybin in bipolar II depression and support further study of psychedelics in this population.”
“DLX-001 Shown to Be Safe and Well-Tolerated Without Producing Hallucinogenic, Psychomimetic, or Dissociative Effects Based on Interim Data from Phase 1 Trial”
The $107,227 early career research grant was awarded to Amanda Tamman at Baylor College of Medicine.
“…those reporting recent cryptomarket use were more likely to report adverse LSD events…”
VIDEOS
ALPS Conference 2023
Recordings of talks delivered at ALPS 2023 are now available on YouTube.
Shayla Love explores the curious intersection between the psychedelics and longevity movements, which was highlighted at this year’s Wonderland Miami conference.
She said her experience with psilocybin “was nice”.
“Bipartisan and bicameral congressional lawmakers have reached an agreement on a large-scale defense bill that contains a House GOP-led section to fund studies into the therapeutic use of psychedelics such as psilocybin and MDMA for military service members.”

This Issue of the Bulletin primarily focuses on psychedelic drug development and business stories, with around 4,000 words of coverage and analysis.
Featured content in this Issue:
LECTURE
Health Economics & Psychedelics, with Elliot Marseille & Jennifer Mitchell
“Recruiting for a CYB003 Phase 3 study is anticipated to begin by the end of Q1 2024.”
“Single IV doses of GM-2505, a novel 5HT2A receptor agonist, demonstrated psychedelic effects lasting 60-90 minutes and were well-tolerated with no serious or severe adverse events.”
UPCOMING EVENTS
reMind Psychedelics Business Forum
28-29 November, Las Vegas, U.S. (info & tickets). Use Josh’s code for 20% off: REMJHARDMAN20.
Horizons NW
1-3 December, Portland, U.S. (info & tickets). Use PALPHA-NW-15 for 15% off.
Psychedelics and mental health (Seminar)
7 December, Imperial College London (Zoom registration)
Rapid-Acting Mental Health Treatment Summit
7 Jan, 2024, San Francisco, U.S. (info & tickets).
Usona Institute is “preparing to launch a larger phase 3 study early next year”, the article states.
“The new industry is generating so much interest in psilocybin that some centers have indefinite waitlists of thousands of people.”
“Psychedelic drugs (or any drug), should not be treated as exceptional and more viable for decriminalisation or legal regulation simply because they are perceived to present lower risks than other drugs, be therapeutically more useful, or be somehow more spiritually significant.”
Our monthly Psychedelic Patent Analysis provides headline figures regarding published psychedelic patent applications and grants, before diving into commentary on a select handful of patent-related actions.
Note: the monthly Psychedelic Patent Analysis is a Pα+ subscriber benefit (join today). Given that this is a new format, we’re making this first issue available without a paywall.
“A group of activists said the campaign printed a labor union logo on their ballot sheets, violating signature-gathering regulations and potentially invalidating thousands of signatures.”
“A Massachusetts campaign says it has overcome a signature collection mishap and will be submitting more than enough new petitions to force legislative consideration of a psychedelics legalization initiative before potentially putting the issue on the 2024 ballot.”
“In this episode, Josh Hardman, the founder of Psychedelic Alpha, explores the multitude of challenges currently being encountered by the psychedelic industry. He engages in discussions regarding the progress of decriminalisation and legalisation initiatives, upcoming clinical trial milestones, and delves into the current state and the potential evolution of the Psychedelic industry.”

We published two mini interviews this week:
“This will be the last issue of the Dose”, reads the top of Tiffany Kary’s latest issue of the newsletter that has covered the cannabis and psychedelic industries for two years.

“The state of New York fined five Medicaid managed care plans a total of $2.6 million for denying claims or failing to pay for specialty behavioral health services.”
Here’s Governor Hochul’s announcement.
“A new coalition of healthcare professionals wants a review of ‘overly restrictive’ [psychedelics] prescribing guidelines, but a GP expert says changes must be done ‘slowly and carefully’.”

This Issue of the Bulletin focuses on various policy-related developments that have materialised in the past few weeks. These include inaugural hearings in the U.S. House, subcommittee meetings and forums, as well as the publication of reports and key changes to major psychedelic policy reform efforts.
In this Issue:
Read the 5,000+ word Issue.

While some aspects of the Natural Medicine program, like cultivation and testing requirements, are more or less settled, the Board is still wrestling with some difficult questions.
The Board is attempting to strike a delicate balance between regulatory safety and program sustainability, a task that may turn out to be the most difficult one of all.
Read the latest via our Bulletin, written by Jeff Fitzgerald and Vicente LLP.
“Based on our assessment of the literature to date, there is still much to learn, and much yet to be understood, about the potential benefits of psychedelic compounds. Our department is not only focused on finding the best innovative treatments and cures but doing so safely.” – VA assistant undersecretary for health, Carolyn Clancy
Regarding MAPS’ Phase 3 program, Clancy suggested the VA’s main concerns are related to a relatively small sample size and even smaller number of veterans included.
“Waterloo Regional Police say they raided a magic mushroom store on King Street in the Preston area of Cambridge on Tuesday.” The shop was reportedly part of the ‘FunGuyz’ chain.

Here, we sketch out what a psychedelic Risk Evaluation and Mitigation Strategy (REMS) might look like, which should go some way to highlighting how these programs could have an outsized impact on the post-approval delivery and accessibility of these treatments.
“I want us to be Patagonia, not Zara”, Beckley Waves’ Dan Love said, referring to NueCo Holdings PBC.
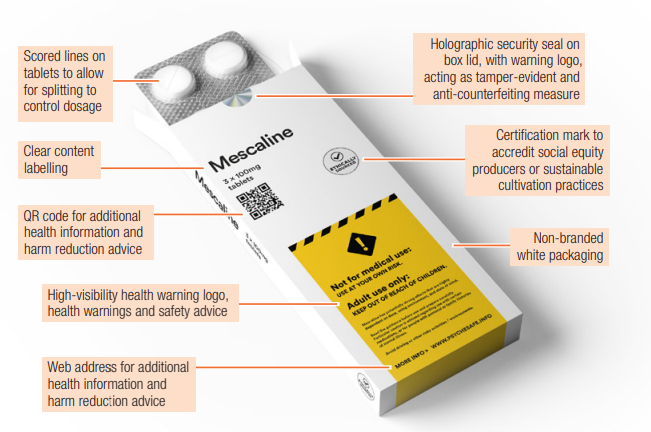
Transform’s Practical Guide is now available to download. Consider ordering a hard copy to support their work.
“[A] challenge is talking to people and saying, ‘We give fish psychedelics and then look at the responses,’” Arinel said. “They ask, ‘Why? Why are you giving tiny little fish larvae psychedelics? What’s the point?’”
Other outlets, including MindSite News, covered the hearing. Our Pα+ Policy Bulletin (149) offers our analysis.
“A growing number of states and cities are legalizing the use of psychedelic fungi in therapy. Some experts think that’s a big risk.”
King’s College London also shared news of the centre’s opening.
“With a strong cash balance of $209M, we are well positioned to continue advancing our clinical programs towards key data milestones”, said CEO Florian Brand.
“Nearly 200 growers submitted 511 mushrooms for sampling at the competition earlier this month at Mile High Station in Denver — far above organizer Jonathan Cherkoss’ expectations.”
“Psychae Therapeutics, which will receive the $4.5 million syndicated investment, is exploring the development of medical-grade, botanically derived treatments and psychedelic-assisted therapies to treat serious mental health challenges such as PTSD, anxiety and addiction.”
“More drugmakers are seeking to harness the medical potential of psychedelics for treating depression, addiction and other hard-to-treat conditions”
WEEKLY POLL RESULTS
We Asked: In 2033, this molecule will have approvals for the most indications in the U.S.:
You answered:
INTRODUCING Pα+
Your one-stop susbcription for psychedelic news, analysis and interviews. Learn more and join.

Pα+ subscribers can access an enhanced version of this article.

The company announces “a firm commitment underwritten offering” of around 67 million units at a price $0.45 per unit for gross proceeds of $30m. Up to an additional $34m could be raised if warrants (priced at $0.51) are exercised.
“Senator Wiener to work with Republicans on a limited therapy-only focused bill”
The AIER, a ‘conservative and libertarian free-market think tank’ published a short article on psychedelics for mental health illness.
The bent of the article is that “it’s time for the FDA to decide” whether to ‘generate an approval process for psychedelics.” But this seems to hinge on a misunderstanding of FDA’s draft guidance on clinical investigations involving psychedelics which does not seek to establish a separate approval pathway for psychedelics.
by Rick Perry.
Pα: Might the fall-out from yesterday’s elections change the odds of success for this effort? If the changing of the guard reaches the ranks of the KYOAAC, such as Bryan Hubbard, the ambitious $42m appropriation could be dead in the water. ∎
“…the Subcommittee is recommending the immediate implementation of a robust research program funded by VAC and the Department of National Defence (DND) in partnership with Health Canada, the Canadian Institutes of Health Research, and all other relevant partners.”
“This is an edited extract from Transform’s How to Regulate Psychedelics: A Practical Guide – by Steve Rolles and Ester Kincová – now available to buy in print. This will be published on Nov 14th! Register for the launch webinar here.”
This is the first issue of our Psychedelic Bulletin that’s exclusively for Pα+ subscribers. Head to our Join page to learn more.
This issue’s contents:
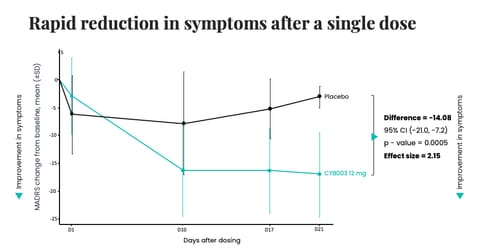
Pα Take: It’s worth stressing once again that this is a small N study, and today’s interim data reflects a subset of that already-small N. Nonetheless, the data appear positive, with a substantial mean MADRS score reduction and safety profile that gives other psychedelic candidates a real run for their money. It now remains to be seen whether these results are reflected in the full Phase 2a data and ultimately sustained in a larger Phase 3 program. With plans to commence a Phase 3 program as early as Q1 2024, along with a Phase 2 study of its CYB004 deuterated DMT candidate for generalised anxiety disorder, it’s going to be a busy couple of quarters for Cybin staff and investigators! ∎ ↗ Psychedelic Alpha
There’s little information about the Friday morning incident: chiefly, whether it’s an accidental car crash or intentional ram-raid (or a pure act of malice, etc.).
Remember that, because they’re operating outside of the law, stores like Shroomyz are often relying on cash for transactions.
The neighbouring store, which has been forced to close, wrote on Instagram that “someone drove their truck into [Shroomyz…] for what I’m sure are some pretty nefarious reasons.”

The striking image was posted by u/Key-Suggestion-1377 on the Toronto subreddit, and is now reported in a number of local outlets.
It’s not the first time this has happened. Back in September, a car was crashed into another Shroomyz store on Dunlop Street West just after midnight on a Wednesday.
Based on images, the crash wasn’t quite as destructive as yesterday’s RAM truck.
WEEKLY POLL RESULTS
We Asked: Do you consider ketamine a psychedelic?
You answered:
Christian Greer and Erik Davis are running a two-week summer school intensive at University of Amsterdam next year. The Psychedelic Universe: Global Perspectives on Higher Consciousness will provide “an in-depth exploration of psychedelic consciousness across space, time, and culture”.
Our friends at BrainFutures are co-hosting an event in San Francisco on January 7th, 2024.
“The ibogaine announcement caused additional controversy. It’s an experimental drug, and, if approved, the $42 million allocation would be the single-largest investment from the commission, which is housed in Cameron’s agency.”
The company will conduct a Ph 2b trial of its psilocybin candidate, RSTP1000, in demoralisation syndrome.
“Eco-anxiety and climate grief are unlike other issues in psychiatry because the feelings extend to something greater than ourselves and our personal narrative.”
“Longtime pot activist Dana Larsen opened the stores on East Hastings, West Broadway and Granville street starting in 2019”
“Larsen claims his operations are “under attack by the city,” and is urging supporters to attend a public hearing about the business licences on Dec. 6 at city hall.”
Over in California, Bakersfield man arrested in illegal marijuana dispensary bust on California Avenue: BPD (Oct 31) ↗ Bakersfield Now
Mattha Busby profiles the Alexander Shulgin Research Institute.
Usona Institute’s scholarship program supports practitioners seeking training or certification in psychedelic-assisted therapy and emerging scholars engaged in post-graduate degree programs focusing on psychedelics.
Deadline: November 3.
↗ AP News also has the story.
“Products in the works at various pharmaceutical companies aim to make ketamine and other dissociative drugs, such as psychedelics like MDMA, even better antidepressants. Some drugs combine ketamine with other medicines to extend its efficacy and reduce the need for such frequent doctor visits. Other research explores ways to cut the hallucinogenic effects from ketamine and psychedelics while holding onto the antidepressant effects.”
Ed Prideaux investigates HPPD.
TREAT California, which had hoped to make its way onto the state’s 2024 ballot and ultimately convince voters to endorse a $5bn psychedelic R&D institute, has withdrawn the project.
In a statement, the organisation said that while voters “have expressed widespread support for the initiative’s objectives”, polling “indicates they do not yet believe in creating a state agency to fund the program.”
Polling conducted by FM3 Research revealed striking political cleavages (which we discussed in Bulletin 147), with the average Californian independent or Republican opposing the measure.
The group announced a newly-formed nonprofit, TREAT Humanity.
WEEKLY POLL RESULTS
We Asked: When will MDMA-assisted therapy be approved by FDA?
You Answered:
“The court documents said he also told police that he had taken “magic mushrooms” for the first time, ingesting them about 48 hours before boarding the plane.”
The story has caused quite a stir, with mainstream media covering it widely and psychedelic advocates bracing for backlash.
Perhaps relatedly, a union representing many U.S. flight attendants (the Association of Flight Attendants-CWA) issued a ‘Know the Facts’ piece about microdosing psychedelics. It’s strange they chose to focus on microdosing.
“Organizers have collected more than 75,000 signatures on a ballot initiative to legalize psychedelic mushrooms.”
The DEA failed to provide adequate justification for its denial of a psilocybin rescheduling petition put forth in the AIMS v DEA case, the U.S. Court of Appeals for the Ninth Circuit court has found. The ruling does not send the petition to HHS for review, however. Rather, it orders DEA to provide an adequate justification for its denial.
Beckley Waves co-founder Daniel Love will act as interim CEO of NueCo Holdings PBC, a newly-formed company that will acquire the assets of Nue Life Health Inc.
“The state has pioneered a therapeutic market for psychedelic mushrooms. Researchers are watching with a mix of excitement and unease.”
As reported last week, a $16m donation from the Gracias Family Foundation has launched a new interdisciplinary program. The Harvard Crimson shares a little more about the initiative.
Blunt is a key proponent of drug policy reform, including psilocybin rescheduling, in the UK.
“Juniper Preserve, a 36-hole resort in Oregon, is applying for permission to conduct research on the performance effects of psilocybin on golfers, which is all to say, the term golf trip could be on the verge of acquiring new meaning.”
The proposed ballot measure, the “Psychedelic Wellness and Healing Initiative of 2024”, would legalise the production, use and – eventually – sales of psychedelics including psilocybin, MDMA, DMT, ibogaine, mescaline and LSD.
The draft text of the ballot measure can be read here.
“Consumption of this substance has reached its highest point since 2017 and is leaving a mark on popular culture. Some defend its therapeutic power, but others warn of its destructive potential”
Otsuka has completed its acquisition of Mindset.
50-minute radio segment, including clips from Psychedelic Science 2023.
50-minute radio segment, including clips from Psychedelic Science 2023.
Beckley’s Phase 2b trial aims to enrol 225 patients across 40 sites in 6 countries. Initial results are expected toward the end of next year.
“A lack of commercial incentives to get the drug approved for treatment-resistant depression means people cannot afford it”
The Australian Capital Territory has now decriminalised small quantities of drugs including LSD, MDMA and psilocybin.
(We first reported the decision in October 2022.)
and lots more.
On Monday evening Roland Griffiths, a pioneer in psychedelic and consciousness research, passed away.
Professor David Nutt shared:
A sad day: my longtime friend and colleague Roland Griffiths died last night after a long battle with cancer during which he showed the inspiring courage and wisdom that characterised his whole career, especially in resurrecting psychedelic research. A truly great scientist.
An interview between Roland and his wife, Marla, and Manish Agrawal displays the pair’s remarkable sense of perspective on Roland’s diagnosis and beyond.
Beyond his immense impact on the present field of psychedelic and consciousness research, his legacy includes The Roland R. Griffiths, PhD, Professorship Fund in Psychedelic Research on Secular Spirituality and Well Being. The Fund establishes (and aims to support, in perpetuity) a world-class psychedelic research program to advance human flourishing and wellbeing.

The ↗ New York Times remembered Griffiths in an article that charts his contributions.
…as did the ↗ Baltimore Banner.
↗ Johns Hopkins shared a tribute to Griffiths, too, which noted that the endowment is at $24 million.
Sponsor the Psychedelic News Feed.
We will soon be opening a limited number of sponsorship opportunities for the Psychedelic News Feed. Get in touch to discuss.
Announces First Patient Enrolled in FDA-authorized Study Evaluating Low-Dose Psilocybin in Human Subjects with Demoralization (Oct 17) ↗ Press Release
The trial hopes to enrol 60 patients to evaluate low-dose psilocybin in demoralisation (which includes feelings of hopelessness and meaninglessness). Diamond does not expect the doses to produce subjective effects.
Pα: Given the dearth of clinical trials evaluating ‘microdosing’, and the equivocal nature of observational studies regarding the practice’s effects (or lackthereof), this should be an interesting study to follow (speaking of which, here’s the trial registry entry: NCT05227742). However, it is worth pointing out that the trial’s dosing protocol – which sees five drug (or placebo) administrations over as many weeks – is dissimilar to popular microdosing regiments. ∎
Reporters at The Denver Post want to know what “burning questions” its readers have about psychedelics in Colorado. (You can also let us know, and we will make sure to cover it in our Colorado Natural Medicine Health Act Tracker, in collaboration with Vicente LLP.)
“Donna Dryer has admitted to conflict of interest and boundary violations in MDMA-assisted therapy study”
Johnson and Johnson’s Q3 2023 results show $183 million in worldwide sales (the vast majority of which were in the U.S.) of Spravato in Q3 2023 vs. $100 million in Q3 2022.
A gift from the Gracias Family Foundation supports the launch of an interdisciplinary effort across the Faculty of Arts and Sciences, Harvard Law School, and Harvard Divinity School.
Quartz also covered the news: A former Tesla director is giving Harvard $16 million to study psychedelics in society and culture
The mental health focused accelerator supports companies at varying stages with a $100k investment and 9 weeks of intensive programming. More info and apply.
“Can toxic company culture be cured with mushrooms? A growing movement of leaders in the business world are willing to give it a shot.”
An attempt to roll back Australia Capital Territory’s decrim. law has been defeated 33-27.
“You can’t take drugs at Athenaeum—but you can talk all about them.”
“Now there’s Super Mario Bros. Wonder, which turns the franchise into a carnival of bizarre delights. Each level includes a wonder flower, a new mechanic that sends Mario on something like a psychedelic trip, reconfiguring the world around him — warp pipes start crawling like caterpillars, rhinos begin a stampede and some mountains gain a set of googly eyes.”
WEEKLY POLL Results
Q: Should Governor Newsom have signed SB-58 to decriminalise certain psychedelics in California?
A: Unsurprisingly, the ayes have it! 80% of you voted yes, with just 5% voting no. 15% of you sat on the fence, answering not sure.
More to come in our next Bulletin.
“Alejandra Lagunes says that psilocybin mushrooms could be a benefit to the country’s mental health crisis.”
The latest issue of History of Pharmacy and Pharmaceuticals revolves around the broad topic of ‘psychedelic capitalism’, and is edited by Neşe Devenot and Brian Pace.
Open Access articles include Matthew Baggott’s rich review of the STP crisis and Shams et al.’s look at the role prior art libraries (in this case, Porta Sophia) can play in the psychedelic patent landscape.
A 30-minute interview with Andy Mitchell, who just published Ten Trips.
This piece provides a summary of a (well, the) psychedelics panel at HLTH in Las Vegas.
“Coming into the sixth month of work, Colorado’s regulated natural medicine program is starting to take shape”, writes Jeff Fitzgerald of Vicente LLP for the latest update from the state’s Natural Medicine Advisory Board.
While comments on ‘giving MDMA to kids’ have caused some hoopla on the conference circuit, if MDMA-assisted therapy is approved in adults then exploring it in adolescents may be the next step on the clinical research agenda. Two researchers published a review article on the topic today, Tuesday 10th (they published a preprint in the Summer).
See our earlier coverage of MDMA-AT’s potential pediatric studies.
“While abandoning mainstream religious affiliation, many turn to alternative expressions, including secular, atheist and psychedelic churches.”
JHU is exploring a reboot of its postwar science TV show, “The Johns Hopkins Science Review”, on a platform like Netflix, and psychedelic research could get airtime: “Psilocybin research and the groundbreaking work of the School of Medicine’s Center for Psychedelic and Consciousness Research is another topic of interest”, the article notes.
NEW JOB LISTINGS
Via our Psychedelic Job Board
Manager/Sr. Manager Medical Information – MAPS PBC
Research Officer – Clinical Psychedelic Lab – Monash University
Director, IT – COMPASS Pathways
“There’s a growing movement among religious leaders to use psychedelics like psilocybin to deepen their faith. The author attended a secret ceremony to find out whether it works.”
Pα: (We will be covering this more extensively in a forthcoming Bulletin – sign up today.) The Governor’s veto is not entirely surprising, given Newsom’s rumoured national political ambitions. Newsom’s associated letter (which is not something a Governor has to accompany a veto with) doesn’t slam the door closed on psychedelic policy reform, either. But it’s clear Newsom would like to see something more closely focused on ‘therapeutic use’, though the term is admittedly vague. Expect to see Sen. Wiener return next year with a new Bill. Our Editor-at-Large Graham Pechenik and I (Josh Hardman) spoke to Wired about the news. ∎

Pα: 3 years ago, Mydecine Innovations Group appointed advisors for a ‘planned listing’ on the London Stock Exchange. That never happened, but earlier this week the company was admitted to the AQSE growth market. Since then, not a single share has been traded. ∎
“Psychedelics are hot in the field of mental health!”, Tweeted the European College of Neuropsychopharmacology alongside a photo of a packed conference hall.
Pα: Our own Josh Hardman attended ECNP’s New Frontiers meeting in France earlier this year, which was entirely dedicated to psychedelics (read our write-up). It’s great to see this interest borne out at the main conference, too. ∎
Videos of presentations at MAPS’ Psychedelic Science 2023 conference are being made available via a dedicated website. Josh’s State of the Psychedelic Sector presentation is among the available recordings.
“Back then, they were associated with a hippy, psychedelic counterculture, and their popularity was given a boost not only by Starr but also Paul McCartney, who had lava lamps on stage with his band Wings, and David Bowie, who was photographed with one in his recording studio.”

Alexis Goosdeel, Director of the European Monitoring Centre for Drugs and Addiction, will present a talk titled, “From entertainment to therapeutic use: effectiveness of psychedelics and cannabis for healthcare”.
“Drugs such as psilocybin can trigger dangerous psychotic experiences and should only be used with medical supervision, warns psychiatrist Jonathan Iliff at New Scientist Live”
Pα: A number of researchers that work with psychedelics have shared concerns around topics like decriminalisation and ‘self-medication’ of late. In response to a WaPo Opinion piece titled “Self-medication with psychedelic drugs is a dangerous plan”, Robin Carhart-Harris said, “I tend to agree”. Jennifer Mitchell, meanwhile, told the LA Times she was against California’s decrim. bill. ∎
Is ibogaine R&D an appropriate use of the state’s Opioid Abatement Advisory Commission funds? A $42m grant would be its biggest to date, by a long shot.
Cameron’s ibogaine initiative “came out of the blue” and “caught everybody off-guard”, according to a source in a Daily Beast exclusive, which alleges conflicts of interest.
Pα: Just as psychedelic advocates question Newsom’s motives for vetoing SB 58, this reporting questions the motives behind KY’s potential ibogaine funding. Opposition researchers have requested ibogaine program-related records from Cameron’s office, so there may be more to come. ∎
Health Canada has given the go-ahead for a Filament-sponsored study of psilocybin for the treatment of opioid use disorder. The Phase 2 trial will take place at University of British Columbia
Pα: In a crowded synthetic psilocybin drug development space, Filament is looking to differentiate itself by using a ‘botanical’ psilocybin candidate. ∎
The case report details how a 49 year old immunocompromised male ended up with a fungal infection in his lungs.
The patient explained that he had cultivated his own psilocybin mushrooms and turned them into a powder using an electric coffee grinder.
“…just three blocks from Grand Central Terminal, tucked within the sprawl of advertising offices, finance firms and chain stores, a community center dedicated to consciousness-altering substances has taken root.”
Free subscribers receive a weekly round-up of news on psychedelics, as well as the occasional article.
Receive regular bulletins, articles, interviews with insiders and quick-take analysis of major stories and developments.
Teams, groups and corporate pricing plans are available, please get in touch via email to learn more.
Front-row access to the psychedelics space: breaking bulletins, in-depth articles, insider interviews and sharp analysis of the moments that matter—from major trial results and funding rounds to policy shifts shaping the future. Plus, full access to our complete archive and Library.
Learn More| Free | ||
|---|---|---|
| Weekly Psychedelic News Feed | ||
| Occasional Articles & Free Resources | ||
| Psychedelic Bulletins (In-Depth Briefings, Multiple per Month) | ||
| Quick-Take Analysis of Major Developments | ||
| In-Depth Articles & Deep Dives | ||
| Exclusive Interviews with Insiders & KOLs | ||
| Quarterly Video Briefings | ||
| Exclusive Tools & Data Resources | ||
| Library of Primers & Explainers | ||
|
Subscribe Now |
||
Independent data-driven reporting, analysis and commentary on the psychedelics space: from business and drug development through to policy reform and research.
By signing up, you agree to our privacy policy. You can unsubscribe at any time.
Aside from group pricing, we also offer bespoke reports and regular briefings. Get in touch to discuss.